Abstract
Polymer latexes are essential components in a wide range of commercial products and formulations such as, paints, cosmetics, coatings, biotechnology, and functionalized supports. Many difficulties are intrinsic to the implementation of polymer latex research, particularly in the purification of the final latex dispersions and the control and reproducibility of particle size, therefore making high-throughput research in this area especially challenging. In this article, we demonstrate how the investigation of the influential synthesis factors on polymer latex materials properties can be swiftly and reproducibly achieved by the combinational use of experimental design, automated synthesis, and a newly developed high-throughput purification process. Through the implementation of flexible automated platforms, a significant increase in the throughput of this previously manual process was achieved. Reaction models were used to examine the synergistic and antagonistic effects of the latex synthesis parameters, thereby allowing the controlled synthesis of fully characterized libraries of surface–functional polymer latexes to be rapidly produced and screened for a wide range of applications.
Keywords
Introduction
Polymer latexes are essential additives to many industrial and commercial products in the coatings, adhesives, and cosmetic industries. The most common and scalable synthetic routes for the production of polymer latexes are via dispersion, emulsion, and miniemulsion polymerization. 1 –4 In the current note, we disclose a flexible process to conduct high-throughput investigations into polymer latex synthesis via dispersion and emulsion polymerizations that have been developed using a Chemspeed SLT 106 Accelerator Synthesis Platform 5 (Chemspeed Technologies AG, Rheinstrasse, Switzerland) (see Fig. 1). This automated platform uses variable sizes of parallel reactor arrays that are temperature controlled and can be loaded with the desired quantities of liquid and solid reagents using the onboard automated dispensing tools while using controlled mixing and an inert atmosphere. The process uses design of experiments (DoEs) 6 to produce a statistically varied work list of experimental recipes to examine the synergistic and antagonistic effects of reaction conditions (temperature, reaction time, reactor size, surfactant, percentage reaction solids to reaction volume, etc) on the physical properties of the final polymer latex, that is, molecular weight, particle size, glass transition temperature, etc. The results of these experiments can be used to produce a synthesis model, which can then be subsequently used to predict optimized reaction conditions to produce latexes with the desired physical properties.

Chemspeed SLT 106 Accelerator Synthesizer layout to conduct HT latex synthesis investigations via experimental design, (a) XYZAlpha cartesian robotic track arm, (b) adjustable spacing septa piecing four-needle liquid-handling tool, (c) gravimetric solid dispensing tool, (d) four independent multivalve 10-mL syringe pumps, (e) bulk reaction and washing reagent storage, (f) 13-, 27-, and 75-mL temperature-controlled synthesis reactors arrays, (g) monomer and initiator reagent storage and final product sample collection vials.
A common problem encountered in polymer latex synthesis concerns the difficulties arising during their purification, where the residual surfactant or stabilizer is removed from the final latex suspension. If these surfactants are not removed this may have undesirable effects on the final application products to which they are added. There are various techniques used to remove these unwanted contaminants each of which have advantages and drawbacks. Filtration, although scalable, can easily be blocked especially if the latex forms a film on drying, such as those used for coating applications. Dialysis is both slow and hard to implement on scale up and chemical methods to remove surfactants can cause damage to some latex materials and may still result in residual impurities. Therefore, the less damaging but still scalable process of using multiple centrifugation, decantation, washing, and redispersal steps to remove the impurities was chosen as the favored purification method. The downside to this method is that it is a very laborious and labor-intensive process and a clear “bottle-neck” in the research workflow and for that reason a clear target for the beneficial application of automation. Therefore, a high-throughput latex purification and analytical sample preparation process was also developed to allow the removal of this bottleneck to the production of fully characterized libraries of functionalized polymer latexes in a high-throughput parallel fashion.
Materials and Methods
High-Throughput Parallel Latex Synthesis Process
Dispersion and emulsion polymerization reactions are influenced by a number of key reaction parameters. 7,8 We therefore used multiple “onion layer” DoE models to identify and quantify these influential synthesis parameters used during the production of latexes over a range of reactor sizes. One such investigation model is discussed below and was used to measure the effects of shear on the synthesis of latexes across different reactor sizes (13–75 mL) by using increasing vortex mixing speeds. By using repeat reactions across the reactor array as part of the experimental design, this allows an assessment of the reproducibility and control of latex weight average molecular weight (M w), number average molecular weight (M n), and hydrodynamic particle radius (R h) to be examined over a range of reaction scales.
The fully automated reaction recipe follows a standard dispersion polymerization protocol, which commences with the cleaning and drying of the reactors under an inert atmosphere. This is followed with the automated addition of the required amount and ratio of monomer, solvent, and stabilizer/surfactant, which is delivered to the reactors using the liquid and solid dosing tools. The program then heats the reactors to the desired reaction temperature and mixing speed. Finally, the correct volume of polymerization initiator is added at the desired addition rate to the reaction mixture and the final latex product collected at the end of the set reaction time. To minimize cross-contamination, both inside and outside of the needles of the liquid-handling tool are washed with solvent between reagent dispensing and latex product sampling.
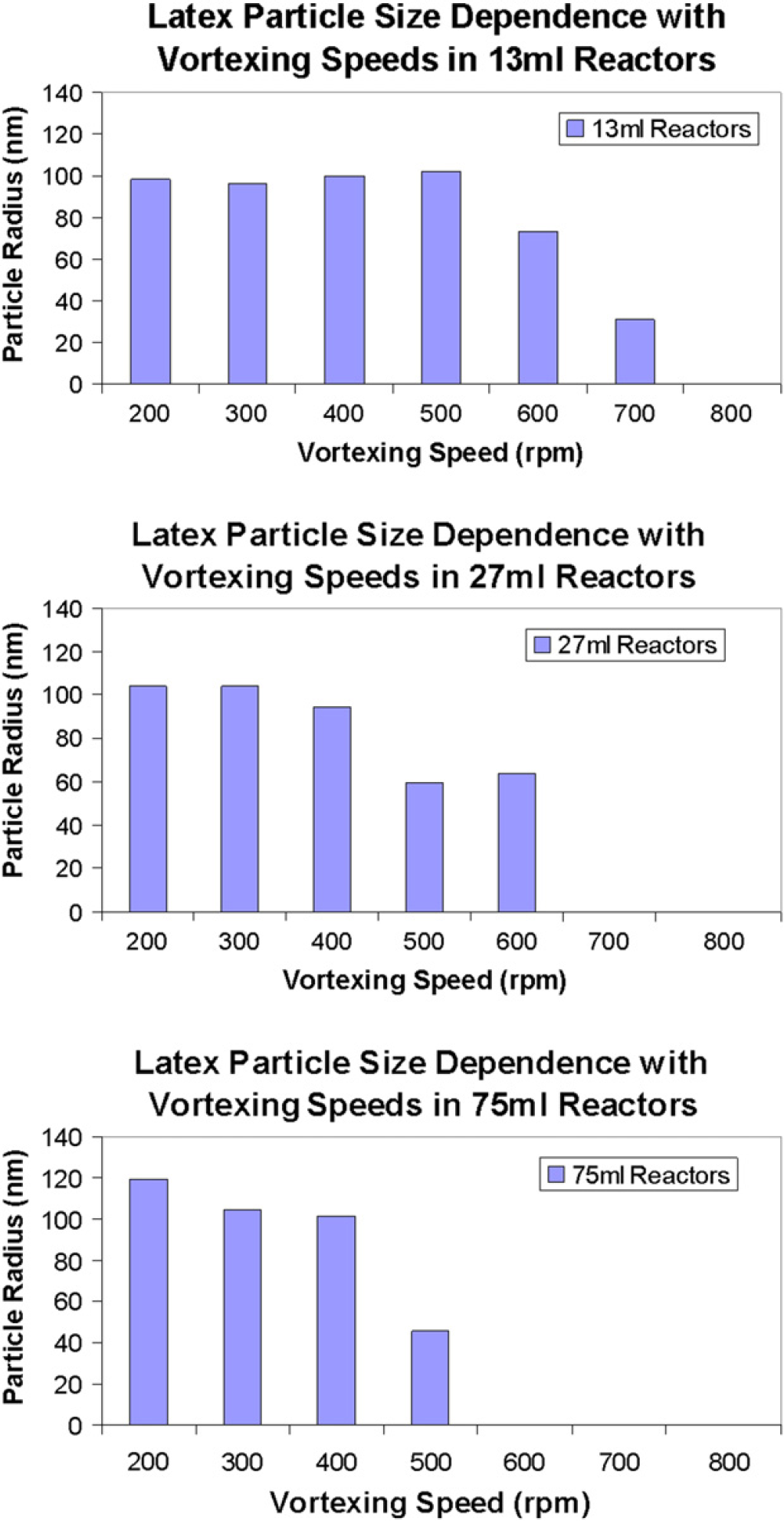
The final latexes produced in 13-, 27-, and 75-mL reactors at increasing vortexing speeds as shown in Figures 2 and Figures 3 (200–800 rpm left to right) show that the higher vortexing speeds cause the reaction to destabilize the gas/liquid interface and undergo “reactor fouling” by coagulating rather than forming a uniform latex dispersion. 9 It can also be clearly observed that this effect is exaggerated as the reactor size increases and thereby highlighting the influence of mixing speeds and reaction scale on the synthesis of latex dispersions.
Polystyrene latexes, including repeat reactions to measure reproducibility, were synthesized via a dispersion polymerization in 13-, 27-, and 75-mL reactors (left, center, and right images, respectively) at increasing vortexing speeds 200–800 rpm (from left to right).

Graphical illustration of the effects of vortexing speed and reactor size on the hydrodynamic particle radius of the latex dispersion (13-, 27-, and 75-mL reactors as left, center, and right graphs, respectively).
A similar trend in the dependence of mixing speed and reactor size on the latex particle size was also observed when applying the same reaction parameters during an evaluation of the automated mechanical stirred 100-mL mini plant reactors, 5 shown in Figures 4.

Independently mechanically stirred and heated 100-mL automated mini plant reactors with online temperature and pH monitoring and multiple reagent feeds for process optimization research.
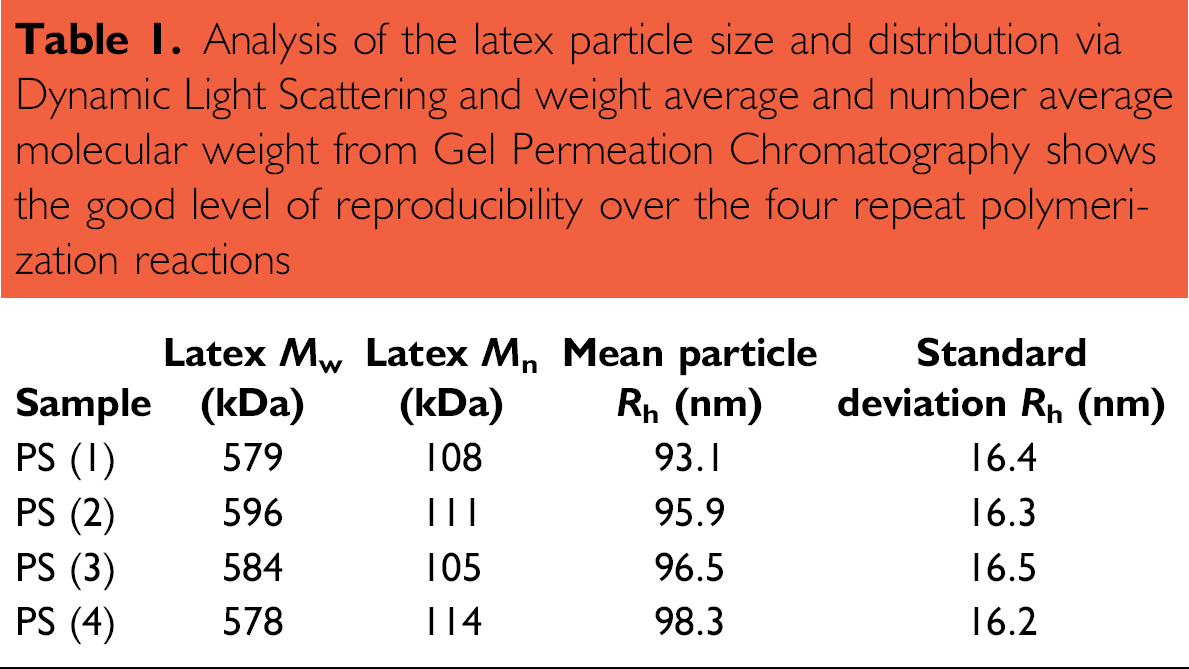
The evaluation of the reaction process showed good reproducibility of R h, M w, and M n in the final unpurified latex dispersion (see table 1 and Fig. 5). To gain further confidence in the robustness of the polymerization process and the automation equipment, the repeat reactions were also randomly distributed across the reactor array.
Analysis of the latex particle size and distribution via Dynamic Light Scattering and weight average and number average molecular weight from Gel Permeation Chromatography shows the good level of reproducibility over the four repeat polymerization reactions
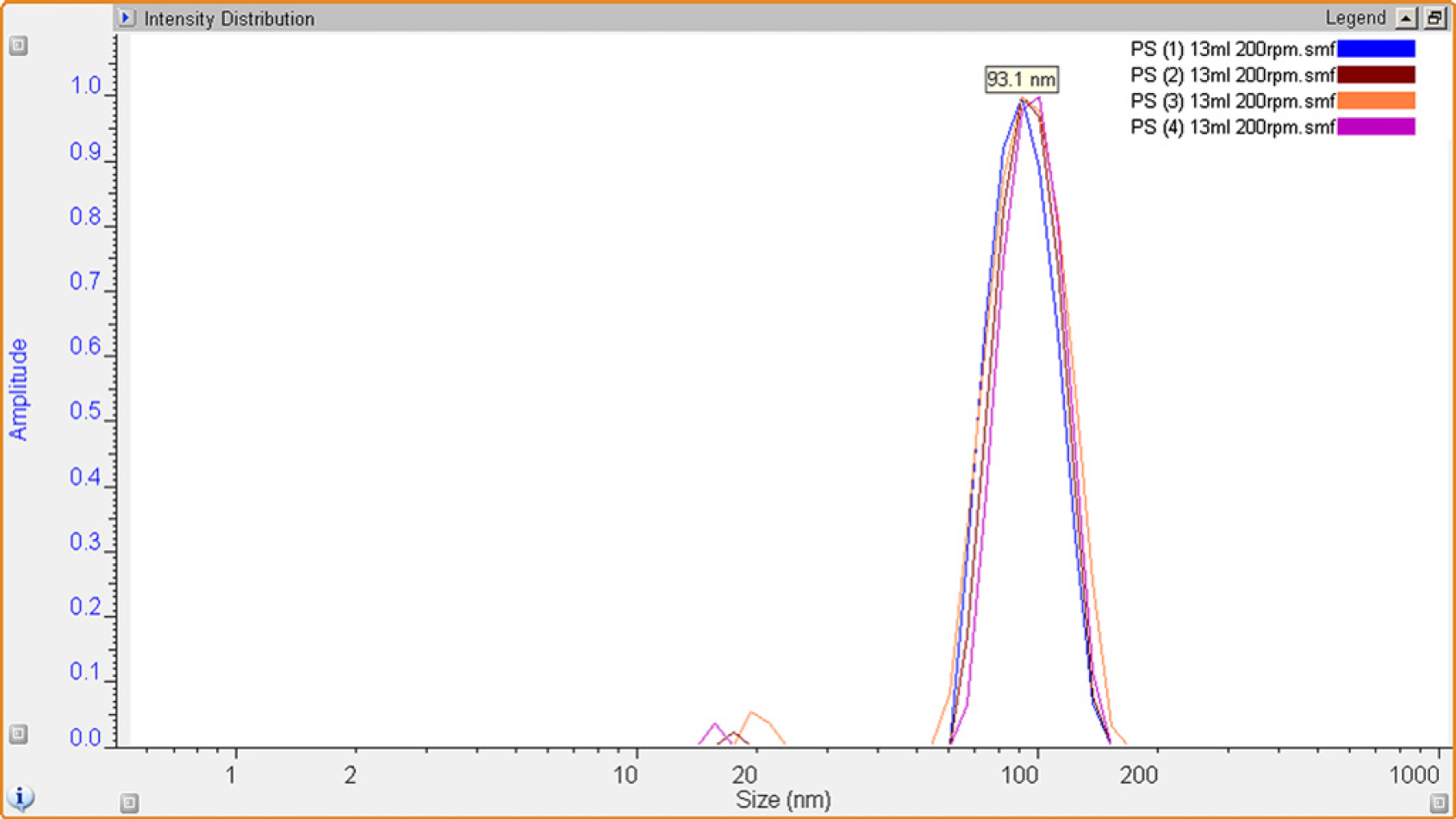
Dynamic Light Scattering (DLS) analysis showed the process to give good reproducibility in both particle size and distribution (R h) of the crude unpurified repeat latex polymerization reactions. Small impurities in the final latex dispersions caused by residual stabilizer, surfactant and unreacted monomer can also be clearly observed.
High-Throughput Batch Latex Purification Process
Once synthesized the latex samples are then fed directly into an automated purification process in batches of up to 96 samples. These are first centrifuged before using the Infrared level detection camera on an Eppendorf epMotion 5075LH liquid handler (Eppendorf North America, Inc., Hauppauge, NY) 10 to measure the liquid level in each centrifuge tube thereby allowing the careful removal of the supernatant liquid without redispersing the latex, thus extracting excess surfactant and unreacted monomer (Figs. 6–8). Fresh

Eppendorf epMotion 5075PC liquid-handling platform layout to conduct HT purification of polymer latexes, (a) sample vials containing crude latex directly from the automated synthesis platforms, (b) centrifuge tube arrays, (c) sampling plates for HT particle sizing and surface tension, (d) washing reagent reservoirs, (e) sampling vials for HT Gel Permeation Chromatography (GPC) polymer analysis, (f) final product library storage plate.

The latexes are centrifuged in batches of 48 samples for 20 min (left) and the sedimented samples returned to the liquid handler (center). The process if also scalable for samples from larger reactor sizes (right).
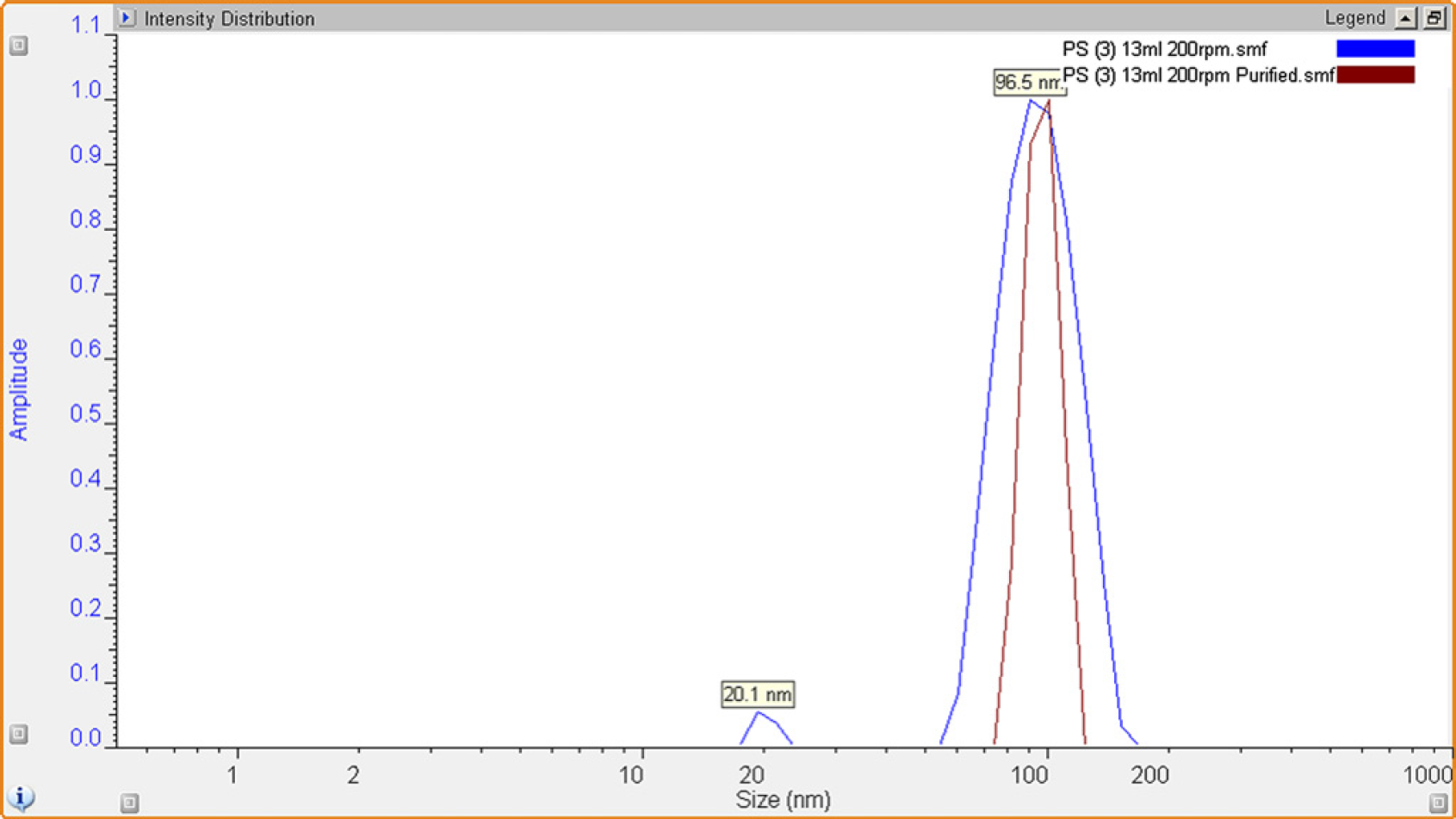
Particle size analysis via Dynamic Light Scattering (DLS) (left) shows the particle distributions of a latex sample before (blue) and after (red) undergoing 10 purification washings. Also, the removal of small reaction impurities can clearly be observed.
Medium is then added and the polymer resuspended before the process is repeated until the polymer is purified. To ensure the stabilizer or surfactant has been fully removed by the repeat washings, a sample of the supernatant is rapidly tested using a Kibron Delta-8 surface tension plate reader and compared to the surface tension of the pure solvent medium. 11,12
By building in process flexibility through multiple platform compatibility of working formats, this allows the samples to be effortlessly delivered from the automated synthesis platforms and placed directly onto the deck of the liquid-handling platform where the centrifuged samples can then be rapidly prepared and processed.
We conservatively estimate this process accelerates latex production over 50 times faster than traditional manual methods and is currently scalable under automation up to 100 mL allowing researchers to rapidly produce fully characterized libraries of functionally diverse polymer latexes on both research and industrial test scales as shown inFigures 6 and 7. Further increases in throughput can be easily achieved by the simple parallelization of another centrifuge.
Results
Once the latexes have been purified, the process prepares analytical samples for Dynamic Light Scattering and Gel Permeation Chromatography 13 (see Fig. 8).
The final purified samples are then prepared as standardized replicates and stored as a 2D bar-coded library for future high-throughput application screening as shown in Figures 9 and 10.

The final library of multiple replicates of each fully characterized latex sample is stored in robotically compatible 2D bar-coded vials and exported for application screening.

Field Emission Scanning Electron Microscopy (FE-SEM) can also be used to image the latex product to show the reproducibility in particle size and morphology. 14
The results from these experiments are then fed into the DoE software to produce a model evaluating the influential synthesis factors on the physical properties of the resultant latex product (see Fig. 11). A number of these models can be combined to produce a fundamental and quantitative understanding of latex syntheses. This is a powerful tool for predicting and synthesizing new latexes with the predetermined physical properties.
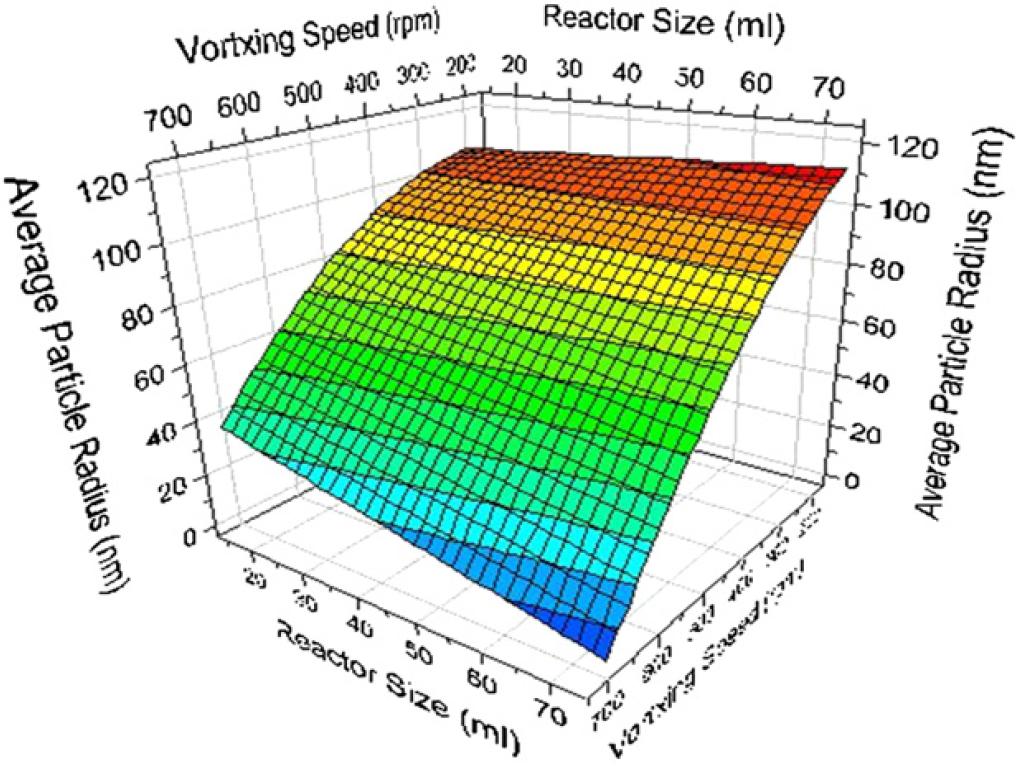
Response surface modeling illustrates the dynamic interaction effects of mixing speed and reactor volume on latex particles size.
Conclusions
We have shown that automation can be used to dramatically accelerate research into commercially significant polymer latex materials. The advantages of using automation for each process step can be clearly shown in the throughput comparison in table 2 below.
Comparison of manual and automated latex sample throughput per process step for each 24-h period for both large and small reaction scales respectfully

Based on the use of common manual parallel synthesis equipment.
Also by combining automation with the implementation of DoE it is possible to efficiently gain an understanding and control of the influential parameters effecting latex synthesis and their physical properties. This allows libraries of diverse functionalized polymer latexes to be rapidly produced for screening for a wide range of commercial applications. It is also believed that these same strategies and processes developed in this project could also be successfully applied to gain similar significant advancements in a number of other research fields.
Acknowledgments
We thank the Northwest Regional Development Agency, and the EU for funding.
Competing Interests Statement: The authors certify that they have no relevant financial interests in this manuscript.
