Abstract
The translation of experimental cell-based therapies to volume produced commercially successful clinical products that satisfy the regulator requires the development of automated manufacturing processes to achieve capable and scaleable processes that are both economic and able to meet the unpredictable demands of the market place. The Healthcare Engineering group at Loughborough has conducted novel demonstrators of the transfer of manual human cell culture processes to the CompacT SelecT (The Automation Partnership) automated cell culture platform, including an osteoblast cell line, embryonic carcinoma cell line, primary bone marrow-derived mesenchymal stem cells, primary umbilical cord-derived progenitor cells, and human embryonic stem cells. The work aims to develop and optimize automated cell culture processes for manufacturing cell-based therapies in a quality system and current good manufacturing practice (cGMP) compliant manner and is underpinned by the application of a six-sigma inspired quality engineering approach.
In this technical brief, we outline the need for automated cell culture systems and automated process engineering for the manufacture of cell populations for therapeutic applications. We review the transfer of a manual cell culture process to an automated process and the subsequent methodology for process improvement using examples from our laboratory of the application of these principles to an important regenerative medicine cell type, the human mesenchymal stem cell. We believe that systematic process improvement methodologies combined with the process stability provided by automation are essential to engineer optimized cGMP compliant manufacturing processes that will be required to realize the promise of cell-based therapies.
Introduction: The Translation of Regenerative Medicine and Tissue Engineering
Regenerative medicine and tissue engineering are fields with major therapeutic potential that aim to use cells or cell and biomaterial constructs to regenerate or replace failed or diseased tissues or organs. The concept isn't new; the field has developed from many disparate lines of research in clinical medicine, engineering, and biological science. New impetus has come over the past decade from a multidisciplinary approach to research and through the discovery of a range of novel potentially therapeutic cell types, particularly niche adult and embryonic stem cells.
Regenerative medicine and tissue engineering are currently in an early translational phase from lab-based experimental disciplines and a nascent industry into a successful industry that can responsively and economically provide a significant range of products to a large market. This transformation is driving an increasing need for robust manufacturing systems for cell-based products to assist the economic and practical feasibility of making emerging therapies by a current good manufacturing practice (cGMP) compliant, a regulated production process. 1,2
Automated cell culture will necessarily be part of a range of strategies used to improve the capability and cost-effective scalability of product manufacture. Cell culture automation is also an enabling platform for the application of further process improvement techniques. Rigorous incremental process improvement systems such as the six-sigma methodology have been applied in many industries to help identify and reduce process variation. Application of these techniques requires automated processes with the scale and stability for large statistically designed experiments.
Historically, automated cell culture has not been significantly used in the cell-based therapy industry due to prohibitive entry costs in a predominantly small business dominated industry. There is also a lack of a clear “trail blazer” that demonstrates the value of automation and a process-orientated approach to the production of a cell-based therapy. This increases the challenge of funding manufacturing process development in a sector where investors have traditionally focused on development of product performance.
The Healthcare Engineering group at Loughborough University is focused on translational research to exploit health care technology, improve the delivery of health care, and facilitate small business growth in its supply chain. An important objective of our work was to provide demonstrators of the use of automation and manufacturing engineering principles in the expansion of therapeutic cell stocks and increase awareness of the value of this approach in the commercialization of cell-based therapies. This article describes the automation of a therapeutic cell culture process and the approach to analysis and process improvement using an important regenerative medicine cell type, primary human mesenchymal stem cells (hMSCs), as an example. hMSCs hold promise in therapeutic areas as diverse as cardiovascular regeneration and cartilage repair.
Methods
The Automation
The Healthcare Engineering group use a CompacT SelecT (Fig. 1) manufactured by The Automation Partnership (TAP) to conduct automated therapeutic cell culture process research. The CompacT is a fully automated cell culture platform consisting of a robot arm that can access a 90 flask T175 humidified incubator or a multiwell plate incubator (inset). Two flask decappers and flask holders, automated media pumping (or pipetting for lower volumes), and a Cedex automated cell counter are also integrated within a laminar flow cabinet allowing most routine cell culture activities such as passage or media changes to be conducted and controlled to a schedule with minimal human interference. The SelecT is a well-established automated system for culture of adherent cell lines for use in pharmaceutical product evaluation. It is an important experimental platform to investigate cell-based therapy manufacture because it allows automated optimization experiments and is a potential production platform.

The Compact Select automated cell culture platform. A—Robot arm, B—Flask incubator, C—Plate incubator, D—Flask decappers, E—Flask holders, F—Media pumps, G—Pipette head, H—Cedex automated cell counter.
Cells, Culture, and Analysis
The primary examples given in this article use hMSC cells isolated from human bone marrow (Lonza) via a histopaque density gradient and subsequent adherence to T175 tissue culture flasks (BD Biosciences). The cell culture media used is 40 mL low glucose DMEM (Lonza), supplemented with 1% Glutamax (Invitrogen), 1% nonessential amino acids (Lonza), and 10% MSC-qualified fetal bovine serum (Invitrogen), unless otherwise stated as an experimental variable. Cell surface marker expression was assessed through flow cytometry (Beckman Coulter Quanta SC) using CD71-FITC-conjugated antibody (Beckman Coulter, clone YDJ.1.2.2), CD105-PE-conjugated antibody (Beckman Coulter, clone 1G2), ALP-PE-conjugated antibody (R&D systems, clone B4–78), and STRO-1-FITC-conjugated antibody (R&D Systems, clone STRO-1). Minitab was used for all statistical analysis and data transformation. The software used multiple regression to fit a general linear model for each response and ANOVA to identify significant (
Manual to Automated Cell Culture Process Transfer
The Healthcare Engineering team has successfully transferred a number of manual cell culture protocols to successful automated cultures on the CompacT SelecT. The objective was to achieve a process platform with the stability to allow the scalable production of cells at the volumes required by therapies and the application of process improvement methodologies. The automation process is therefore designed to mimic the manual process as closely as possible to maximize chances of success and to avoid process modifications based on assumptions about the sources of process output variation. The resultant automated process is required to be reproducible and have low noise to improve the sensitivity of further process optimization. Data from such a system are also more likely to be acceptable to a clinical regulator.
In our laboratory, subtle differences in process output are often found between manual and automated cell culture processes. The importance of this clearly depends on whether the different output falls within the customer (clinical/regulatory) specification. The customer specification for automated cGMP processes for creating therapeutic cell stocks is likely to be more demanding compared to that for the current use of automation to produce cell lines for applications such as drug testing. The automated hMSC expansion process provides an example of these discrepancies as it does not produce the same cell yield with the same characteristic cell marker patterns as the manual process (Box 1). These differences are not apparent through visual cell examination.
A comparison of the performance of an automated and manual cell culture protocol to expand a human mesenchymal stem cell population. Statistical significance of difference in cell numbers,

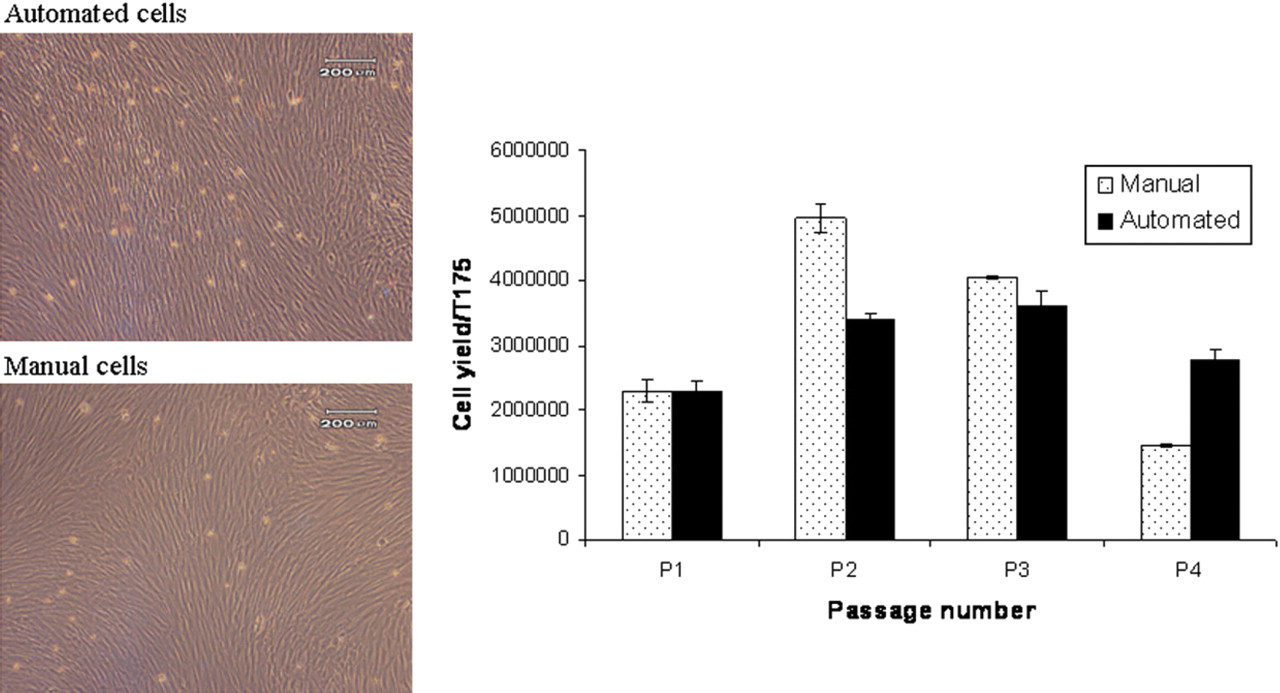
Interrogation of both the manual and automated processes identified procedural differences that may contribute to differences in process output. Experience with more cell types is required to discern generic issues from those that are specific to a given cell type or process. Preliminary experimental work leading on from process mapping has identified the lack of a centrifugation step in the automated process and resultant residual trypsin in the culture media as a major procedural difference that may influence the fate of sensitive cell types. The process engineering techniques described below can be applied to identify process input variables such as this that can be manipulated to attain the desired specification for each cell type.
The Process Improvement Methodology
The Healthcare Engineering group uses a six-sigma-guided approach to improve automated therapeutic cell culture processes. This focuses on the data-driven DMAIC tool for process improvement: definition of the critical to quality issues, measurement of the relevant parameters, analysis of the process, improvement of the process and validation, and control of the improved process.
Definition of Critical to Quality Issues
The lack of a real current customer specification for many speculative therapeutic cell types requires “critical to quality” issues to be defined for a generic regenerative medicine customer based on consultation with the industrial and academic science base. Recurring nonspecific problems include inadequate process reproducibility (particularly in periods of product and process technology transfer), the high cost of product manufacture, uneconomic cell yield while maintaining product quality, and the high cost of commercial scale up. Automation has intuitive value in some of these areas through economic scalability and removal of operator associated variability. In the case of the automated expansion of an hMSC population, we identified low yields of progenitor cells as a critical quality issue for automated processing to which to apply the process improvement methodology.
Measurement and Quality Specification
The second step in the DMAIC cycle is to measure the performance of the automated system against the customer specification. However, product quality specification and the associated measurement systems for process control and product validation are a consistent problem in automated cell culture for therapeutic purposes. Cell input to clinical trials have tended to rely on known starting materials and a controlled process to produce a cell population that is then screened for microbial safety. A definitive functional specification, for example gene expression or surface marker, is still elusive for many therapeutic cell types. It is difficult to prove that a cell expressing a certain gene combination or surface marker profile is committed to a specific phenotypic fate, or conversely is unable to adopt a potentially damaging fate. hMSCs and human embryonic stem cells (hES cells) are both examples where surface markers and gene expression provide guidance to cell character but are not specific to the cell type and do not guarantee cell efficacy or safety.
Conducting manufacturing process research in such an ill-defined environment requires an incremental approach to product specification. After consultation with industrial, academic, and clinical partners, we are using a surface marker profile including STRO-1, CD105, CD166, ALP, and CD71 for characterizing hMSC cells and a profile including SSEA3, SSEA4, OCT3/4, and the absence of SSEA1 to characterize hES cells. 3 However, some markers are expressed on more specific cell subsets than others, and none are definitive.
Measurement of the critical to quality progenitor issue for automated hMSC processing is shown in Box 1. The automated system produces a cell population with lower STRO-1 expression (and potentially higher ALP expression), and hence lower progenitor potential, compared to the expert manual process. We therefore aimed to improve the progenitor population yield in the automated process to at least the yield of the manual population. This preliminary data further allowed a short-term capability analysis of the automated process. This understanding of process variation is necessary to exclude special causes of variation before designing statistical experiments and also to allow power analysis of the experiments. Process analysis and improvement are conducted based on this initial quality and variability data.
Analysis of Process
The third step in the DMAIC cycle is to analyze the automated process. This involves constructing a process map identifying all of the key subprocesses and corresponding key process input variables and key process output variables. This provides a living document for developing process knowledge and a systematic approach to identify potential sources of process variation. Figure 2 shows a simple high-level subprocess map of the automated expansion of an hMSC population. Table 1 shows how an example simple subprocess (automated media change) is broken down into component parts that may contribute to variation. Other subprocesses, such as cell passage, are far more complex and have longer lists of input and output variables. Process maps are usually accompanied by cause and effect diagrams that help identify, sort and display possible causes of a problem or quality characteristic. These are particularly hard to construct at an early stage in the development of a process where the relationships between input and output variables are so poorly understood, but they are enriched with data through iterative experimental cycles.
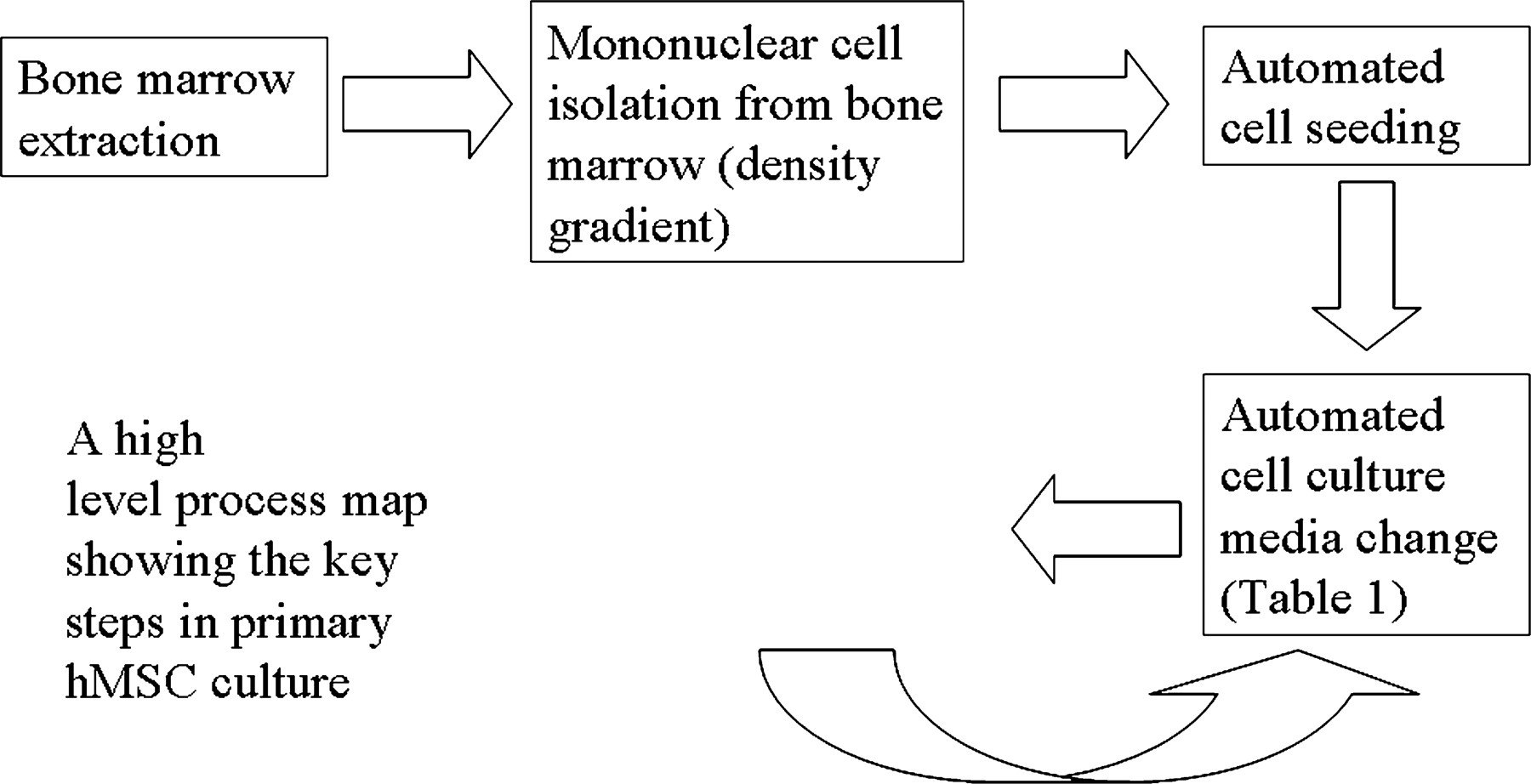
A high-level process map showing the key steps in primary human mesenchymal stem cell culture.
A breakdown of the automated media change sub-process to help identify key variables

Improvement of Process
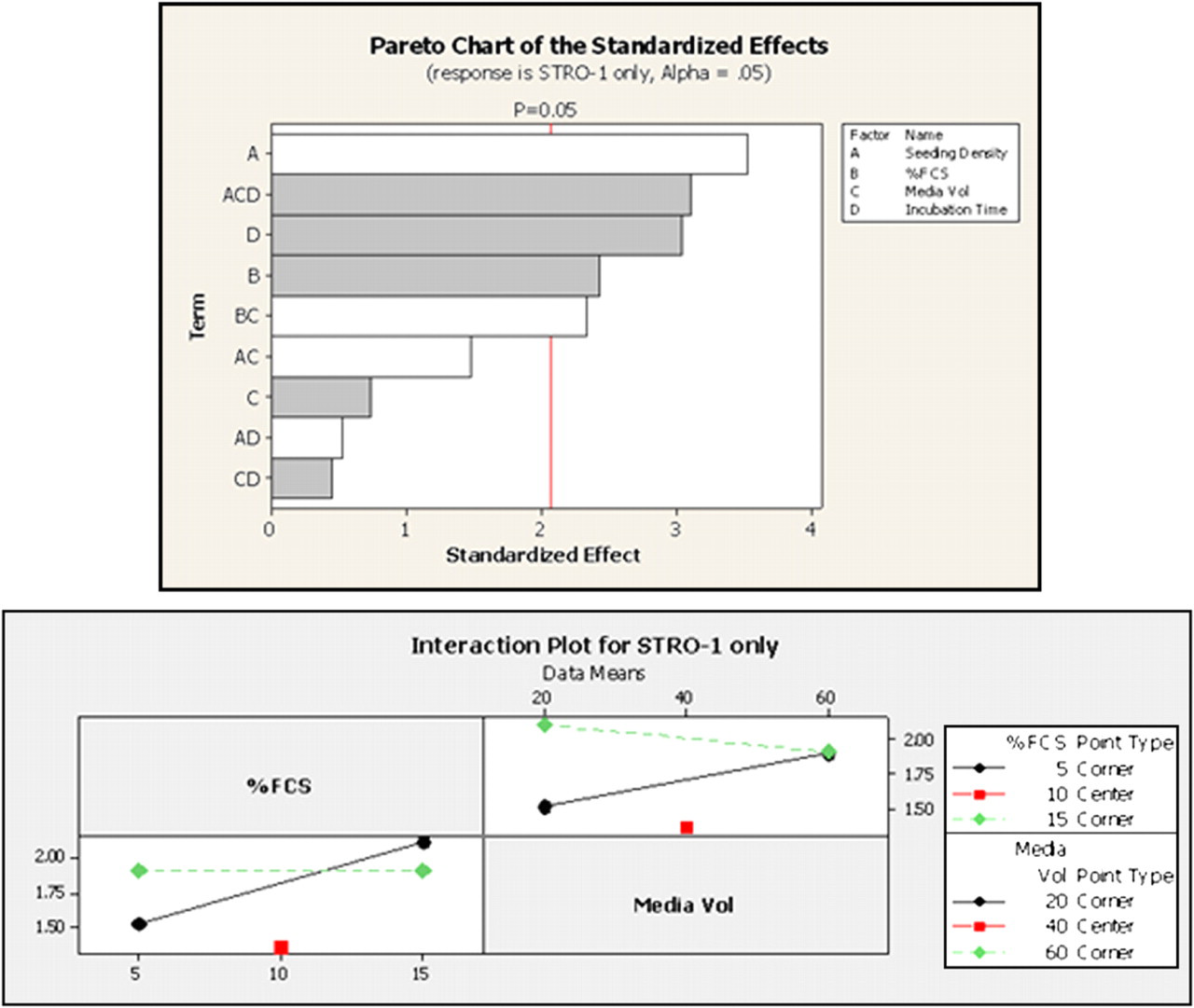
The fourth step is to improve the automated process based on the analysis. The analysis identifies so many cell culture parameters that a standard single variable experimental approach would be impractical in terms of time and resource. However, the number of, and potential for interaction between, the culture factors and the ability to culture at large scale makes the automated process an ideal candidate for a factorial experimental approach. The systematic route to improvement in this scenario would involve conducting a partial factorial screening experiment to identify main parameters contributing to automated process output variation. This would be followed by a full factorial analysis of the major identified sources of automated process variation to characterize their independent and interacting effects on the automated process output. Box 2 shows an example of data we have obtained in our laboratory through use of a statistically designed full factorial screening experiment to investigate major sources of process variation in the automated expansion of hMSCs. The example pareto chart shows which cell culture factors will either reduce or increase the expression of STRO-1. The example interaction chart shows how the cell culture factors “serum” or “media volume” effect STRO-1 expression in a non independent manner, that is, the change in process output in response to a change in one factor is dependent on the level of the interacting factor.
An example of the output from a factorial experiment. White lines in the pareto chart (right) indicate a negative effect on STRO-1 expression, a progenitor marker, and gray lines a positive effect. The nature of the significant interaction between serum % and media volumes is shown in the interaction plot (below).
Validation and Control
The final step of the DMAIC cycle involves verifying the results of the optimization experiments and ensuring the process remains in specification. However, this is only achievable if the variability of undefined categorical cell culture variables (e.g., serum batch, primary cell source) that were held constant in the improvement phase are maintained or understood in the validation phase. This presents problems for developing capable automated production processes for autologous therapies where cell source is a poorly understood variable and restricts the application of the results from experiments using primary cells (such as the hMSC example given here) to a strict set of laboratory conditions. Work is therefore ongoing to widen the process investigation to address the primary cell source variability and standardize other culture inputs necessary for the full validation and practical applicability of the hMSC results to autologous clinical application. However, the demonstration of the value of the methods for analyzing automated cell culture processes and identifying sources of variation in important outputs has initiated similar process improvement projects on banked cell lines for putative allogeneic therapies. In these cultures, the output will be easier to validate due to the low level of variability in the input material.
Summary
At Loughborough University, we have demonstrated that automated cell culture coupled with the application of process engineering principles will be an important tool for the successful commercial production and early-stage process development of therapeutic cell cultures. The approach is particularly powerful because the process control of the automation provides power to statistical analysis and enables large numbers of variables to be manipulated when compared to possible manual experiments. The automation platform also has the benefit of being a realistic potential production unit thereby adding relevance to the output. The tools and examples outlined are an introduction to the power of a multidisciplinary team-based approach to automated manufacturing process issues in tissue engineering and regenerative medicine. These tools are also being applied to add value for other process outputs such as process time, costs, and other identified critical outputs. Through the application of these techniques, we hope to develop optimized validated processes for automatic production of multiple cell types for cell-based therapeutic applications.
Acknowledgments
This work forms part of the UK Engineering and Physical Sciences Research Council funded Innovative Manufacturing Grand Challenge in regenerative Medicine—Remedi. Remedi is a partnership of Loughborough, Nottingham, Cambridge, Birmingham, Ulster and Liverpool Universities, and industry and agency stakeholders. We are particularly grateful for the support of Richard Archer and TAP in this work.
