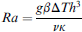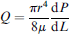Abstract
We have designed a novel convective flow-based thermocycling system capable of performing high-speed DNA amplification via the polymerase chain reaction in a simplified and inexpensive format. Successful amplification of a 191 bp influenza-A target is demonstrated within 25 min using a 10 μL reaction volume with no modification to standard laboratory protocols. The system is simple to assemble and can be readily integrated with existing laboratory instrumentation for automated operation. (JALA 2006;11:217–21)
Introduction
DNA sequence information generated from the Human Genome Project continues to stimulate new research in a variety of areas including medical diagnostics, genomic analysis, and drug discovery.
1
DNA fingerprinting assays, for example, are becoming an increasingly common component of many diagnostic procedures. Many of these genomic analysis strategies rely on the polymerase chain reaction (PCR) to amplify a specific region of interest from a longer DNA template before subsequent separation, purification, and detection steps can be performed. However, PCR continues to be one of the key rate-determining steps in the overall analysis, imposing limitations to the ultimate level of achievable throughput. The timescales required to perform a typical amplification remain relatively slow (> 1 h), not due to reaction kinetics but as a result of the highly inefficient design of conventional thermocycling hardware (generally incorporating high-heat capacity metal blocks).

The underlying biochemistry of the PCR requires reagents (template DNA, primers, dNTPs, thermostable Taq polymerase enzyme, etc.) to be repeatedly cycled approximately 30–40 times through three distinct temperatures associated with denaturation, annealing, and extension processes. This thermal cycling can be accomplished in a variety of ways including the following: (i) placing tubes containing the PCR reagent mixture in a stationary metal block whose temperature is electronically varied, (ii) loading PCR reagents into high surface-to-volume capillary tubes that are heated and cooled using forced air streams (e.g., LightCycler, Roche; SmartCycler, Cepheid; PRCJet, Megabase Research Products), and (iii) mechanically transporting the PCR reagents between individual blocks or baths maintained at constant temperatures (e.g., Robocycler, Stratagene). Although these designs are conceptually straightforward, the desire to provide rapid cycling while maintaining adequate temperature control often introduces a considerable degree of electronic and mechanical complexity. Among these approaches, the metal block design is one of the most commonly used, however, the time and energy required to repeatedly ramp the block temperature up and down makes this approach extremely inefficient. Recent advancements in microfluidic technologies have enabled PCR thermocycling to be performed in a miniaturized format, however a variety of issues remain to be addressed before performance and versatility comparable to conventional instruments can be achieved.2–6
An emerging approach to tackle these complexities involves harnessing natural convective forces to perform thermal cycling.7–11 Convective thermocyclers use the same thermal energy required to maintain PCR temperatures to simultaneously circulate reagents through each temperature zone thereby reducing unnecessary ramping time and energy requirements. Convective flows have been extensively studied and characterized in literature,12, 13 and occur as a result of buoyant instabilities that develop due to differences in density at different locations within the fluid. At very large scales, for example, convective currents in the atmosphere and oceans can give rise to devastating hurricanes. When the underlying physics is considered, a dimensionless parameter called the Rayleigh number emerges that expresses the ratio of buoyant forces to the restoring thermal and viscous diffusive forces
12
Here, g is the acceleration due to gravity, β is the thermal expansion coefficient of the fluid, ΔT is the temperature gradient across the fluid body, h is a characteristic length scale, ν is the kinematic viscosity, and κ is the thermal diffusivity. When Ra is below a critical value, there is insufficient driving force to initiate fluid motion, and heat transfer occurs primarily by conduction. When a critical value is exceeded, however, a convectively driven flow field is established, and heat transfer is dominated by convection. In the case of fluid confined between two infinitely wide horizontal surfaces separated by a vertical distance h, for example, the onset of convective flow occurs at a value of Racrit of approximately 1700.
Convective flows can occur in a variety of formats, including cavities and closed loops (Fig. 1). In the closed loop case, often referred to as a thermosyphon, convection can be generated either by applying horizontal or vertical temperature gradients across the loop. The closed loop design with horizontal temperature gradient is particularly attractive for thermocycling applications because unidirectional flow through the loop can be generated allowing residence times within specific temperature zones to be precisely controlled. Rapid cycling times are achievable because the convected fluid elements are able to quickly attain thermal equilibrium with their surroundings as they are transported through successive temperatures without the lag time associated with heating and cooling inactive hardware components (e.g., metal blocks). In addition, the value of Racrit is quite low in these systems (of order 1–10) meaning that convective flows can be readily generated under PCR temperature conditions.
11
These unique characteristics are ideally suited to meet the requirements of thermally actuated biochemical reactions like PCR.
5
Most studies to date in the area of convective flow thermocycling, however, have been primarily directed at a proof-of-concept level, lacking capabilities for integration and automation. In this article, we describe a compact closed loop thermocycler design suitable for integration with automated upstream and downstream analysis and detection processes, and capable of performing rapid DNA amplification with little or no modifications to existing laboratory protocols.
Buoyancy-driven convective flows in cavity and closed loop geometries.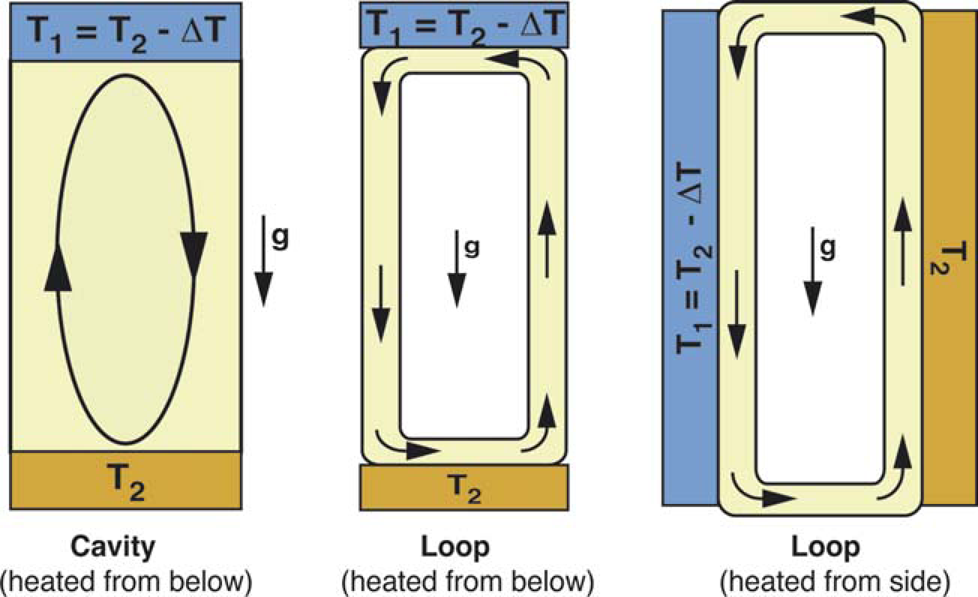
Design and Testing of a Prototype Convective Closed Loop Thermocycler
The thermosyphon concept can be straightforwardly applied to perform PCR thermocycling by simply applying a temperature gradient across a closed loop geometry. We have developed a prototype closed loop thermocycler (CLTC) consisting of a triangular flow geometry designed to replicate the three stages in a PCR cycle (i.e., denaturing, annealing, and extension). The device consists of two independently controlled thermoelectric heaters (Catalog # HT4-6-21X43, Melcor Corporation) affixed on opposite sides of a triangular aluminum scaffold as shown schematically in Figure 2. When one of the heaters is maintained at 94 °C (denaturing) and the other at 72 °C (extension), the third side of the scaffold passively attains 55–60 °C (annealing). This arrangement provides sufficient driving force to initiate circulation of PCR reagents through the loop. The denaturing and annealing processes occur almost instantaneously, whereas extension proceeds at the rate of about 60 bases per second.
14
Based on these considerations, the triangular geometry should be designed such that the extension leg of the loop is sufficiently long to allow replication of the target fragment of interest. Flat thermocouple probes (Omega Engineering, Inc.) are affixed on the heaters to monitor surface temperatures. A 9-cm-long segment of fluorinated ethylene propylene (FEP) tubing (Zeus Industrial Products, Inc.) of 320 μm inner diameter and 10 μL volume is loaded with PCR reagents after inserting one of the ends inside a shorter sleeve of flexible polyvinylchloride (PVC) tubing of slightly higher diameter. After pipetting the reagents into the tube segment, the free ends are joined together by hand using the PVC sleeve, and the loop is wrapped around the triangular assembly. Slightly overfilling the tubing before joining minimizes the possibility of trapping air bubbles. Aluminum adhesive tape is wrapped around the tubing to ensure thermal contact. The choice of FEP tubing was motivated by its attractive combination of biocompatibility and optical clarity. Cycle times of 102 s were achievable under PCR conditions using this design.
Schematic of the CLTC device incorporating Peltier heaters on two sides of a triangular scaffold. When the heaters are maintained at 94 and 72 °C, the third side of the scaffold passively attains annealing conditions (55–60 °C).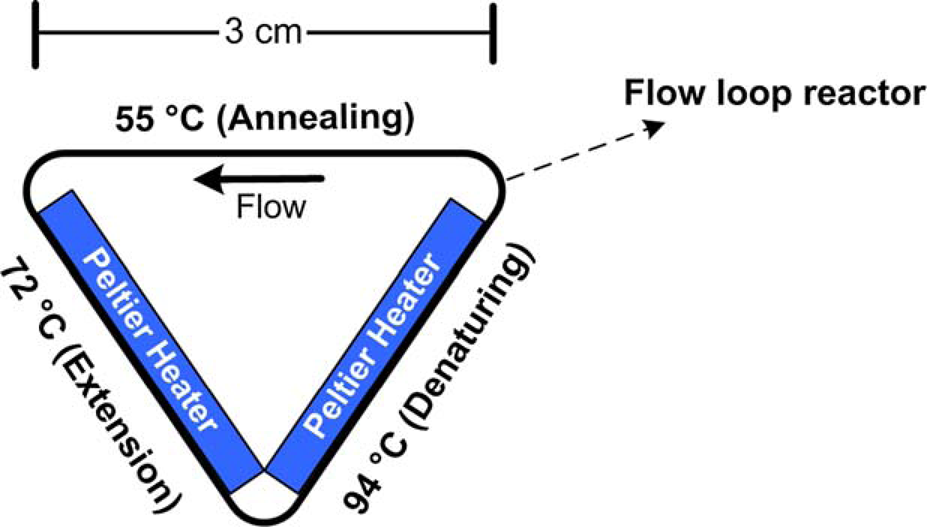
We used this CLTC device to successfully amplify a 191 bp target associated with the M1 and M2 channel proteins of the influenza-A virus within 50 min. To determine the minimum reaction time required to replicate the target amplicon (191 bp), we performed a series of reactions with reaction times ranging from 20 to 55 min. As expected, the electrophoresis results of the amplified products show a reduction in the product band intensity as the reaction time is decreased (Fig. 3A). Robustness was investigated by performing a series of reactions at varying denaturing temperatures ranging from 90 to 96 °C. Figure 3B shows that successful amplification can be achieved over a range of denaturing temperatures.
(A) Electrophoresis gel picture showing an amplified 191 bp target associated with the membrane channel proteins M1 and M2 of the influenza-A virus from a 3.9 kb template (Maxim Biotech, Catalog # SP-10377) at seven different reaction times. (B) Gel picture showing the amplification results of the 191 bp target at seven different denaturing temperatures. Standard 50 μL reaction mixes contained 30 μL of optimized buffer/dNTP mix, 10 μL of primer mix, 8.75 μL of ddH2O, 1 μL of template DNA, and 0.25 μL of 5 units/μL AmpliTaq polymerase (Applied Biosystems). On completion of the reactions, products were aspirated from the flow loops, run on a 2% agarose gel (5 μL loaded product volume; Mini Sub-Cell GT, Bio-Rad Laboratories, Inc.) at 60 V for 1 h, and stained with 100× SYBR-Green I (Molecular Probes).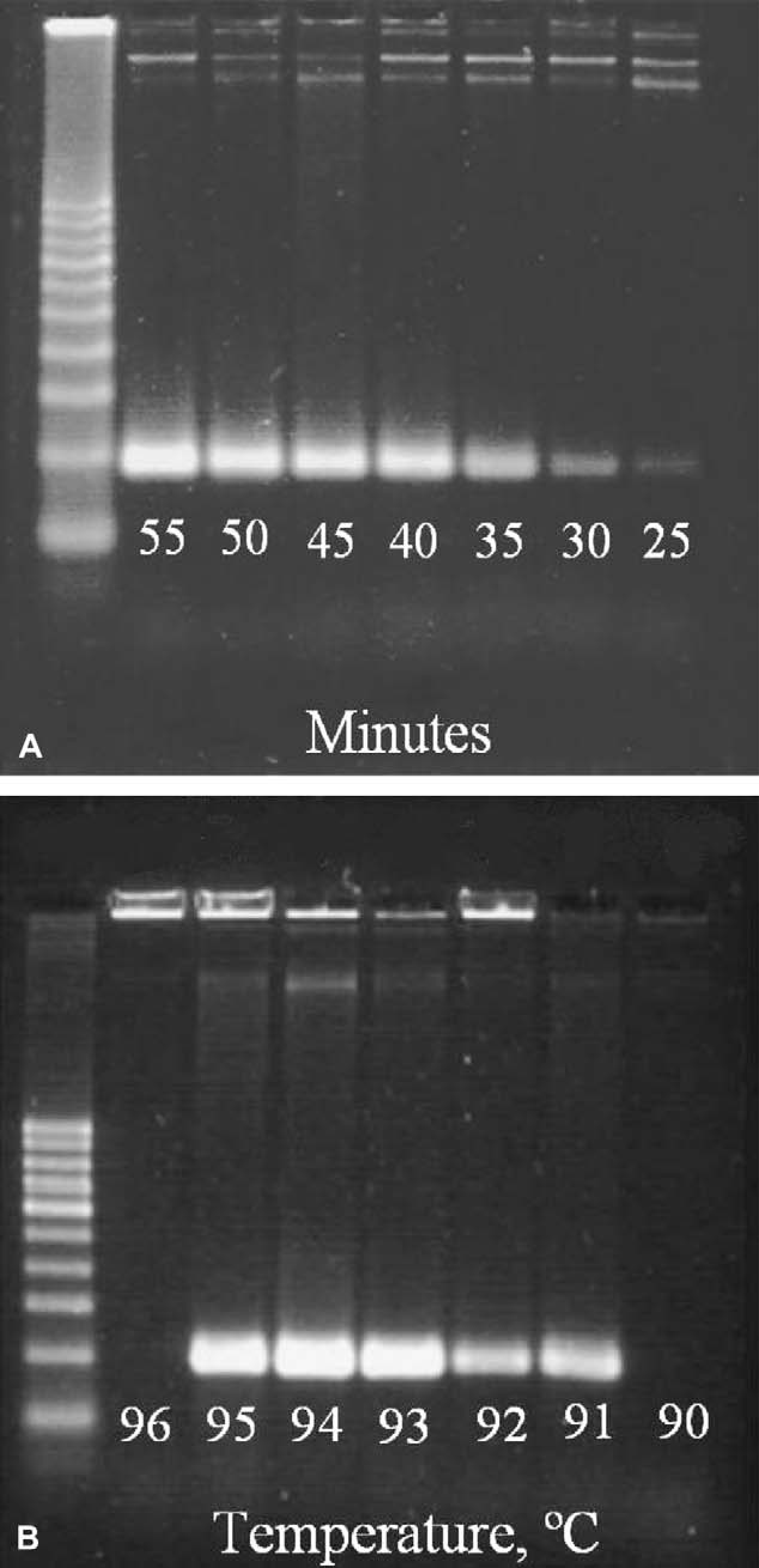
Overall cycling times and residence times within specific temperature zones are both determined by the flow rate through the loop. A general picture of the interplay between temperature and flow rate can be obtained by considering the relationship between the pressure drop dP/dL and flow rate Q for laminar Poiseuille flow through a tube of a Newtonian fluid with viscosity μ
where the geometric parameters r (tube radius) and L (loop length) are the primary control variables. In a convective flow loop, a pressure drop is associated with the buoyant forces that arise in response to variations in temperature along the loop perimeter. We characterized this by performing flow visualization studies after loading a series of 9-cm-long FEP tubing segments of different inside diameters (320, 400, and 510 μm) with an aqueous suspension of 6 μm diameter fluorescent latex microspheres (Polysciences, Inc.). Flow velocities were monitored by observing microsphere motion using a microscope equipped with a mercury arc illumination source and CCD camera. As expected, the flow rates increase with increasing temperature gradient, and the flow rate at a particular temperature gradient increases with channel diameter (Fig. 4). The prototype design is capable of delivering 30 cycles in about 21 min using the 510-μm diameter tubing, although these times can be significantly reduced through the use of shorter loops. By performing momentum and energy balance analysis for a closed loop system with constant wall temperature boundary conditions, a model can be developed to enable the design of CLTC reactors incorporating virtually any combination of reaction time and volume.
15
Flow rates determined from observing the motion of fluorescent microsphere tracers as a function of applied temperature gradient in three different tubing diameters. The flow rate increases with the applied temperature gradient at a fixed diameter, and increases with diameter at a fixed temperature gradient.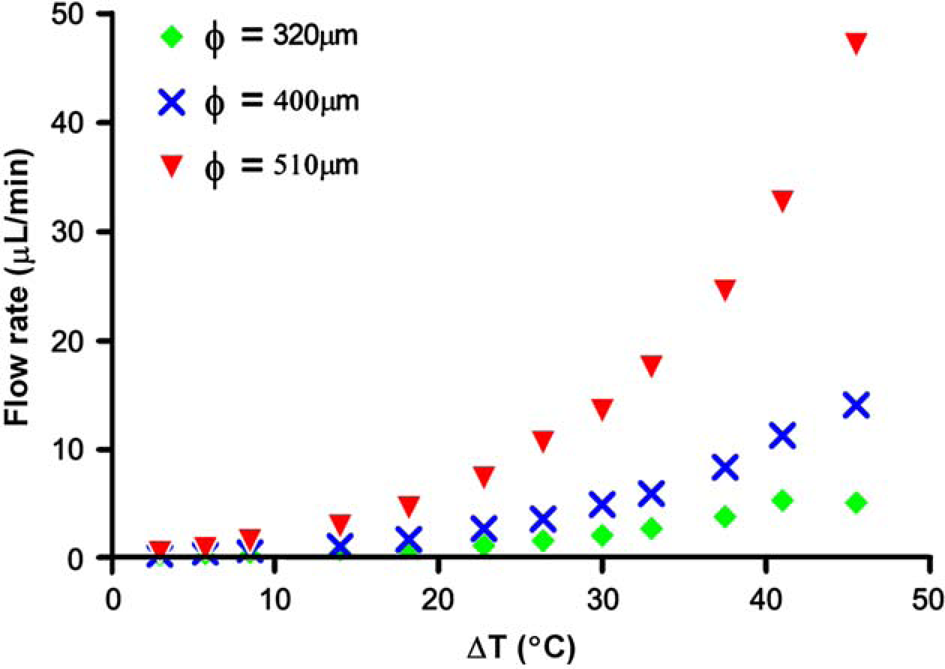
The CLTC offers advantages over conventional benchtop thermocyclers in terms of speed, portability, and cost. A format equivalent to the multiwell arrangement in benchtop thermocyclers can also be achieved by using an array of reactors arranged in parallel on the triangular scaffold. In terms of power consumption, the dual thermoelectric heater design used in this prototype draws about 3.5 W, with each 4-cm-long heating element capable of comfortably accommodating 10–12 tubing loops (including both the tubing outside diameter and the larger diameter PVC sleeve used to join the free ends, and accounting for a 2-cm-long inactive area occupied by temperature sensors and mounting hardware). Based on these results, a scaled-up design capable of accommodating 96 loops would require a heating element of about 18 cm in length (calculated using a basis of 12 loops per 2 cm heater length, plus a 2-cm-long inactive zone) that would be expected to consume about 16 W of power (calculated using a basis of 3.5 W per 4 cm heater length). Even considering the case where all three temperature zones are actively controlled (power consumption would increase to about 24 W), this analysis implies a power requirement of 100 W or less for a 384-reactor array. For comparison, the conventional benchtop thermocycler used in our control studies (TGradient, Biometra) lists a 310 W maximum power rating. Although these power consumption values are approximate, they can serve to establish order of magnitude estimates for comparison. Finally, we note that the FEP loops are inexpensive and disposable, and the entire system is suitable for use in a variety of applications including cycle sequencing and real-time PCR assays.
Potential for Automation
Owing to its tubing-based design, the CLTC offers excellent potential for automation with both upstream sample preparation and downstream analysis operations (Fig. 5). For example, a fluidic interface capable of metering discrete amounts of sample, mixing with PCR reagents, and pumping into the flow loop could be constructed using electronically controlled valves. Once the reaction is complete, the products can then be extracted for subsequent analysis followed by a rinsing operation so that the same loop can be used to perform multiple reactions. Alternatively, because each loop is inexpensive, an ensemble of flow loops could be used in conjunction with a flow circuit capable of switching to a new loop for each reaction. Reagents could also be incorporated into replaceable dispenser cartridges containing a sufficient quantity to perform multiple reactions.
An automated DNA analysis scheme incorporating CLTC-based PCR integrated with upstream sample preparation and downstream analysis operations. The tubing-based design is adaptable for use with automated liquid-handling instrumentation.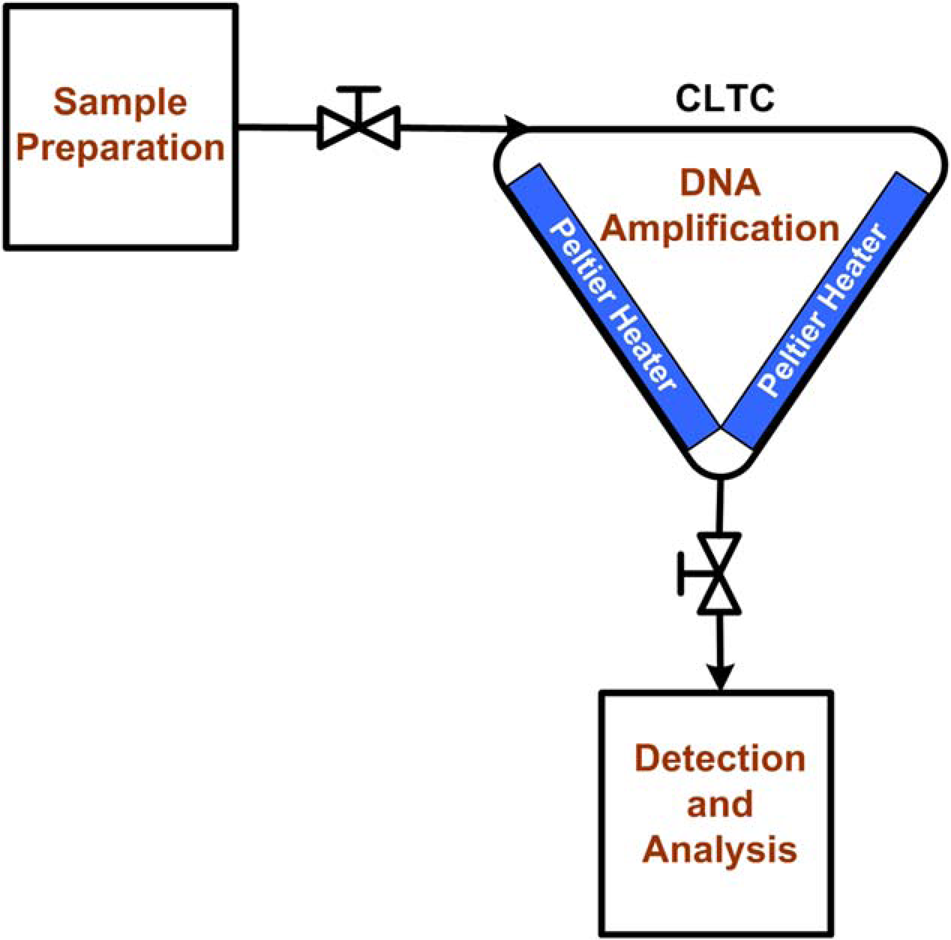
Conclusions
Despite incredible advancements in PCR biochemistry, existing thermocycling hardware continues to suffer from limitations associated with slow timescales and extensive energy requirements. Convective flow thermocycling offers an innovative approach toward addressing these limitations. We have demonstrated this potential through the design, construction, and testing of a convectively driven closed loop thermal cycling device capable of performing PCR in a rapid, inexpensive, and greatly simplified format. Successful amplification of a 191 bp influenza-A target has been demonstrated within 25 min using a 10 μL reaction volume with no modification to existing laboratory protocols. The FEP tubing reactors are biocompatible, require no surface treatment, and are optically transparent enabling PCR detection in a real-time format.
Footnotes
Acknowledgment
This work was supported by the National Institutes of Health under grants K22-HG02297 and R01-HG003364.