Abstract
Our laboratory has been testing ways to reduce costs, sample volumes, and decrease labor in microsatellite (or short tandem repeat polymorphism) genotyping. Microsatellite genotyping involves polymerase chain reaction amplification of a short (100–400 bp) fragment of chromosomal DNA that encompasses the tandem repeats followed by electrophoresis to size the amplification products. Using a continuous polypropylene tape (array tape) embossed with 384-well arrays, conforming to the microtiter plate standard, we have been able to perform the amplification reactions in smaller volumes and to decrease handling of stacks of microtiter plates. Instruments were constructed in-house to achieve these results. However, the problem of removal of the samples from the tape for electrophoresis remained. We report here efficient piercing of the tape seal for extraction of the samples using a CO2 laser. Scoring of the seals with the laser weakens it sufficiently to permit extraction of the samples with a syringe array. CO2 lasers are robust systems that do not contain a lot of frequently replaced parts, and do not require frequent recalibration. In addition, the laser is software controlled allowing for highly reproducible scoring and easily switching between 384-, 1536-, and 96-well formats. (JALA 2006;11:260–7)
Introduction
Microsatellite genotyping has traditionally been a highly labor-intensive process involving large amounts of technician time. Multiplexing markers and increasing the number of detection dyes have led to higher throughput, but vast amounts of sample plates still need to be handled manually or with expensive robotic arms and stackers. In addition to the cost of handling all of these plates, they require large amounts of storage space. To aid in automation, Marshfield Clinic Research Foundation (MCRF) has been developing a microtiter plate replacement consisting of continuous polypropylene tape (array tape) embossed with 384-well arrays, conforming to the microtiter plate standard.1–3 Array tape also allows smaller reaction volumes to be used and can be easily manufactured to have wells that match the desired reaction volume.
Commercial equipment was not available for purchase when work with the array tape started. This required several instruments to be developed, including a transfer unit to replicate the unique samples into each well (Fig. 1A).
4
The transfer unit currently incorporates a commercial positive displacement head capable of dispensing volumes down to about 500 nL (Tomtec, Hamden, CT). A barcode print and apply is attached to the transfer unit to aid in sample tracking. The common reagent, or test, is dispensed into the entire array using a noncontact solenoid valve system (Fig. 1B).
5
This inkjet-type dispense was developed in conjunction with Oak Ridge National Laboratories. Sealing required for water bath immersion is a key component. The tape-sealing unit developed allows the use of pressure seal or heat seal (Fig. 1C). The samples are amplified using polymerase chain reaction (PCR) in thermal cycling water baths (Fig. 1D). Previous work in the array tape was performed as closed tube assays.
6
To make array tape viable as a complete microtiter plate replacement, it was necessary to be able to remove the sample from the sealed wells. A pooling unit (Fig. 2A) similar to the transfer unit was developed to facilitate extraction of the samples from the array tape. The unit contains three independent X-axes including an array tape indexer, a four-position plate shuttle, and a dual position wash station (Fig. 2B). The samples from successive arrays, each containing a different marker dye, are combined with size standards and a loading dye into a 384-well microtiter plate. The pooled samples are then loaded into polyacrylamide gels for analysis.
(A) A 384-well unique sample transfer unit to replicate the unique samples into each well. The transfer unit currently incorporates a commercial positive displacement head capable of dispensing volumes down to about 500 nL. (B) The common reagent, or test, is dispensed into the entire array using a noncontact solenoid valve system. (C) The tape-sealing unit uses pressure or heat seal to prepare the tape for water bath immersion. (D) The samples are amplified using PCR in thermal cycling water baths. (A) Pooling unit with CO2 laser to facilitate extraction of the samples from the array tape. (B) The unit contains three independent X-axes including an array tape indexer, a four-position plate shuttle, and a dual position wash station.

Methodology
Array Tape Background
Array tape (also called Microtape) is a continuous strip of polypropylene tape embossed with arrays of reaction wells conforming to the microtiter plate standards (Fig. 3). Array tape can replace microtiter plates in virtually any high-throughput process. Embossed tape has been used extensively in the electronics industry for years. Circuit components such as resistors and capacitors are packaged into wells in the tape, sealed, and are then transported to circuit board assembly plants where the components are plucked out of the tape by robotic arms and placed onto circuit boards. Massive amounts of tape are manufactured for this application. In the 1980s, tape rapidly displaced individual plastic trays in circuit board production. The tape manufacturing process is mature, and the progression from individual component handling to continuous format was proven in the electronics industry.7–8
Array tape is a continuous strip of polypropylene tape embossed with arrays of reaction wells conforming to the microtiter plate standards.
Array tape has several important advantages over microtiter plates. One of the most important is that array tape is less expensive to manufacture than microtiter plates. Microtiter plates are made by a process called injection molding in which plastic is heated until malleable and then injected at high pressure into a metal mold. Array tape is made by a type of thermoforming called embossing. A section of blank, flat plastic is heated until soft and then is punched with a metal tool to produce wells.9–10 Because the manufacturing process is simpler and less plastic is used, the cost of the array tape is 10% or less the cost of individual microtiter plates.
It is easier to change the design of the array tape than that of microtiter plates. The metal molds used for the microtiter plate manufacturing process cost on the order of $200,000. Changing the design of a microtiter plate is therefore cost prohibitive. In contrast, the metal tools used to stamp the array tape cost on the order of $12,000. This makes it much easier to alter the shape or geometry of the array tape. Eleven different versions of the tape have already been tested. Array tape can be manufactured (Global Array, Minneapolis, MN) in a wide variety of different plastics and geometries. The wells of the tape can be coated with many different agents.
Because it contains less plastic and can be spooled, array tape is also much more compact than microtiter plates. A polypropylene 384-well tape array weighs less than 20% of the corresponding 384-well plate. A reel of 600-tape arrays is only about 3.5 in. wide by 22 in. in diameter (90 mm by 560 mm). Such a reel is much easier to transport and store in a lab than the corresponding number of plates.
Thermal transfer is also more rapid during thermal cycling in the array tape compared to that in the individual plates (Fig. 4). These profiles were generated using microtemperature probes within the wells. The temperature transitions of the tape are much sharper than those of the plate as the material is transferred from one water bath to another. The temperature dips in the tape occurred during the brief time when the tape was in the air between baths. This problem has been solved by enclosing the coil of tape in a plastic shield to insulate the tape as it travels from one bath to another.
Comparison of thermal transfer characteristics of array tape and microtiter plate using microtemperature probes within the wells.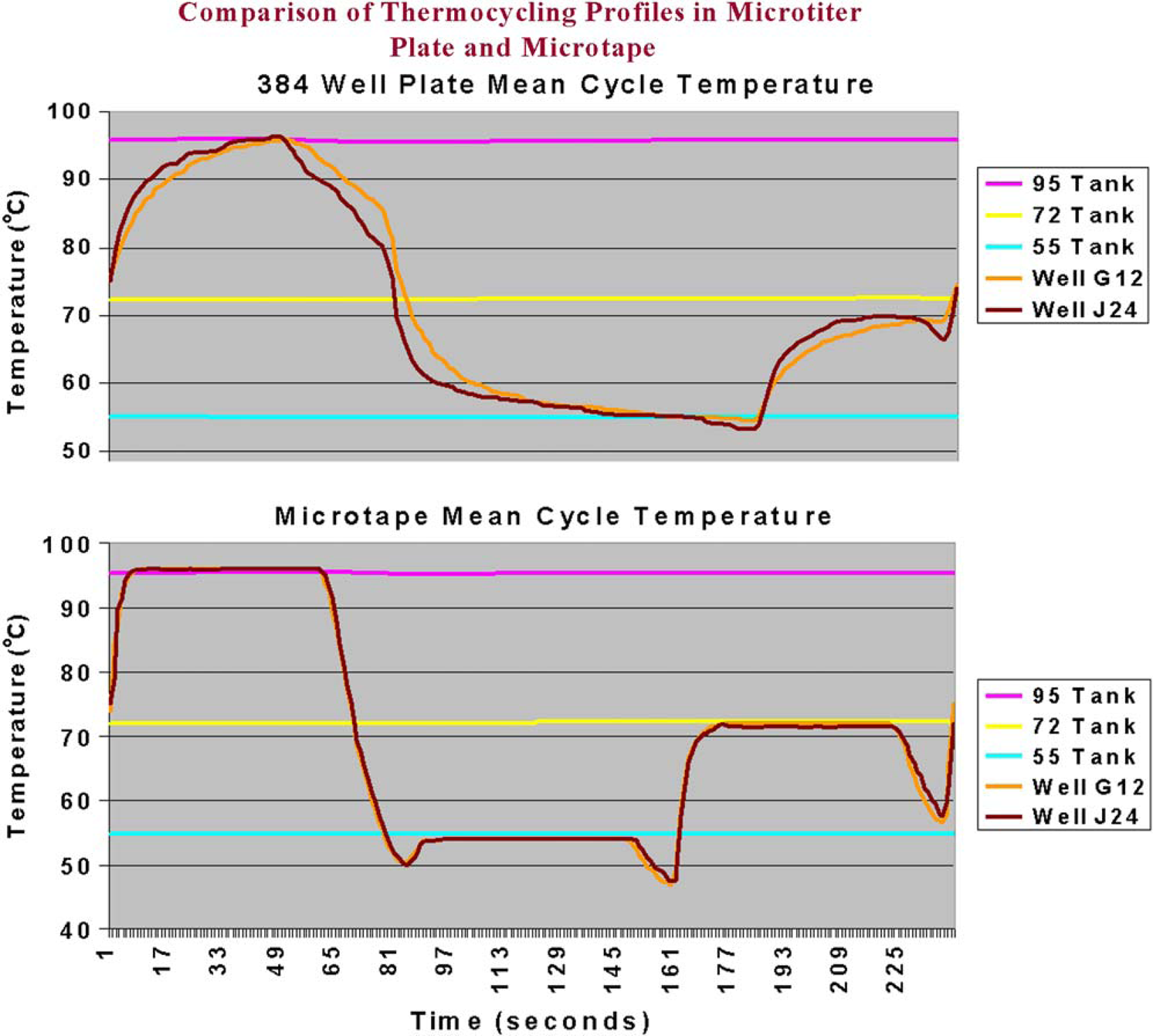
Although the advantages described above are important, perhaps the greatest advantage lies in the indexing holes that run along either edge of the tape. These index holes serve the same purpose as the holes along the edges of old computer paper that was used with tractor feed printers. The holes allow the tape to be fed in a continuous, automated process through various liquid handling, detection, and other instruments. This is a far more elegant process for high-throughput testing than the current use of robotic arms for moving individual plates. The robotic translation devices for the plates are substantially more complicated, expensive, and prone to failure than tape translation systems. This simplicity leads to greater efficiency.
Amplification comparison tests were performed in 96-well plates and 384-well array tape.11–13 The 96-well plates served as the control and contained 3 μL of DNA per well at a concentration of 15 ng/μL. The DNA was allowed to air-dry in the wells before the 4 μL of PCR mix was added to the wells. Four 96-well plates were used, one for each marker dye used, and each contained three to four multiplexed markers from the Mammalian Genotyping Service (MGS) standard screening set of 400+ markers.
14
The array tape was prepared in the same manner into four consecutive arrays with the volumes halved to 1.5 μL of DNA and 2 μL of PCR mix per well. The DNA transferred was the same template, and one PCR mix per marker dye was made that was then divided between the plate and corresponding array. All plates and arrays were sealed and run simultaneously on the same water bath thermal cycler. The samples from the four 96-well plates were then hand pooled with a 12-channel Hamilton syringe along with size standards and loading buffer into another 96-well plate for loading. A razor blade was used to slice the seal of the array tape, and the samples from the first quadrant of the four arrays were then hand pooled with a 12-channel Hamilton syringe along with size standards and loading buffer into a 96-well plate for loading. Both sets of pooled samples were then loaded into polyacrylamide gels for analysis. Half of the gel contained the samples from the 96-well plates, and the other half contained the samples from one quadrant of the 384-well array tape. Since both halves of the gel contained the same DNA template amplified with the same markers, the results should have been identical. Results of the scan do in fact show the two methods to be very comparable (Fig. 5) in both completeness and accuracy. With the confirmation of the results of the amplification in the two different reaction vessels, it then became necessary to automate the sample removal process to increase efficiency.
Details of marker allele calls from an amplification comparison test performed in 96-well plates and 384-well array tape. The 96-well plate results are shown in the upper image, and the array tape results are in the bottom image.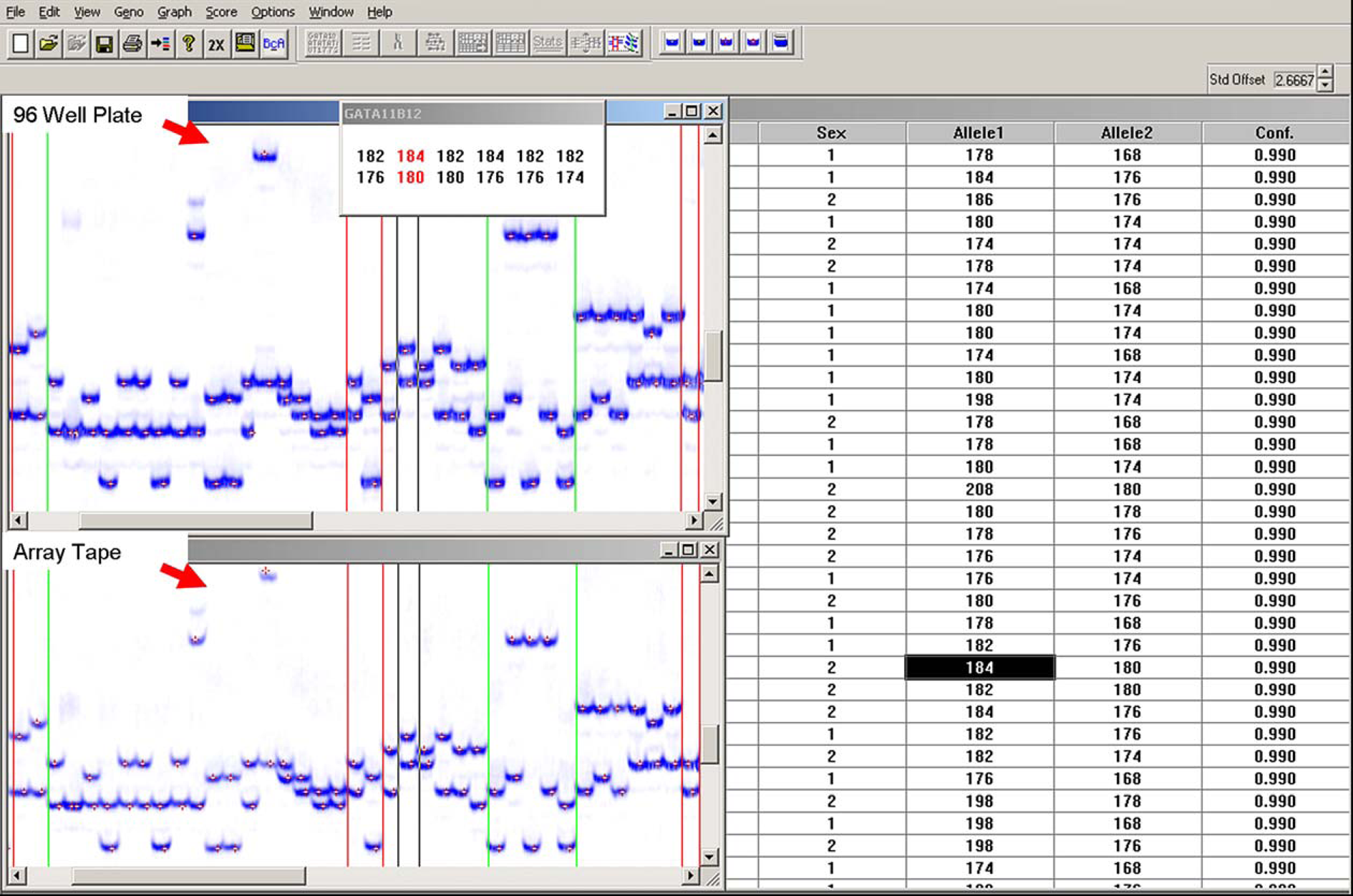
Sample Extraction Methods
Peeling the Seal off Sealed Tape.
Water bath immersion and the surface area that contacts the seal (Brandel, Gaithersburg, MD) prevent peeling of the seal like a microtiter plate. A large amount of force or heat is required to peel the seal off, however, this force can cause the seal to tear and the heat causes the array tape to melt. Large amounts of adhesive also remain, covering some of the wells (Fig. 6).
Water bath immersion and the surface area that contacts the seal prevent peeling of the seal like a microtiter plate. Peeling of the seal causes the seal to tear and large amounts of adhesive to remain, covering some of the wells.
Pierceable Seals.
Piercing a single well was relatively easy, but piercing the entire array proved to be more difficult. Only one pierceable seal capable of water bath immersion was found. However, the 384-tip instrument tended to stretch the seal to the bottom of the shallow wells, and never actually pierced. It takes approximately 200 g or 1.96 N of force for one tip to pierce the seal, which is fairly minimal. Piercing 96 wells with the same force is still relatively easy, requiring 19 kg or 188.16 N of force. However, piercing 384 wells requires 76.8 kg or 752.64 N of force (this is the equivalent of an average-sized person standing on the tips).
Pins.
An array of sharpened pins was used to poke holes in the seal. A large amount of force was required to break through the seal. The seal also tended to stretch before finally breaking and trapped part of the sample between the seal and the side of the well. This approach was ultimately abandoned due to the need for intensive cleaning between arrays to eliminate the risk of carryover.
Razor Blades.
A column of extra heavy-duty razor blades was aligned over the tape, with one blade for each row of wells (Fig. 7). The tape was indexed under the blades to produce horizontal slits across the well columns. This approach was problematic due to the pressure required for the blades to score the seal sufficiently and uniformly. Indexing proved to be extremely difficult, because the high blade pressure required caused several different motors to stall. Variations in the blade edge made finding matched sets of blades difficult. Frequent adjustments and blade replacements were required as the blades wore. As the blades made contact across columns, the risk of carryover increased. The blades also proved to be difficult to clean.
Column of extra heavy-duty razor blades aligned over the tape, with one blade for each row of wells to produce horizontal slits across the well columns.
CO2 Laser.
Industrial CO2 lasers (Worldwide Laser, Gilbert, AZ) are robust systems that do not contain a lot of frequently replaced parts. A 30-W CO2 laser operating at 30% power is coupled to a high-speed galvo head that is controlled through Windows-based software. This system provides highly reproducible scoring of the seal and is only limited by the positional accuracy of the indexing drive. The software optimizes the galvo pattern for minimal movement and repetition (Fig. 8A). Marking patterns can be imported from drawings and can be easily switched between 96-, 384-, and 1536-well patterns (Fig. 8B). CAD software also makes it easy to compensate for pattern distortion that may be caused by the optics (Fig. 8C). This distortion increases with distance from the center of the focusing lens. Pattern design has centered on minimizing the size of flaps of seal caused by the laser-marking process. Large flaps of seal pose the threat of blocking the sample from the tip and inhibiting extraction. Current marking patterns consist of a star pattern that has been optimized to correct for optical distortion and a crosshatch pattern that uses multiple passes over each well to counteract optical distortion. The star pattern generates eight flaps per well and the crosshatch pattern generates four to six flaps per well depending on the well position in relation to the focusing lens. Since the laser is noncontact, carryover and sample cross contamination are highly unlikely.
(A) Snapshot of optimized galvo-driven pattern. The high-speed galvo head that is coupled to the CO2 laser is controlled through Windows-based software. Marking patterns can be imported from CAD drawings and can be easily switched between 96-, 384-, and 1536-well patterns. (B) Crosshatch pattern using multiple passes over each well to counteract optical distortion. (C) Star pattern optimized to correct for optical distortion.
Discussion
Various approaches have been tried to expose the wells for sample extraction following PCR. Each one was found to have limitations ranging from carryover to high cost. Peeling, piercing, and slicing were all relatively inexpensive to set up, but all proved to be difficult to automate and were not very flexible. The CO2 laser carries a higher setup price, but it more than makes up for it with reliability and adaptability.
Commercialization will be key to acceptance of array tape as a microtiter plate replacement. MCRF has been working with Douglas Scientific to develop commercial array tape instrumentation (Fig. 9). The Douglas Scientific unit is a modular system that allows numerous configurations and is a streamlined package.
Douglas Scientific array tape instrument. This modular system allows numerous configurations in a streamlined package.
Over the years, improvements in instrumentation and software have led to dramatically increased efficiency in the MGS. In 1993, MGS genotyping output was 140,000 genotypes at a total cost of $3.00 per genotype. In 2005, MGS output was 11.57 million genotypes at a total cost of $0.27 per genotype. Switching to array tape and autoloading the samples will result in a 25% increase in throughput from runs loaded at night, and will significantly lower the total cost per genotype.
Conclusions
It has been shown that array tape is an acceptable medium to replace microtiter plates in microsatellite genotyping. Implementation of array tape will dramatically decrease the amount of sample plates that need to be handled and stored. Array tape also allows smaller reaction volumes to be used and can be easily manufactured to have wells that match the desired reaction volume. Smaller reaction volumes allow conservation of precious samples and expensive reagents, which can result in more tests being performed on the same quantity of DNA, or the same amount of tests with less DNA.
Current instrumentation has greatly increased automation and reduced labor in sample preparation, and MGS now has the ability to pierce the seal with a CO2 laser to facilitate extraction of the samples from the reaction vessels to load into gels for analysis. The final hurdle in automating microsatellite genotyping is automating the loading of the samples into polyacrylamide gels for analysis. MCRF is implementing an autoloading system complete with thermal blocks for denaturing the samples before loading and 12 needles that are connected remotely to a 12-syringe pump (Fig. 10A and B). The ultimate goal of this system is to automatically load three to four consecutive runs at a time without any intervention.
(A) Polyacrylamide gel and autoloading system showing thermal blocks for denaturing the samples before loading and 12 loading needles that are connected remotely to a 12-syringe pump. (B) Close up detail of needles loading samples into the polyacrylamide gel.
Suppliers
Brandel, Gaithersburg, MD; Seal Part Number 9795BR. Global Array, Minneapolis, MN; Array tape.
Tomtec, Hamden, CT; 384-well positive displacement head Part Number 020869.
Worldwide Laser, Gilbert, AZ; LP8030 30W CO2 Laser system.
Footnotes
Acknowledgments
Supported in part by NIH grant HL62681 and contract HV48141. We thank the entire staff of the Mammalian Genotyping Service.
