Abstract
Xylella fastidiosa (Xf) is the causative agent of Pierce's disease in a variety of commercially important plants such as citrus, coffee, and grapes. By blocking the xylem, Xf disrupts water and nutrient transport. Xf is a gram-negative phytopathogen that can form biofilms. Twelve genes have been identified in Xf that can regulate exopolysaccharides, a major component of biofilms, including aconitase, which responds to intracellular iron levels. We have employed a quantitative assay for biofilm formation referred to as minimal biofilm elimination concentration (MBEC) assay that is amenable for highthroughput screening. Biofilm formation by Xf (Napa, CA) can be blocked using iron chelators such as lactoferrin (LF), ethylenediaminetetraacetic acid (EDTA), and S, S' ethylenediaminedisuccinic acid (EDDS). Incubation of Xf in the presence of LF at 1000 μg/mL for 3.5 days showed inhibition of biofilm formation (42%) as well as inhibition (32%) of planktonic growth (liquid-phase bacteria). EDTA at a concentration of 15 mg/mL inhibited 99.7% of biofilm formation and 98.9% of planktonic growth in a 24 h incubation. In contrast, EDDS at a concentration of 38.2 mg/mL showed 64.7% inhibition of biofilm formation and 33.6% inhibition of planktonic growth. Iron deprivation could serve as a first step toward eradication of Pierce's disease via blockage of biofilm formation.
Keywords
Introduction
Xylella fastidiosa (Xf) is a gram-negative phytopathogen that can form biofilms, broadly defined as “communities of microbes associated with a surface, typically encased in an extracellular matrix”. 1 Xf has been isolated in different global locations infecting a variety of commercially important plants. One of the 12 genes that regulate exopolysaccharides, a major component of biofilm, is aconitase, which responds to intracellular iron levels. Iron is also essential for the growth of pathogenic bacteria. 2 It was thus considered to be of interest to investigate the potential role of metal chelators in biofilm formation in Xf.
We have incubated Xf (Napa, CA) in the presence of three different metal chelators and measured biofilm formation as well as growth of planktonic cells (liquid phase). Lactoferrin (LF) is a glycoprotein that belongs to the iron transporter or transferrin family. LF is present in milk and in exocrine secretions and is released from neutrophil granules during inflammation. Possible antibacterial activity of LF could be due to its high affinity for iron (Kd ∼ 10−22 M). 3 Due to its availability, conalbumin was also used. Conalbumin is also known as ovotransferrin and is an iron transporter. S, S' -ethylene-diaminedisuccinic acid (EDDS) is a structural isomer of EDTA and has been used as a biodegradable substitute for EDTA. 4
In this study, we have employed a recently developed quantitative assay for biofilm formation referred to as minimal biofilm elimination concentration (MBEC) assay for physiology and genetics (P&G) that is amenable for high-throughput screening. Traditionally, flow cell systems are used to produce biofilms in observation chambers under a wide range of flow rates and nutrient conditions. This assay method is considered to be an open system allowing the continual replenishment of fresh nutrients, in a manner similar to “field conditions” observed in the environment. Biofilm flow cell systems have been commonly used to study the attachment of single bacterial, fungal, or algal cells to various surfaces. By counting individual cells over time the surface concentration (cells/unit area) and accumulation rate can be calculated for assessing the fouling potential of different surfaces. 5,6 A flow cell available from Stovall Life Science, Inc contains triple channels covered with a glass cover slip, attached to the cell with acrylic adhesive. Typically flow cell systems are able to hold up to four flow channels that provide 12 growth channels. This method can be used to study microbial communities under confocal laser-scanning microscope for structural analysis. Compared to traditional flow cell systems containing 12 different growth chambers, the MBEC assay is capable of producing 96 statistically equivalent biofilms. The MBEC assay can be used to test a range of dilutions of antimicrobial compounds, allowing a study of effectiveness of antimicrobials in a high-throughput fashion. The design of the MBEC assay includes pegs that insert into each well of the 96-well microplate, facilitating the separation of planktonic and biofilm bacterial populations that normally requires multiple steps.
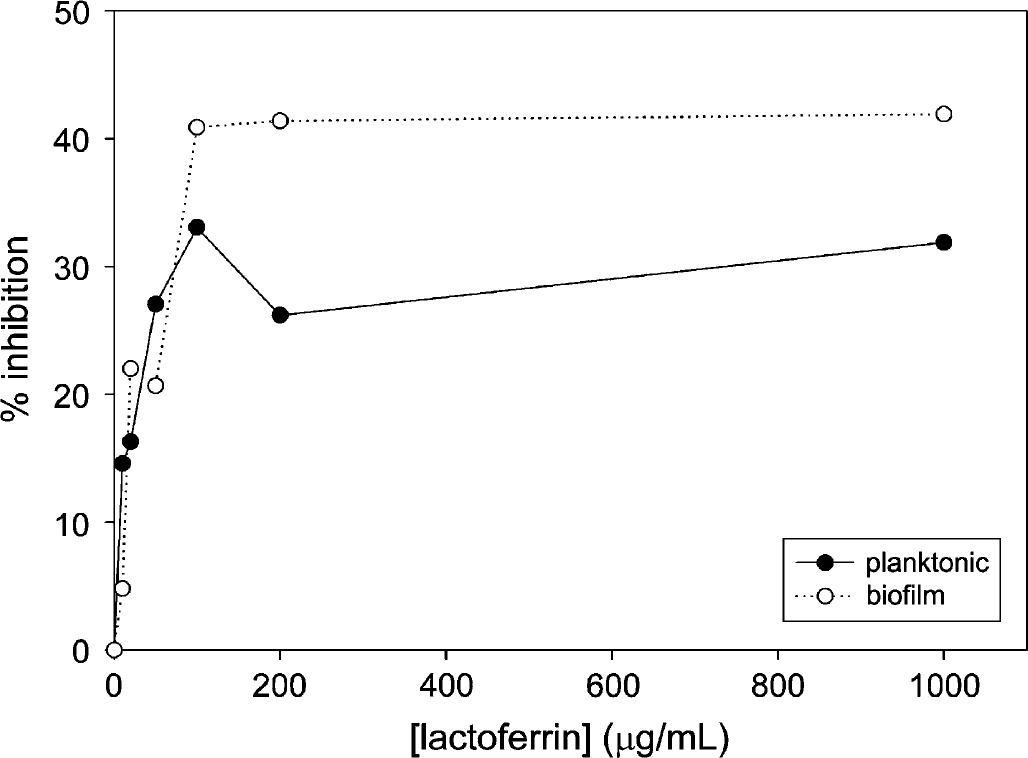
Inhibition of Xf planktonic growth and biofilm formation during 3.5 days of incubation in the presence of LF. Each data point shown represents the average of 10 wells.
Materials and Methods
Xf was purchased from the American Type Culture Collection (ATCC number 700963 isolated from Napa, CA) under a United States Department of Agriculture permit (USDA permit number 65800). Xf was inoculated in ATCC medium/2202 PWG medium, except L-glutamate was substituted with L-glutamic acid due to its solubility. Liquid culture was incubated with no shaking at 28 °C until it achieved an optical density of greater than 0.5 at 600 nm. Biofilm formation and planktonic growth were measured using the MBEC assay for P&G (MBEC Bioproducts, Edmonton, Canada) according to the manufacturer's protocol. Briefly, 150 μL of liquid culture, 25 μL of a metal chelator at various concentrations, and 25 μL of 14.4 mM of NaHCO3 were placed into each well of an MBEC P&G assay 96-well microplate and incubated for various periods at 28 °C. MBEC microplates include pegs inserted into each well treated to promote biofilm formation on the surface of the peg. The microplate pegs were then washed with phosphate-buffered saline solution to remove planktonic phase, and transferred to a fresh 96-well microplate containing 150 μL of media, 25 μL metal chelator solution, and 25 μL of 14.4 mM NaHCO3 per well for 24 h at 28 °C. Optical density was then measured at 600 nm as a readout for planktonic growth in the original microplate. Microplate pegs were placed in a 96-well microplate containing 150 μL media, 25 μL of metal chelator, and 25 μL of 14.4 mM NaHCO3 and sonicated for approximately 12min (Bransonic Ultrasonic cleaner model 3510) to dislodge surviving biofilm. Optical density was then measured at 650 nm as a readout for biofilm formation.
Results and Discussion
Xf was incubated in the presence of LF or conalbumin as substitute in the range of 0-1000 μg/mL over the course of 5.5 days, and optical density was measured in approximately 24 h intervals (see Figs. 1–3). Incubation of Xf with LF at 1000 μg/mL over a time course showed the highest inhibition after 3.5 days—31.8% for planktonic phase and 41.9% for biofilm. Xf was also incubated with EDTA for 24 h ranging from 0 to 50 mg/mL (see Fig. 4). Incubation of Xf with EDTA at 15 mg/mL for 1 day resulted in almost complete eradication of both planktonic and biofilm. Planktonic growth was inhibited by 98.9% and biofilm formation was inhibited by 99.7%. Xf was incubated with EDDS in the concentration range of 0-38.2 mg/mL for 24 h (see Fig. 5). Incubation of Xf with EDDS at 38.2 mg/mL for 1 day inhibited the planktonic phase by 34% compared to 65% for biofilm formation.
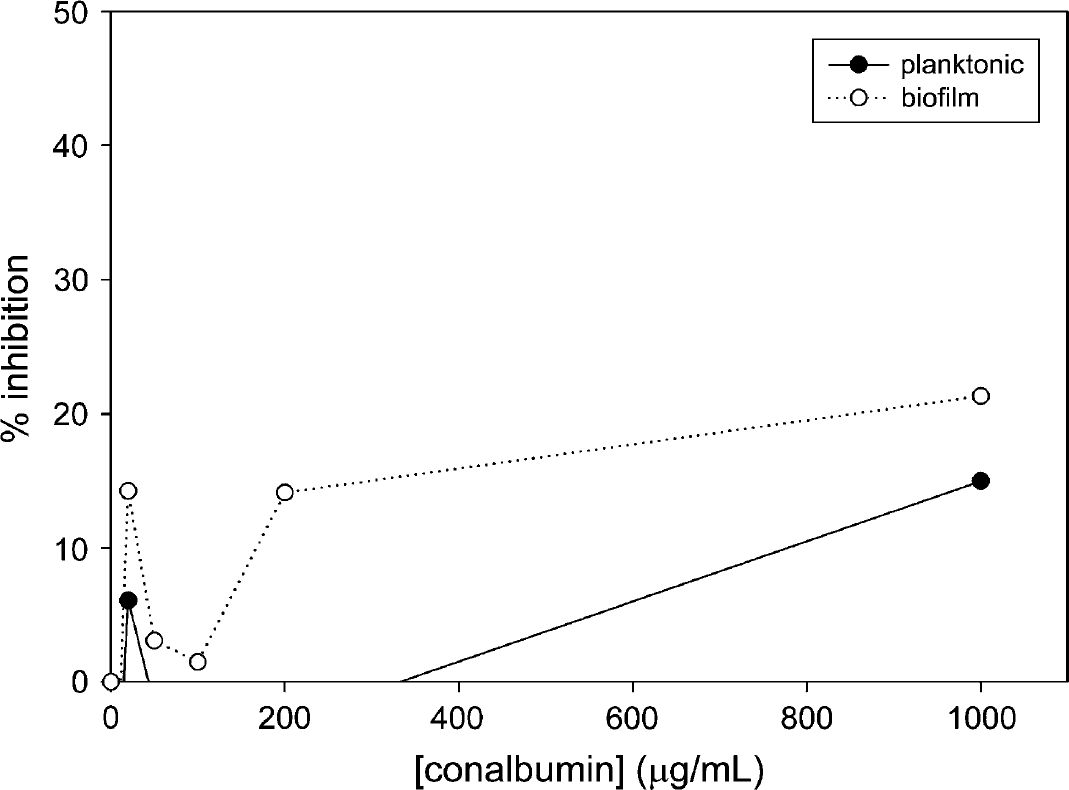
Inhibition of Xf planktonic growth and biofilm formation during 2 days of incubation in the presence of conalbumin. Each data point shown represents the average of 10 wells.
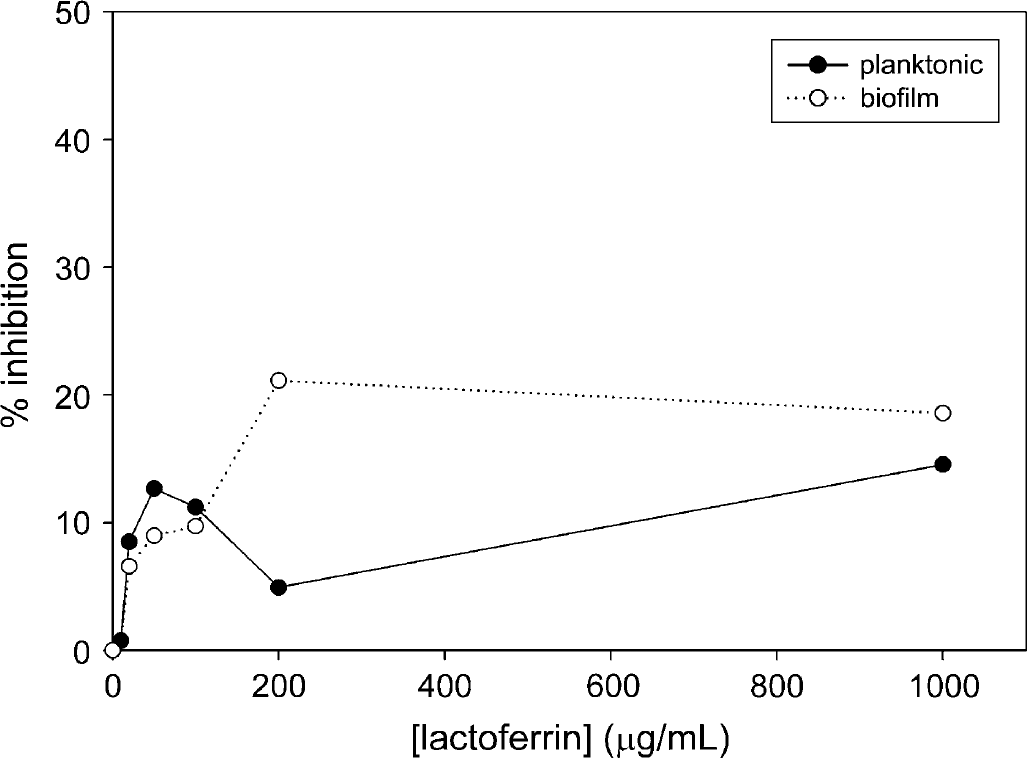
Inhibition of Xf planktonic growth and biofilm formation during 5.5 days of incubation in the presence of LF. Each data point shown represents the average of 10 wells.
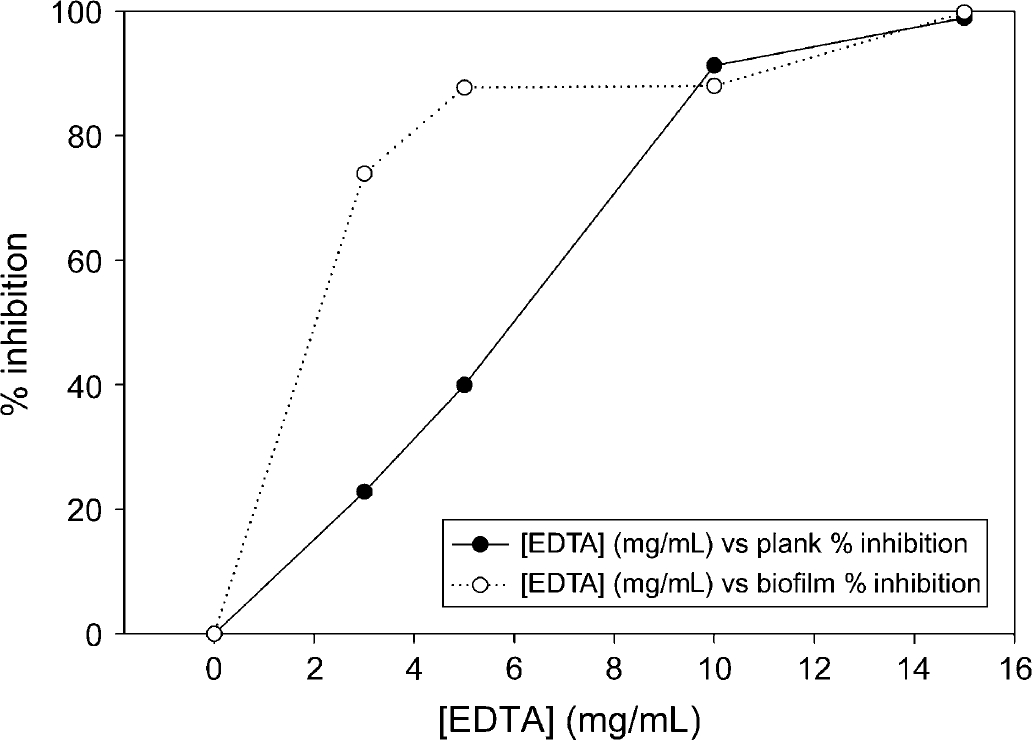
Inhibition of Xf planktonic growth and biofilm formation during 24 h of incubation in the presence of EDTA. Data points greater than 15 mg/mL are omitted due to inhibition > 100%. Each data point shown represents the average of 10 wells.
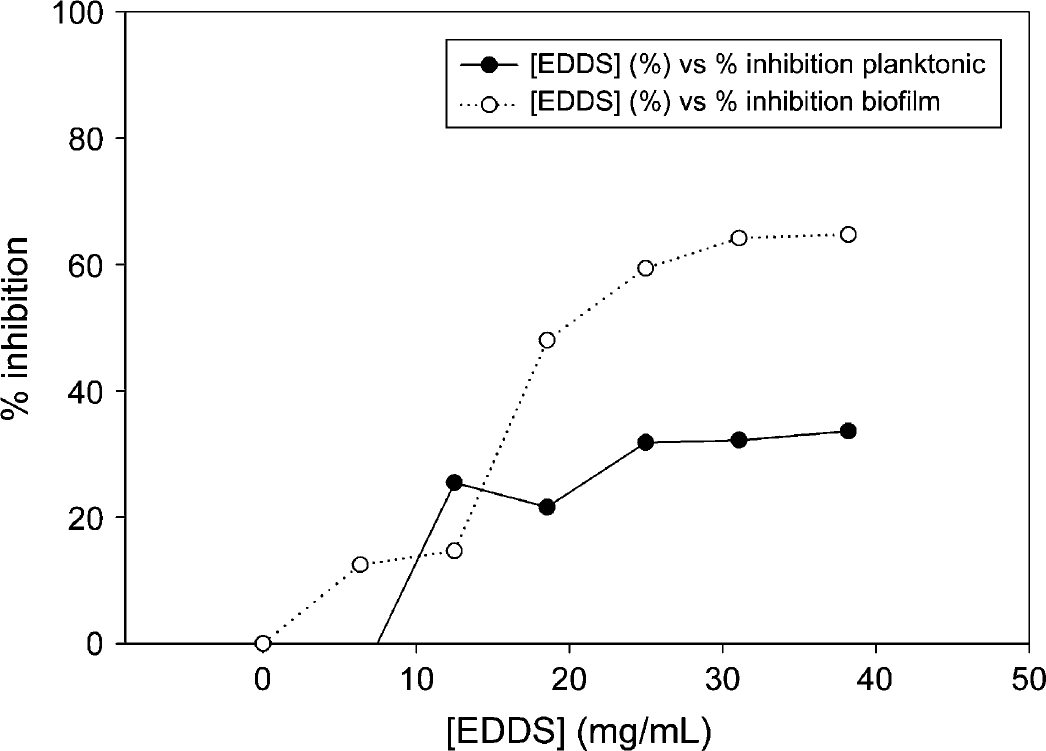
Inhibition of Xf planktonic growth and biofilm formation during 24 h of incubation in the presence of EDDS. Each data point shown represents the average of 10 wells.
Traditional assays for biofilm formation have employed flow cells that are not amenable for high-throughput screening. 7 This study has made use of the MBEC assay designed to produce biofilms in 96 individual wells under sheer stress, allowing for rapid screening for compounds that can block biofilm formation. Pegs of the MBEC lid are designed so that biofilm can be easily removed by sonication, followed by a quantitative measurement using a standard microplate reader.
Taken together, our studies indicate that Xf biofilm is more sensitive to iron levels in the environment compared to the planktonic phase. This approach could be a first step toward treatment of Pierce's disease using metal chelators.
Acknowledgements
This work was supported by the Herman and Margaret Sokol endowment.
