Abstract
A high-throughput cell-based fluorescence screening assay was used to determine the cytotoxic activity of various cytostatic reference agents, unknown compounds of natural sources and pharmaceutic standard excipients in different cell lines such as HEK-293 and DLD-1 cells. In this paper we exemplarily show results for a selection of compounds and excipients. The cytostatic agent piposulfan has a much higher cytotoxic activity in DLD-1 cells (EC50=0.001 mg/mL) than in HEK-293 cells (EC50=0.3 mg/mL) in contrast to busulfan and vinblastine where the EC50 values are close together in both cell lines.
From 113 tested compounds from natural sources the cytotoxic activity of 75 compounds showed no difference in both cell lines, 34 of them had a higher activity in HEK-293 cells than in DLD-1 cells and 4 compounds showed less cytotoxic activity in HEK-293 cells than in DLD-1 cells. We demonstrate the rather potent cytotoxic action of Cremophor EL; this finding is in accordance with previously published observations. The presented results demonstrate that the used assay system is reliable, enables the ranking of the cytotoxic potential of compounds of various chemical classes, and allows the determination of cell type-specific cytotoxicity. (JALA 2004;9:159-62)
Introduction
Huge efforts are made to find effective treatments against cancer. New compounds are screened for anti-cancer activity, 1 known compounds are used in new combination or treatment regimens. 2
The toxicity of most compounds is very un-specific, damaging neoplastic tissue as well as healthy tissue. Cytostatic agents that are drug candidates for the treatment of cancer are generally tested in animal models for efficacy and toxicity. 3,4 A preliminary test can also be done at lower cost and with higher throughput capacity in carcinoma and in normal cell lines. 1,5
Toxicity and pharmacological activity of cytotoxic drugs were tested in a robotic high throughput screening (HTS) cell assay, which allows the prediction for the efficacy of the drug in different carcinoma cell lines and for toxic effects in non-carcinoma cells. The toxicity data are a tool to distinguish between two drug candidates with equal activity but different toxicity.
The drug candidates are frequently either of low solubility or of low stability when conventional formulation components are used. 6,7 That is why different formulation components were included in the test.
Materials and methods
A robotic high-throughput screening system was used to carry out the assay.
Cells of different lines (e.g. HEK-293, DLD-1) were seeded (50 μL cell suspension, 2×105 cells/mL) into 96 well plates (Cellstar®, Greiner Bio-One Ltd., Frickenhausen, Germany) and incubated overnight (37°C, 5% CO2)to allow the cells to recover. Then test compounds in serial dilutions were added (200 μL/well) followed by a further incubation for 48 h (37°C, 5% CO2). After incubation a tetrazolium salt based indicator dye (EZ4U, Biomedica Ltd., Vienna, Austria) was added and the cells were allowed to metabolize the dye (see Fig. 1) for 1–2 h at 37 °C/5% CO2. The absorption which directly correlates with the cell number was measured at 492 nm.
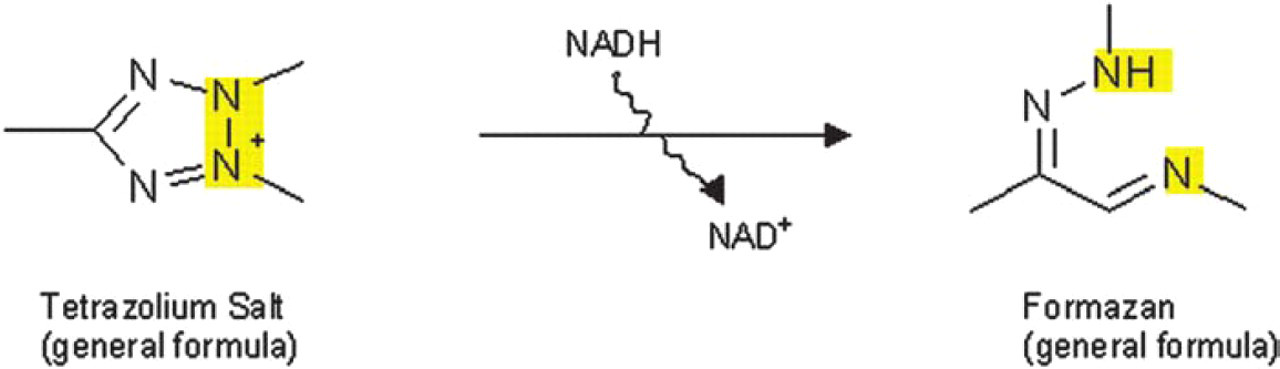
Principle of the reduction of a tetrazolium salt to a colored formazan by metabolically active cells.
The accumulation of dye in the remaining vital cells was plotted against the dilution of the test substance. The concentration of test substance where the absorbance is half of the maximum was calculated (EC50) and used for comparison of the cytotoxic activity of the test compounds. The activity was calibrated against the activity of substances with known cytotoxic activity and toxicity.
Results
The activity of cytostatic drugs and compounds from natural sources as well as of several commonly used formulation components such as Cremophor EL, PEG 400, DMSO, acetone, and β-cyclodextrin were tested against carcinoma and non-carcinoma cell lines. Figures 2 and 3 show exemplarily the cytotoxic activity of different compounds in HEK-293 (human emryonic kidney) cells compared to the human colon carcinoma cell line DLD-1.
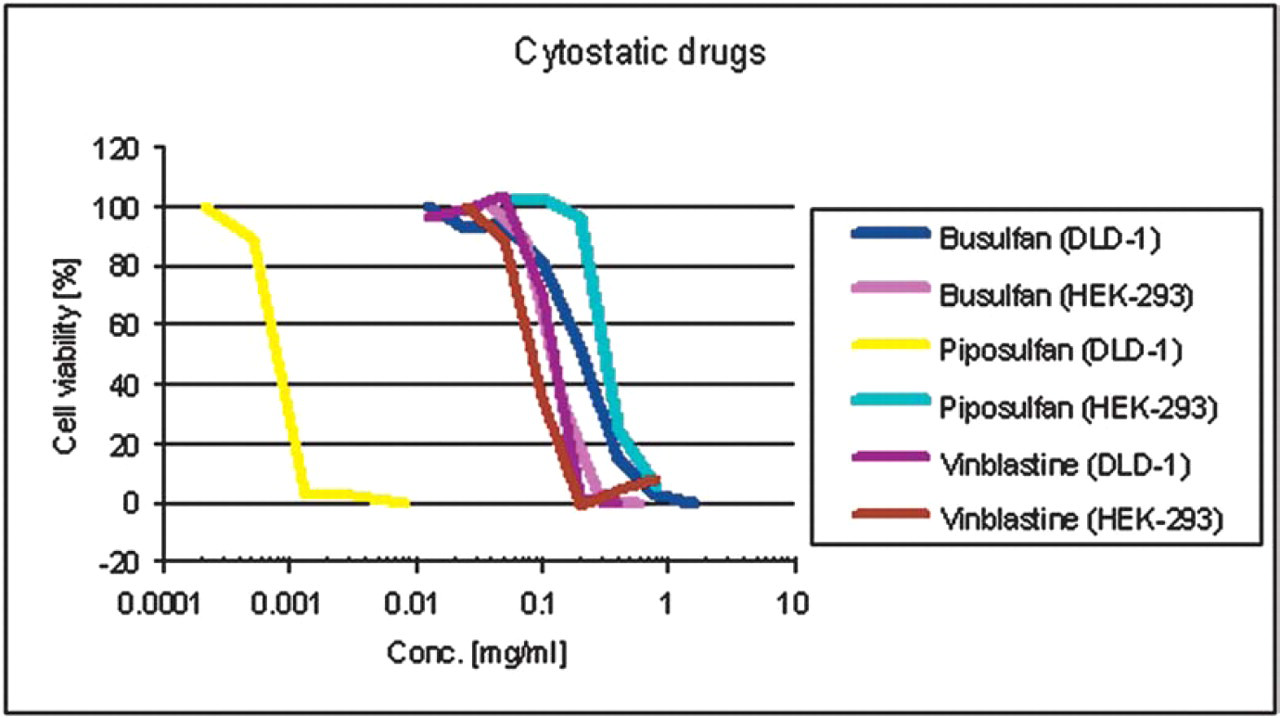
Cytotoxic activity of busulfan, piposulfan, and vinblastine in HEK-293 cells compared to cell line DLD-1.
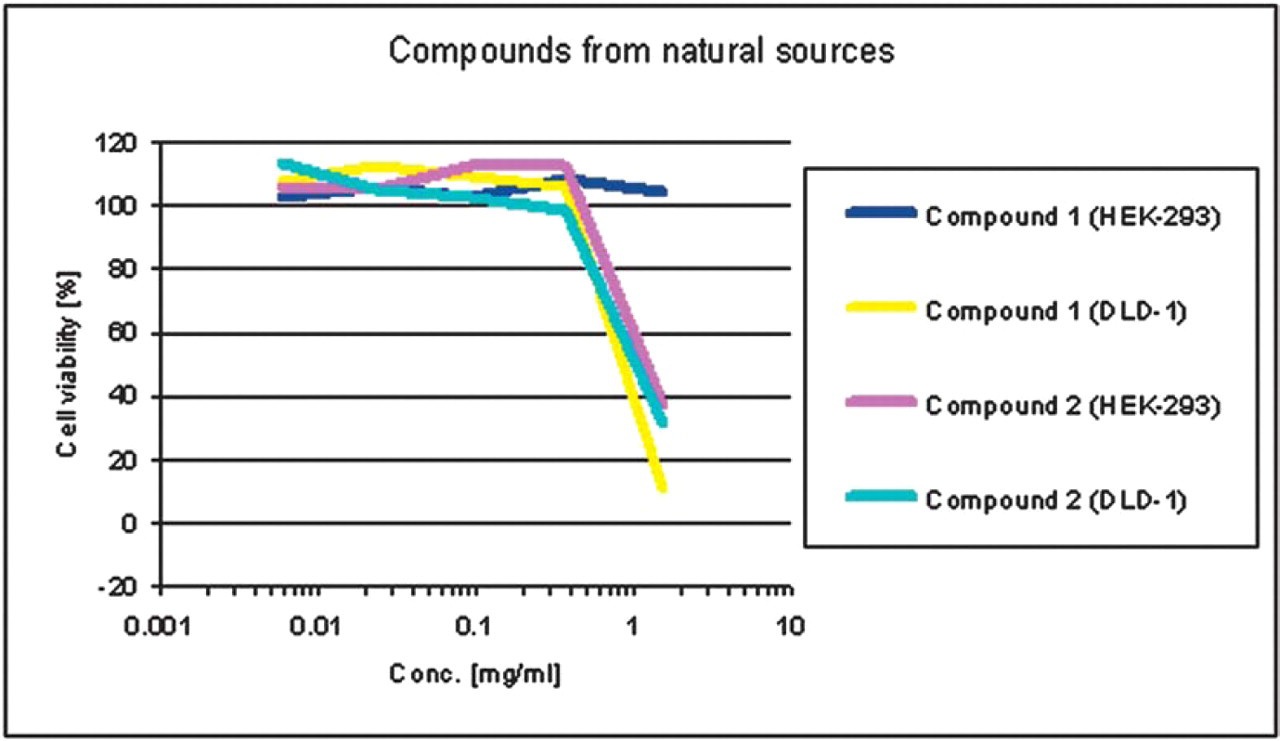
Cytotoxic activity of compounds from natural sources in HEK-293 cells compared to cell line DLD-1.
Figure 2 illustrates that the cytostatic agent piposulfan has a much higher cytotoxic activity in DLD-1 cells (EC50 = 0.001 mg/mL) than in HEK-293 cells (EC50 = 0.3 mg/mL) in contrast to busulfan and vinblastine where the EC50 values are close together in both cell lines.
Figure 3 shows exemplarily the results from two unknown compounds from natural sources where compound 1 has higher cytotoxic activity in DLD-1 cells (EC50 = 0.9 mg/mL) than in HEK-293 cells (EC50 > 2 mg/mL). Compound 2 has the same cytotoxic activity in both cell lines.
From 113 tested compounds from natural sources the cytotoxic activity of 75 compounds showed no difference in both cell lines, 34 of them had a higher activity in HEK-293 cells than in DLD-1 cells and 4 compounds showed less cytotoxic activity in HEK-293 cells than in DLD-1 cells.
In Figure 4 the graphs of five different formulation components in DLD-1 cells are presented. The results show that the neutral formulation component Cremophor EL has a relatively high cytotoxic activity (EC50 = 0.03 %) compared to the other tested excipients (EC50 values in the range of 4–12 %).
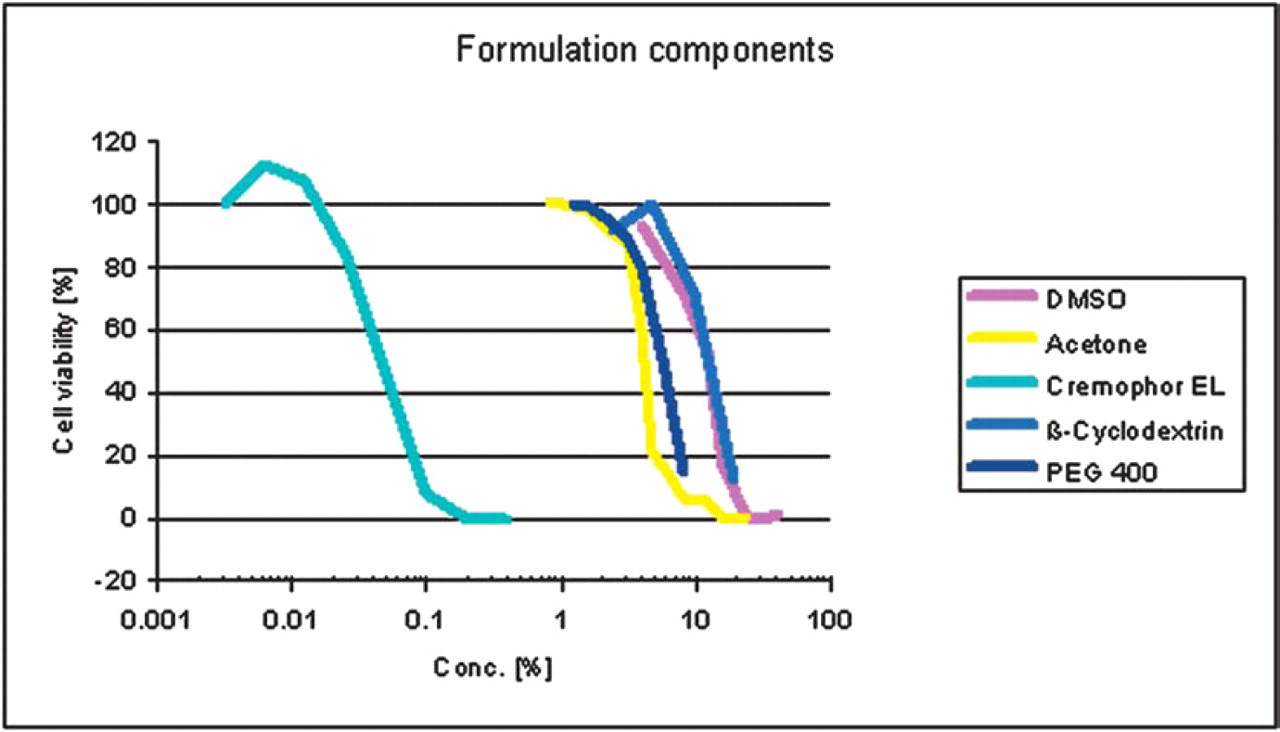
Cytotoxic activity of several formulation components in DLD-1 cells.
CONCLUSION
Our data demonstrate that the used cell assay is reliable and enables the ranking of cytotoxic compounds according to their relative cytotoxicity; this was examplified for a series of cytostatic reference drugs and unknown compounds of natural sources, respectively. This assay is suitable for screening of cytotoxic compounds for efficacy in carcinoma cell lines as well as for toxic effects in non-carcinoma cells. Furthermore, the assay is also indicative of cytotoxic effects of excipients. Thus, we could demonstrate the rather potent cytotoxic action of Cremophor EL; this finding is in accordance with previously published observations which suggest a synergistic action of the excipient with various antitumor drugs. 8,9
Acknowledgments
The authors thank the Institute for Biomedical Technique (IBMT), University Rostock, for cultivation and continuous supply with several cell lines.
