Abstract
The Department of Chemical Pathology of the University of Zimbabwe (Medical School) has set up an external quality assurance scheme, Zimbabwe External Quality Assessment Scheme (ZEQAS) to assess the quality of clinical chemistry laboratory performance in Zimbabwe. The programme has been in place since 1989. 43 clinical chemistry laboratories, both public and private, are currently participating in the quality assessment scheme. Analytical laboratory performance for participants is examined for twelve analytes. A standardized computer software package is used to process data and for producing performance charts. The information highlighted by a computer print-out for each participating laboratory includes the following: MRVIS (mean running variance index score) for all analytes, overall MRVIS for the laboratory, table of results for each analyte (with its mean value and cofficient of variation) and trimmed results for each analyte by individual method which include number of laboratories using a particular method, mean results for that particular method, SD (standard deviation) for that particular method and CV (coefficient of variation) for that particular method. Since the introduction of ZEQAS, there has been a drastic improvement in clinical chemistry laboratory performance in Zimbabwe. The overall mean running variance index score (OMRVIS) for all laboratories over a period from 1990 to 1999 has improved from 163 to 98.
INTRODUCTION
Quality control has been considered as one of the most essential tools to evaluate the quality of clinical laboratory data. Clinical laboratories seek to achieve the quality control goals through quality assurance programmes, which include pre-analytical, analytical and post-analytical elements of performance (Whitehead, 1976). These laboratories need to have comprehensive internal quality control programmes and need to participate in external quality assessment (EQA) schemes as an integral part of good laboratory practice (Tan et al., 1990). Although EQC schemes are important in laboratory medicine, the majority of developing countries are lagging behind in concepts and implementation of sustainable quality assurance practices. In Zimbabwe, most clinical chemistry laboratories are participating in the Zimbabwe External Quality Assessment Scheme (ZEQAS). ZEQAS was found in 1989 by the University of Zimbabwe, Medical School's Department of Chemical Pathology with the assistance of the Department of Clinical Chemistry, Wolfson Research Laboratories, Queen Elizabeth Medical Centre, Birmingham, UK. The main objectives for the establishment of ZEQAS were to provide independent information on the quality of clinical chemistry tests in laboratories which provide patient care services; to improve the quality of test results and promote agreement between laboratories; to provide a secure basis for the establishment and development of ZEQAS in clinical chemistry; to provide and promote training in principles and practice of quality assurance; to identify laboratories with problems and assist them in improving their performances and to provide a secure basis for a potential regional EQAS within Southern African community
MATERIALS AND METHODS
Participants: Clinical chemistry laboratories were recruited and forty-three are currently participating in the programme. The participants are from both Government public health laboratories and the private sector. The majority of the laboratories are from urban areas.
SPECIMEN DISTRIBUTION AND RESULT ANALYSIS
Lyophilised bovine serum in labelled vials is distributed by post to each of the participating laboratories on a monthly cycle. The specimens are normally reconstituted with 5 or 10 ml of water before distribution. The results are returned by post to the Department of Chemical Pathology to be evaluated by a computer programme developed and donated by the Wolfson Research Laboratories. Data entry for all laboratories is done soon after receiving the results. The computer programme generates important information for all tests and laboratories. The report produced for each participate includes MRVIS for all analytes, overall MRVIS for the laboratory, table of results for each analyte and trimmed results for each analyte. After data processing, individual laboratory reports are sent to participants. The processed data shows how each laboratory or analyte is performing by the value of the MRVIS and other variables. The data also shows how all laboratories or analytes are performing by the values of overall MRVIS (OMRVIS).
RESULTS
Laboratory performance
The overall laboratory performance of all participants has greatly improved since 1990 (Figure 1). This is illustrated by the drastic fall of OMRVIS from above 160 in 1990 to about 98 at the beginning of 1999. There has been a sudden fall of OMRVIS after the participants' workshop in 1992 (Figures 1 and 2). The performance of most laboratories improved after this workshop (Figure 2).
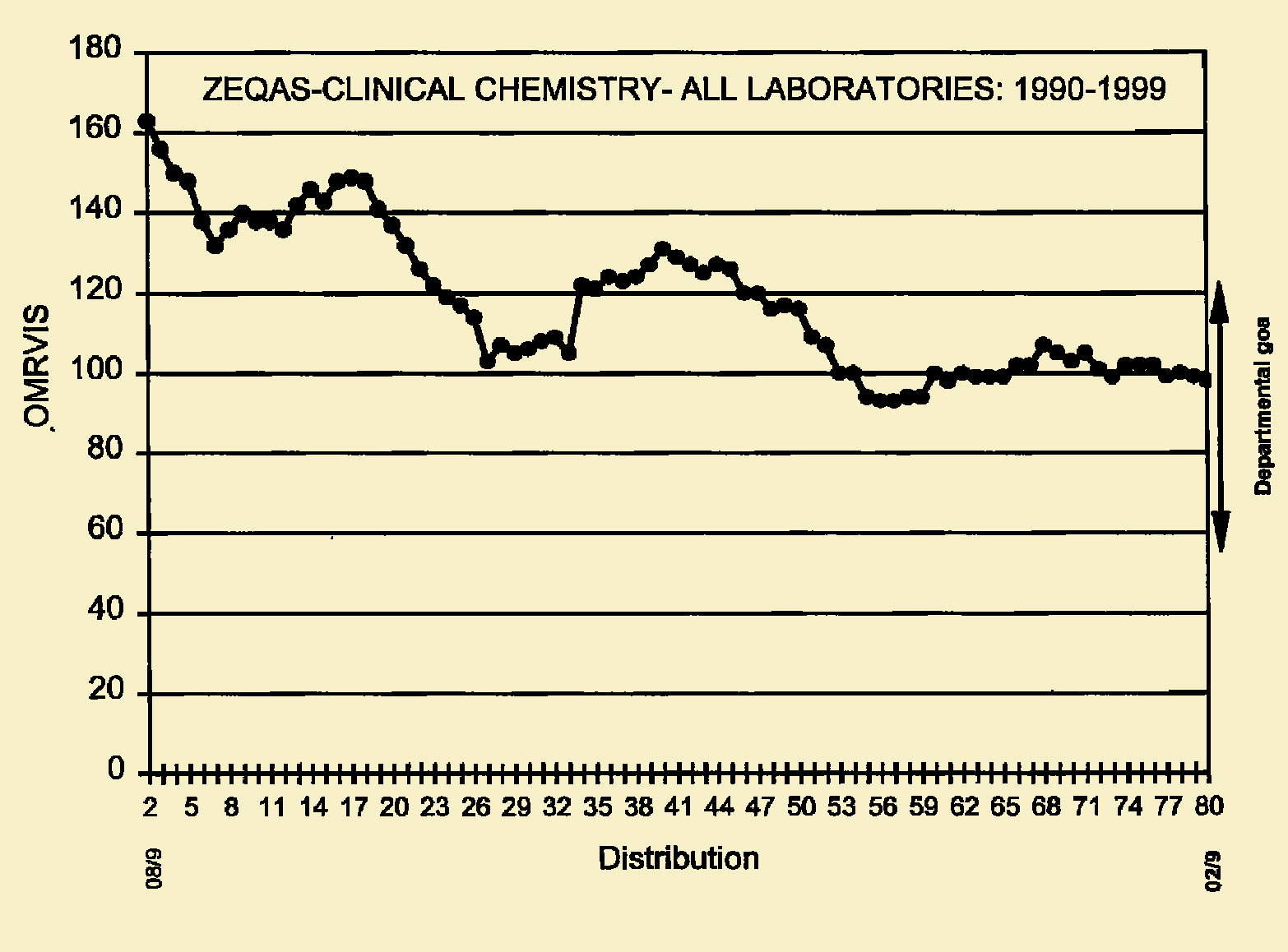
Changes in laboratory performance over time. Note that the training workshop was held at distribution 16 and ZINQAP was launched at distribution 40. There was deterioration of the overall laboratory performance between distribution 26 and 39. This was probably attributed to the joining of new participants into the scheme.
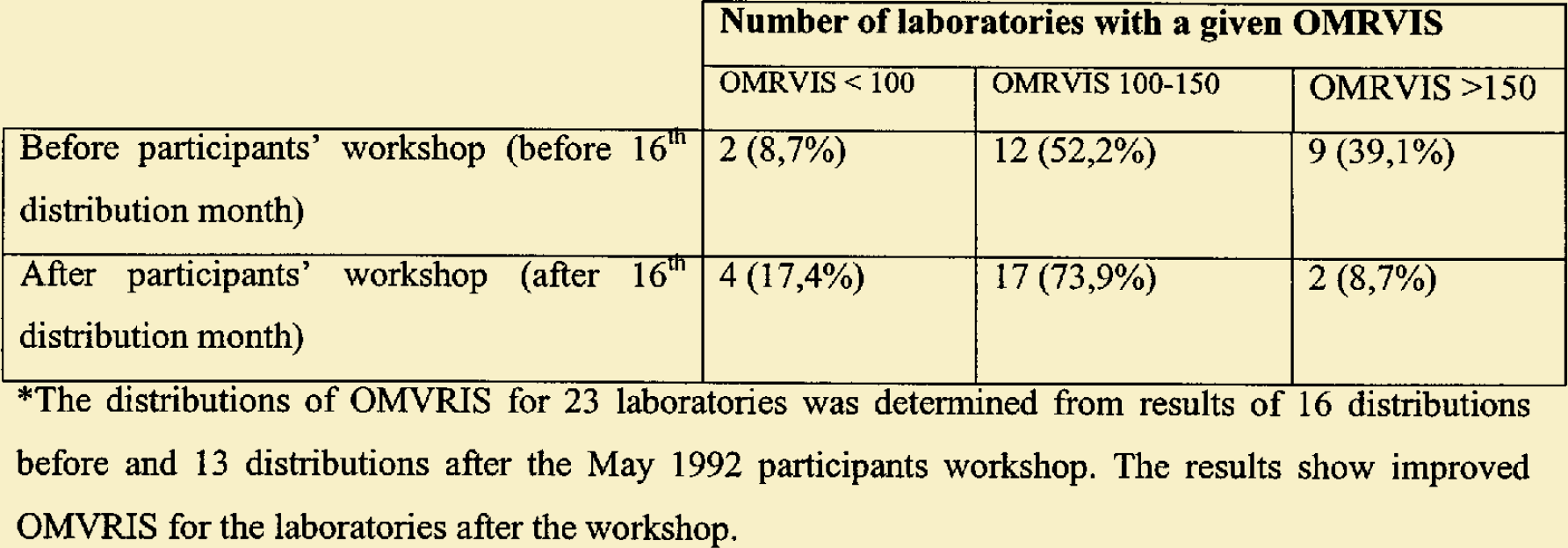
Zimbabwe EQA distribution of OMRVIS before and after participants meeting in May 1992*. (Numbers refer to numbers of laboratories in each category).

Zimbabwe EQA distribution of OMRVIS before and after participants meeting in May 1992*. (Numbers refer to numbers of laboratories in each category).
Analyte performance
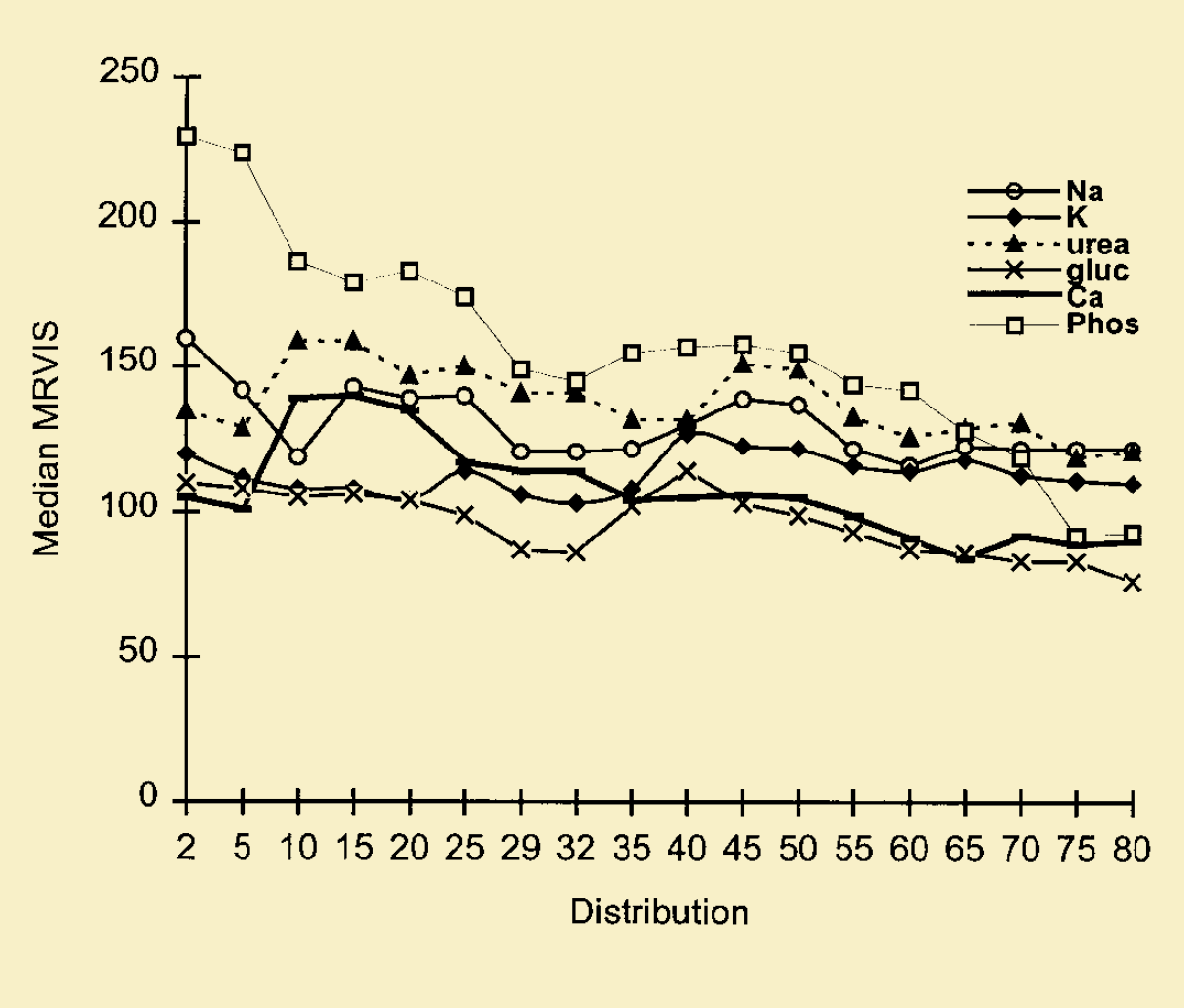
Median MRVIS is used to assess performance of each analyte by all participants. Figures 3 and 4 show the performance of all the participants for each of the 12 analytes (urate, creatinine, bilirubin, total protein, cholesterol, albumin, sodium, potassium, urea, glucose, calcium and phosphate). Creatinine and glucose are doing very well with MRVIS of less than 80 at distribution 80. For creatinine, the MRVIS dropped from 145 (distribution 2) to 79 (distribution 80), which is really a great improvement. Other analytes, mainly total protein, urea and sodium do not show much improvements and their MRVIS is still above 120 (at distribution 80) (figures 3 and 4).
SCHEME VALIDATION
A number of studies have been essential to ensure that good reliable material was distributed to laboratories. Firstly, a study was carried out to determine the stability of various analytes in samples preserved in ethylene glycol under various conditions. The analytes have been found to be stable for more than 7 days at room temperature, confirming experience elsewhere. Comparisons of consensus values from ZEQAS with those obtained in the UK NEQAS for the material provided by Wolfson Research Laboratories was then carried out. The results confirmed close agreement. Graphical analyses of between laboratory consensus values (cv) against concentration for each analyte was done. This revealed no significant differences in pattern for the liquid serum and lyophylised materials. The behaviour of the liquid material was therefore considered for use in the scheme. The validity of the material used and of the designated values for the assessment of laboratory performance were therefore established.
PUBLICATIONS AND CONFERENCES
Since its inception, ZEQAS has generated results which have already been published and presented at conferences. Results were presented at conferences in South Africa (1992), Kenya (1993), Australia (1993), UK (1996), Japan (1996) and Italy (International Federation of Clinical Chemistry, 1999). Manuscripts have been prepared for publication in medical journals for use by participants and scientists. An article entitled “The Zimbabwe External Quality Assessment Scheme (ZEQAS) in Clinical Chemistry: Results of a Pilot Programme” were published in the Central African Journal of Medicine (Mujaji et al., 1994). ZEQAS hopes to make more publications as a way of further promoting its communication with participants, updating knowledge on quality control and dissemination of results to other external quality assessment programmes within the region and the international arena.
Number of laboratories with a given OMRVIS OMRVIS < 100 OMRVIS 100–150 OMRVIS >150 Before participants' workshop (before 16th distribution month) 2 (8,7%) 12 (52,2%) 9 (39,1%) After participants' workshop (after 16th distribution month) 4 (17,4%) 17 (73,9%) 2 (8,7%)*The distributions of OMVRIS for 23 laboratories was determined from results of 16 distributions before and 13 distributions after the May 1992 participants workshop. The results show improved OMVRIS for the laboratories after the workshop.
DISCUSSION
The past decade has seen great achievements by ZEQAS. A fall in the overall mean running variance index score (Figure 1) shows that the clinical chemistry laboratories are now performing exceptionally well in Zimbabwe. Workshops such as the one launched in 1992 also contributed to improved laboratory performance. Communication between participants and scheme organisers at workshops, introduction of training, up-dating knowledge and quality assurance awareness have all contributed to improvement of OMRVIS for participants from above 160 in 1990 to 98 in 1999. The programme can now be rated comparably with the long established UK NEQAS. In a pilot programme, the consensus values obtained from ZEQAS agreed closely to those of UK NEQAS despite the relatively small number of participants in Zimbabwe (Mujaji et al., 1994). Median MRVIS for most analytes are also falling, showing laboratory improvement in performance. Nevertheless, there are some laboratories and analytes with very high MRVIS. Most of these laboratories are likely to have problems such as poor laboratory management, problems with instruments, reagents or calibrators and method shortcomings. Despite these problems, ZEQAS envisage further successes and great strides in clinical chemistry in the next millenium. It has been proposed that the range of analytes be expanded to include enzymes (such as lactate dehydrogenase, alanine and aspartate aminotransferases, alkaline phosphatase, gamma-glumyltransferase, alpha-amylase, creatine kinase, acid phosphatase, pseudo-cholinestrase and hydroxybutyrate dehydrogenase), tumuor markers (such as alpha—fetoprotein, carcinoembryonic antigen, tissue polypeptide antigen, carbohydrate antigens, prostatic acid phosphatase and prostatic specific antigen) and hormones. The scope of the scheme is also expected to increase the number of participants by inclusion of laboratories at district level. There is also a potential for collaborative activities of ZEQAS with neighbouring countries and participation of laboratories in Southern and Central African countries such as Zambia, Namibia, Botswana, Malawi, Mozambique, Lesotho, Swaziland and South Africa. Continued education and professional training will continue to be essential so as to reinforce the use of the most appropriate quality assurance measures. Holding seminars and visiting laboratories showing unsatisfactory and successful performance are also key to the success of ZEQAS. Currently ZEQAS operates under ZINQAP (Zimbabwe National Quality Assurance Programme) which monitors both external and internal quality assessment schemes in all disciplines of pathology.
