Abstract
The authors automated an enzyme-linked immunosorbent assay to detect porcine serum antibodies to Toxoplasma gondii. Two thousand swine sera can be assayed in two eight-hour shifts using a robotic workstation. The automated-ELISA programming is not complex and the test configuration is flexible. This high-throughput screening (HTS) a-ELISA can achieve a 10-fold increase (100→ 1000 tests) in test capacity over the manual method. The assay has been validated according to the requirements of the ISO/IEC 17025 standard. These include repeatability, reproducibility, and optimal threshold value studies. Other requirements are proficiency panel testing, analyst training, standard operation procedure, and equipment certifications.
Introduction
A number of manual enzyme-linked immunosorbent assays (ELISAs) for detecting antibodies to Toxoplasma gondii have been published.1–5 Automated ELISAs (a-ELISA) for detecting different pathogens and their specific antibodies have been developed.6,7 In this paper, we have described the use of a robotic liquid-handling workstation in conjunction with a mini sample-tube system to automate a manual ELISA for high-throughput screening (HTS) of swine antibodies to T. gondii.
T. gondii is the causative agent of toxoplasmosis. It is a protozoan parasite with worldwide distribution that affects man and other warm-blooded animals. 8 Cats, including wild Felidae, are the only definite hosts. Swine are considered a major source of human infections by this parasite. 9 The United States Department of Agriculture (USDA) has listed toxoplasmosis as one of the most important food-borne diseases linked to red meat in the U.S. 10
The greatest food safety concern attributed to this parasite is for a woman to consume infected pork and acquire the parasite during pregnancy. If she does not already have protective antibodies against T. gondii, there is a 20 to 50% probability that her fetus will be infected. 11 Many of the infected fetuses will have disease manifestations, including chorioretinitis and mental retardation, later in life. 12 In the U.S., the estimated annual cost of treating these patients was $5.3 billion in 1993. 13 With a worldwide trend of escalating medical costs, the most effective method to minimize the social and economic impacts of this disease is by removing the cause of infection. In order to control the spread of this parasite, the U.S. has recently completed a large-scale surveillance study on T. gondii. The USDA screened over 14,000 porcine sera nationwide for T. gondii antibodies in its National Animal Health Monitoring System (NAHMS) swine 2000 study. 14
The objective of this paper is to automate and test validate a commercial T. gondii antibodies ELISA and transform the protocol into a high-throughput, cost-effective assay method that can be employed in any large-scale surveillance study in Canada. We are also required to validate the test protocol based on the requirements of the ISO 17025 standard.
Materials and Methods
Assay Development
The sequence consists of a number of steps, which include the acquisition of reference serum controls (both positive and negative), the selection of target population, sampling/preparation of serum, use of bar code recognition for specimen tracking, the selection of a commercial ELISA and its automation using a robotic workstation, throughput comparison between the manual and automated procedures, ISO 17025 test validation, and interpretation of the results.
Reference Sera
Reference positive serum control (N = 30) and negative serum control (N = 20) were purchased from IVD Research (Carlsbad, CA). They were prepared from T. gondii infected pigs and have been confirmed either positive or negative by both the ELISA and agglutination test by the manufacturer. The serum sample was divided into four aliquots to be used in/as (1) reproducibility and repeatability studies, (2) analyst proficiency testing, (3) reference controls in the a-ELISA, and (4) spare samples. In the test validation experiments, we encrypted the reference sample identities so that the positive or negative nature of the serum controls was unknown to the laboratory analyst during tests, thus avoiding any testing bias.
Test Sera
Finishing pigs (N = 2000) were randomly selected for screening by the a-ELISA for T. gondii antibodies. These were animals within 45–60 days of shipment from the farms. Each animal was assigned a unique bar code number for specimen tracking purposes. Several mL of blood was collected from each animal. About 1 mL of serum was harvested aseptically per blood sample. Only sera devoid of bacterial contamination and haemolysis were used in this project.
Sampling
The sera was collected into a miniature sample tube system (Micronic BV, The Netherlands). Each unit of the system is made up of 96, 1.4 mL polypropylene tubes in a support rack with lid. This system is in the Society of Biomolecular Screening (SBS) standard 96-well plate format. With the identical format being used, direct transferring of serum from any one tube in this system to a corresponding position on a 96-well microplate can be done without possibility of mistake. Each serum occupies the same location, e.g., A1, in the sampling unit as in the ELISA plate. A minimum of 90 serum samples can be stored in each system and kept at −40°C until used.
Specimen Bar Code Recognition and Specimen Tracking
In order to accurately identify and track each specimen throughout the project, a unique bar code label/number was assigned to each animal/specimen. The individual label was generated from a SATO CL (Series “e”) thermal transfer printer controlled by “Label View Pro for Windows” software (Teklynx, Eau Claire, WI). Freezer-proof bar code labels, measuring 1.75″ by 0.25″, were used. The type of bar code symbologies chosen was Code 39. These labels were then attached to the tubes and the miniature sample tube racks (Figure la). A database (MS Excel spreadsheet) was created to store the specimen information (Figure lb), including case number, identification number, animal type, submission type, storage location, test mode, and type of pathogen, which can be linked through the use of the unique bar code assigned to each specimen. A Cobra bar code scanner (Symbol Technologies, Holtsville, NY) was used as an accurate input device to register the bar code number of the specimen into the database. This scanning device eliminates human error. Furthermore, the bar code system was used to create an ELISA worksheet (Figure 2) by assigning each test serum to a designated position on the 96-well microplate. This was used to track specimens throughout the project.

Miniature sample tube system (96-tube format).

Database for specimen tracking using MS Excel spreadsheet.
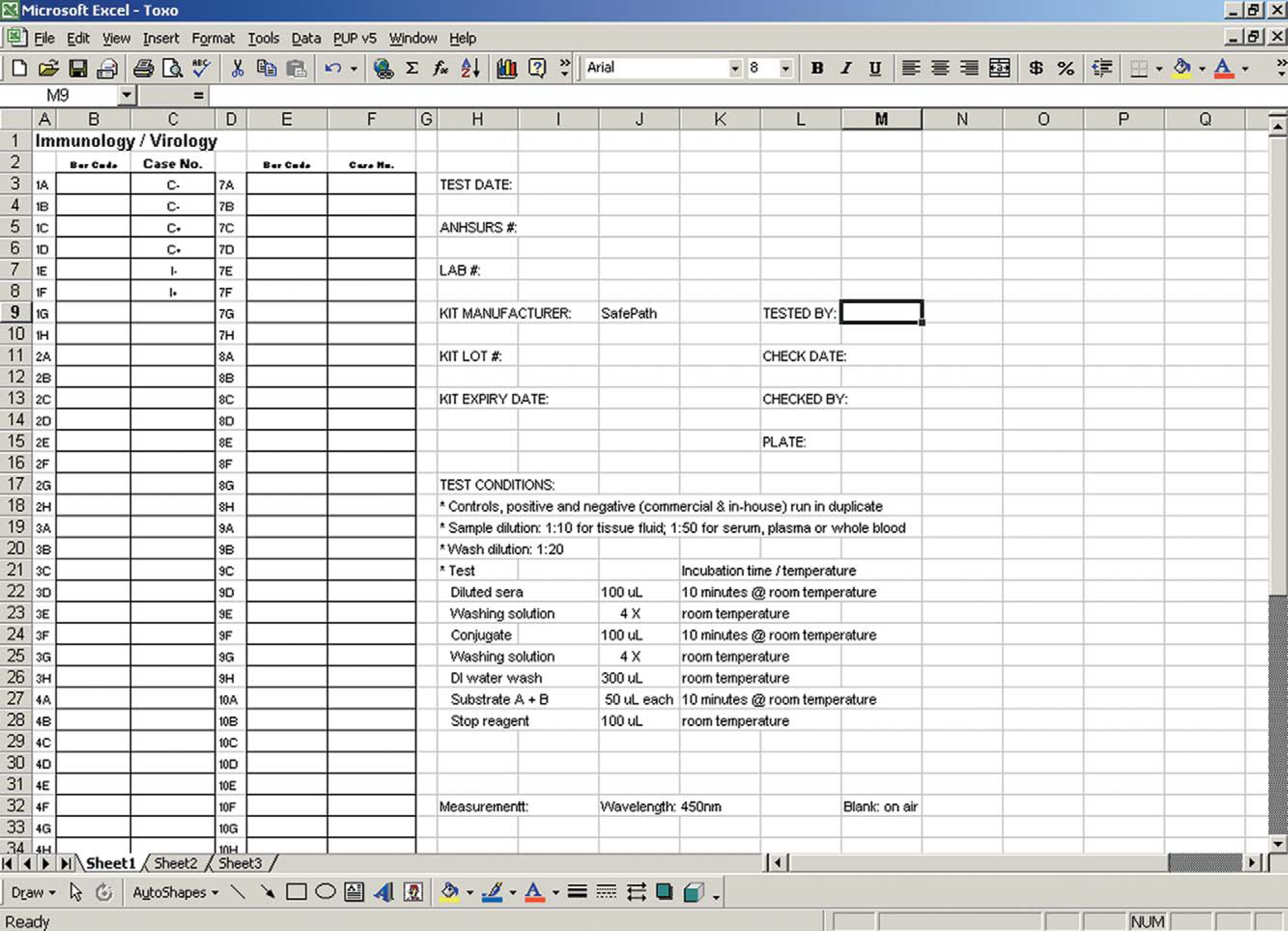
ELISA worksheet designed for a 96-well microplate.
Automated Enzyme-Linked Immunosorbent Assay
A Toxoplasma gondii Antibody Test Kit (SafePath Laboratories, Carlsbad, CA) was purchased (a microwell ELISA test). Specific Toxoplasma antigens are coated in each test well of a 96-well microplate. Porcine serum samples were diluted by simultaneously transferring aliquots of all 96 samples from the miniature tube rack to a microplate containing the diluent buffer. The final dilution was 1/50. During the first incubation, the specific antibodies present in the test sera were bound to the coated antigens in the microwells. After three washings to remove the nonspecific immunoglobulin (IgG), the antigen-antibody complex formed was allowed to bind to an anti-swine IgG peroxidase conjugate. After another three washings to remove the unbound enzyme conjugate, the antigen-antibody–conjugate (enzyme) complex was allowed to react with the chromogen (tetramethybenzidine) and the substrate (hydrogen peroxide). A specific blue color developed. In the last step, a stop solution (1 M phosphoric acid) ended the reaction and turned the color to yellow. The optical density (O.D.) values of the microwells were read with an ELISA reader at 450 nm. According to the manufacturer's recommendation, an absorbance reading greater than or equal to 0.2 O.D. units is considered positive, and an absorbance reading of less than 0.2 O.D. units is considered negative.
The different liquid handling steps were carried out by a Biomek FX® (BFX) robotic workstation (Beckman Coulter, Fullerton, CA), which can handle 96 or 384 microwell transfers. In order to increase the workstation's capacity, it is connected to a Stacker Carousel® (SC) (Beckman Coulter) (Figure 3). The design of this automation unit is “open” with the incorporation of several independent modules (equipment) to the core, or backbone, workstation. This modular design provides high adaptability in the configuration of the equipment, depending on the requirements of the ELISA procedure. The major peripherals include the microplate washer, an ELx405® (BIO-TEK Instruments, Winooski, Vermont, USA) (Figure 4) and the ELISA reader, a VmaxELISA reader® (Molecular Device, Sunnyvale, CA) (Figure 5). Automation flexibility is further enhanced by the use of interchangeable automated labware positioners (ALPs) for specific purposes, such as locations of reagent transfers, disposable tips, plate replications, microplate shaking, and waste discard.

Biomek FX workstation linked to a stacker carousel.

Bio-Tek microplate washer.

Molecular Device ELISA microplate reader.
This automated workstation is equipped with a 96-channel pipetting head, the pod, with an integrated gripper to enhance system throughput. All transfer steps are designed to be performed simultaneously on all 96 wells. This pipetting head produces consistent and reproducible results in the volumes of 1 to 250 μL during plate-to-plate transfers. We have used 20 out of 25 possible positions of the workstation deck in our test. With the linking of the BFX and SC through the use of the integrated gripper of the 96-channel pipetting pod, we can use a total of 20 microplates and tip boxes and 10 reservoirs in our a-ELISA continuously and greatly increase the walkaway time (WAT) of the technologist.
Automation programming was performed with a Pentium III microcomputer with Windows NT® (Microsoft, Redmond, WA) operating system. The integrated software (Biomek 2.1®; Beckman Coulter) controlled the workstation and other peripheral equipment. With this software, we wrote an in-house program specifically for the Safepath ELISA by using the graphic interface and specific icons incorporated in the software. The robotically controlled X-Y-Z positional liquid handling steps were programmed in the order of the ELISA protocols.
Once the reactions of the ELISA were completed, O.D. values from the 96-well microplate were read by an ELISA microplate reader at a wavelength of 450 nm. The O.D. values were transferred from the plate reader, captured, and stored in the computer.
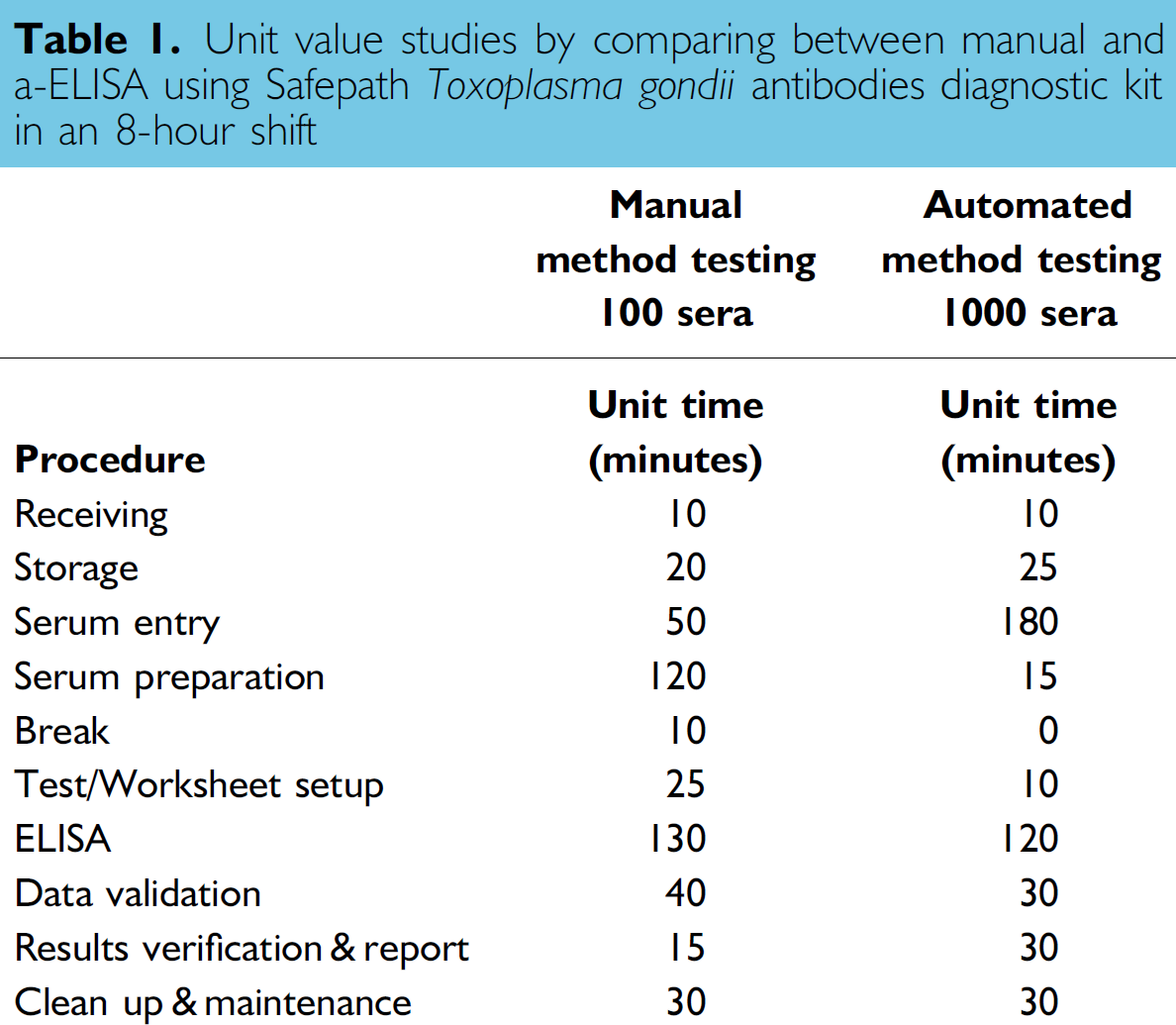
Unit time evaluation was carried out by comparing the T. gondii ELISA performed manually or with automation (Table 1). The different steps involved were: receiving, storage, and tracking of specimen, serum preparation, test/worksheet setup, assay, data validation, results verification, and reporting. Each unit value equals 1 min. The timeframe considered was an eight-hour shift period. Basically, it involved estimating the number of T. gondii antibodies ELISA, manually or automated, we could process in an eight-hour shift period by one analyst. An eight-hour shift was the time period we adopted to estimate throughput in our laboratory.
Unit value studies by comparing between manual and a-ELISA using Safepath Toxoplasma gondii antibodies diagnostic kit in an 8-hour shift
Data Validation
In our laboratory, we are mandated to validate this a-ELISA according to the requirements of the ISO17025 standard. 15 We have used methods described previously.16,17 We have employed a test-specific validation program based on a database of past test results of the reference positive and negative control sera. The criteria examined included reproducibility, repeatability, analyst testing proficiency, cutoff value determination, and the O.D. values consistency of the positive and negative serum controls.
A two-by-two table for reproducibility studies of positive and negative serum controls
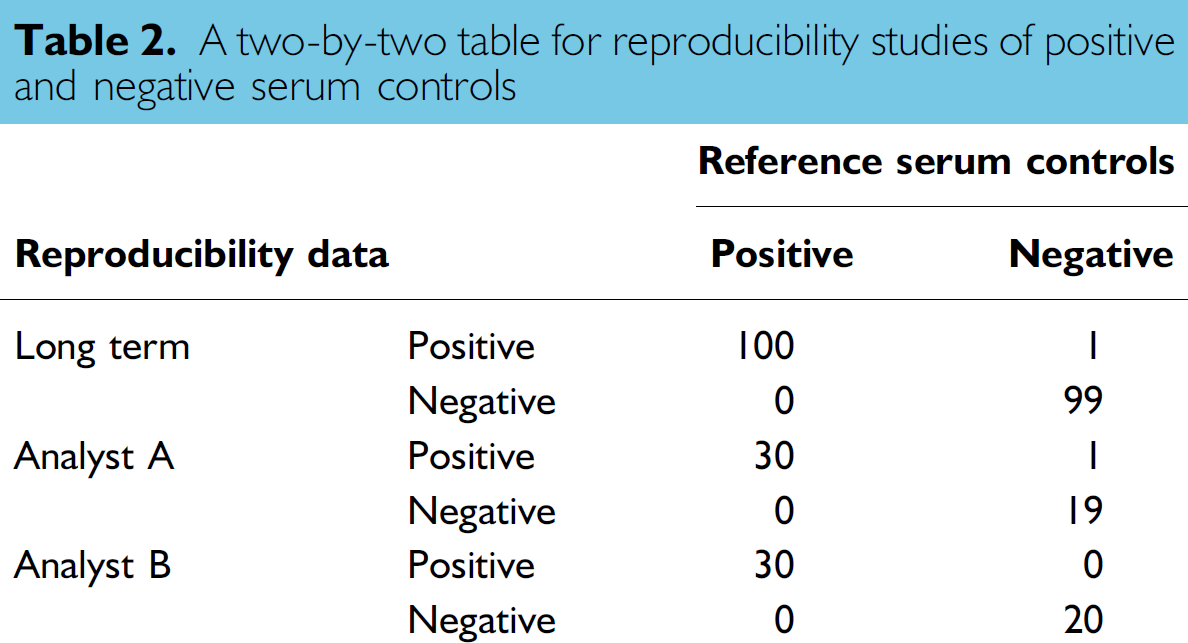
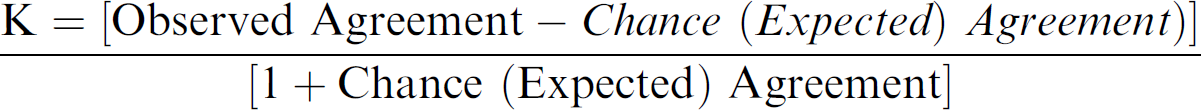
The analysts were scored from zero (0%) to 1 (100%). The passing grade is 0.95 (95%). The closer to unity, the more reproducible and accurate is the test result.

Repeatability graphs for positive and negative serum controls (intraplate). (The x-axis includes 30 test dates, whereas the y-axis represents the O.D. of the control sera.)

Repeatability graphs for positive and negative serum controls (interplate). (The x-axis includes 30 test dates, whereas the y-axis represents the O.D. of the control sera.)
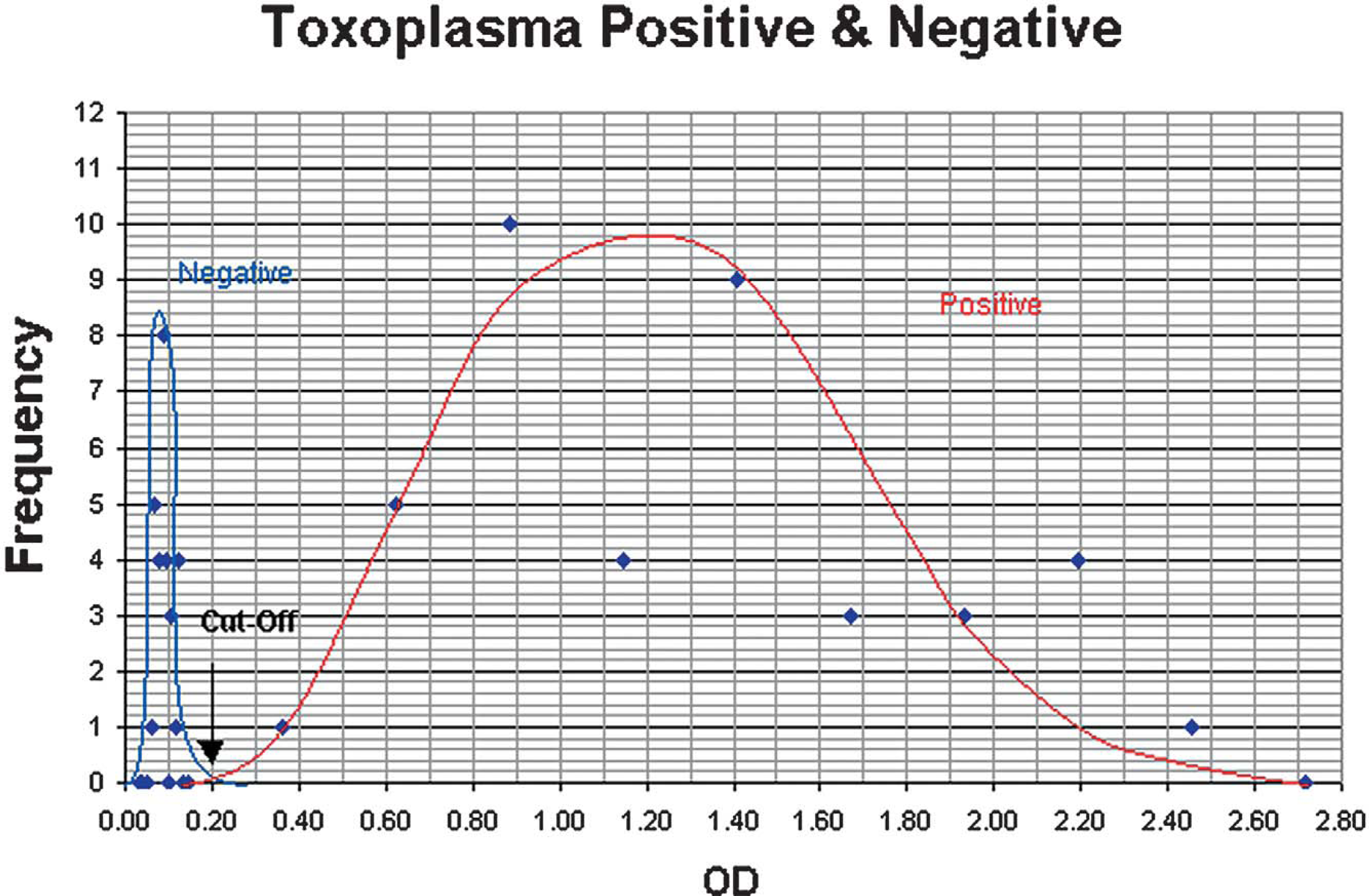
Frequency distribution curve for positive and negative serum controls.
On the basis of the combined check results of the above-described parameters, an overall judgement can be made in real time to indicate, for instance, the necessity of a retest, even at times when the duty analyst is not a T. gondii antibodies ELISA specialist. After validation, the results of all the test sera are calculated and scored as either positive or negative using the cut-off values.
Results
Throughput
In our unit value study of the T. gondii ELISA, we examined the maximum throughput of one analyst in an eight-hour shift using both the manual and a-ELISA (Table 1). The major duties involved: receiving, storage, preparation of specimen, setting up and running the assay, data collection, validation, submission verification, and result reporting. Under optimal conditions (on average), one analyst can manually process 100 assays for T. gondii antibodies in one eight-hour shift. Using the T. gondii a-ELISA, around 1000 assays can be completed each shift. The turnaround time (TAT) improvement and the decrease of labour cost for the test is 10-fold.
Reproducibility
For the long-term reproducibility studies (Table 2), the K value was 0.99 (99%). The test/analytical sensitivity and specificity were 100% and 99%, respectively. For the analyst proficiency tests (Table 2), for technologist A and B, respectively, the K values were equal to 0.98 (98%) or 1.00 (100%), respectively. The passing grade is 95%. The analyst proficiency test for each analyst is required annually.
Repeatability
For the repeatability studies, O.D. values of the positive and negative control sera of the a-ELISA were examined. In the intraplate study (Figure 6a), for the positive serum control, the mean was 1.20, the SD was 0.05, and the CV was 4.2%. For the negative serum control, the mean was 0.050, the SD was 0.0005, and the CV was 1.0%. In the interplate study (Figure 6b), for the positive control serum, the mean was 1.187, the SD was 0.182, and the CV was 15.3%. For the negative control serum, the mean was 0.049, the SD was 0.005, and the CV was 9.4%. Examining Figure 6a, intraplate analysis, the results of the 30 individual runs of either control were all within ± 2SD O.D. values. The same observation was recorded in the interplate analysis (Figure 6b). These charts showed that the a-ELISA developed was a precise test procedure since the results were repeatable in both intraplate and interplate analyses.
Threshold Values
In the frequency distribution graph of the negative and positive controls, the two bell-shaped curves intercepted each other at the threshold or cut-off values. The O.D. values of the cut-off were 0.20 units (Figure 7). This result matched the recommended validation criteria of the manufacturer for this ELISA (Figure 8). Examining the frequency distribution graph on the two sides of the cut-off line, we may have regions of false negative (left-hand side) and false positive (right-hand side). Moving the cut-off line to either side would affect the diagnostic sensitivity and specificity of the a-ELISA. Increasing the diagnostic sensitivity will result in a decrease of the diagnostic specificity and vice versa.
Manufacturer validation criteria (positive and negative cut-off values).
Discussion
In developing this a-ELISA, we considered several important requirements. First, we should be able to configure the automation workstation and its peripherals with great flexibility to accommodate the multistep, multiequipment requirements of the immunoassay procedures. At the same time, the degree of complexity of the working of the assay should be kept to a minimum so that it is easy to write up the operational program, preferably one single program per assay. Second, the robotic workstation should be accurate, precise, simple to operate, robust, and easily maintained to avoid forced downtime due to human and or machine failure. Third, the assay should be able to perform high-throughput screening for the antibodies to T. gondii. Fourth, the operation should allow (in between assay steps) ample walkaway time (WAT) for the technologist to multitask, such as sample receiving, sample preparation, test validation, result interpretation, and information reporting. Of equal importance, we must examine the cost effectiveness of the automated assay. At the end, developing a standard operation procedure (SOP) is necessary. This SOP should describe the steps and critical control points of the methodology, the validation criteria, the performance characteristics, and the data management program of the a-ELISA. These will quality assure that the assay is accurate, precise, and consistent.
We achieve a high degree of automation flexibility by integrating the automation workstation with its peripherals by using the software purchased from the manufacturer. This open design, reinforced by a modular approach for setting up the hardware during the planning and scheduling of the test, provides great flexibility. The different ALPs suitable for a variety of functions allow easy adjustment for the different liquid handling steps. All necessary liquid handling steps, including liquid transfer, microplate shaking, incubation, washing, and reading steps, can be automated. The degree of operation complexity can be kept to a minimum for this a-ELISA by writing the specific operation program with the software provided. Each time the end user is required only to use the “run” command to start the program. We wrote one specific program for the T. gondii a-ELISA for controlling the different essential steps of the test.
In order to prevent forced downtime due to machine failure, the workstation has been maintained in good working condition by the manufacturer and our routine maintenance measures. The workstation is maintained and certified for accuracy by the manufacturer annually. The technologist performs routine workstation alignment procedures on a weekly basis. Forced downtime due to human fatigue between steps of the assay has also been eliminated.
Improving the throughput of this ELISA for screening specific antibodies was the main purpose of this project. One technologist applying the described a-ELISA can run more than 10 times the number of specimens that can be tested in the same timeframe by a manual ELISA. There are several significant time savers in our a-ELISA compared to the ELISA run manually. There is an improvement by using a 96 micro-tube system together with individually labelled serum tubes in sample collection and preparation/dilution, as compared to manual labelling and sorting. Instead of transferring the specimens individually to a dilution tube and finally to the 96-well ELISA microplate, the robotic workstation can transfer the diluted samples to the microplate in one single step. And throughout the a-ELISA, the robotic workstation uses the 96-channel pipette head in all liquid handling steps, as compared to individual and eight wells transferred at a time found in the manual method. The use of a bar code recognition system, 96 micro-tube system, 96-channel pipetting and electronic handling of data in the a-ELISA improve the speed and reliability of the assay. The final result is a significant decrease in the cost-per-test due to increased test capacity and decreased turnaround time (TAT).
By the nature of any automated procedure, one built-in advantage is enabling the analysts to increase walkaway time (WAT) during each screen. The time can be used to perform specimen receiving, preparation, storage and discard, automation analysis, data generation, test validation, and results reporting. These are functions that have to be coordinated in order to produce valued information (science-based information about the food-borne pathogen, T. gondii) in real time for our client. We have attempted to electronically link all these functions together using different computer hardware and software programs and a government local area network.
Since our provincial facility is an accredited laboratory, it is a requirement that the a-ELISA be validated and all quality assurance tests maintained. The procedures include repeatability, reproducibility studies, and determination of threshold or cut-off values. Repeatability or precision studies require a minimum dispersion between tests, and for reproducibility or accuracy studies there should be a minimum shift from the expected value in each assay. The repeatability studies of the positive and negative controls provided excellent results. The ISO 17025 standard also requires a system for regular analyst training using on-site proficiency panel testing to ensure that the laboratory consistently produces reliable test results. The development of a standard operating procedure for the assay completes the test validation requirements. The robotic workstation and the peripheral equipment used are certified by the manufacturers annually.
In Canada, Alberta swine producers raise more than 3 millions pigs annually. When there is a need to carry out T. gondii surveillance studies to screen large numbers of animals, an automated HTS assay such as an a-ELISA becomes necessary. This provides a rapid, highly flexible assay method that is capable of producing rapid reporting of surveillance information. Because T. gondii is one of the most important food-borne pathogens to consumers, it is important to stay vigilant to the possibility of exposure to this parasite at some point along the “gate to plate” pathway. Early detection and the ability to break the chain of infection will be economically beneficial to the swine industry and ultimately become important as a public health and food safety measure.
Acknowledgments
This work was supported totally by the Agri-Food Laboratories Branch, Food Safety Division, Alberta Agriculture Food and Rural Development, Alberta, Canada. The authors would like to thank Murray Kennedy and Jane Calvert for reading the manuscript and providing valuable suggestions. Mention of trade names or commercial products in this article is solely for the purpose of providing specific information and does not imply recommendation or endorsement by the Alberta Department of Agriculture Food and Rural Development.
