Abstract
Purpose
To evaluate functional electrical stimulation (FES) neuroprothesis as a method to improve gait in hemiplegic patients, using three different gait scoring methods as measures.
Methods
Five hemiplegic patients (four with cerebral palsy at GMFCS I, one with diffuse pontine glioma) with a mean age of 16.5 years were given a FES neuroprosthesis (NESS® L300™) that was applied and calibrated individually. After an adaptation period during which the participants increased their daily use of the neuroprosthesis, gait was assessed with the stimulation off and with the FES on. Kinematic, kinetic, and temporal spatial data were determined using motion analysis and summarized by three scoring methods: Gait Profile Score (GPS), Gait Deviation Index (GDI), and Gillette Gait Index (GGI). Indices were calculated using the Gaitabase program available online. Patients were followed for a minimum of 1 year.
Results
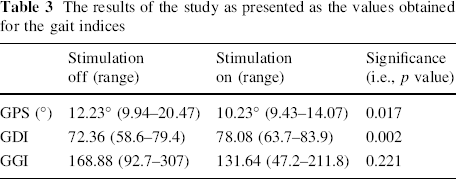
When comparing gait with and without stimulation, all scoring methods showed improvement. GPS and GDI of the affected leg were significantly improved: 12.23–10.23° (p = 0.017) and 72.36–78.08 (p = 0.002), respectively. By applying the movement analysis profile, the decomposed GPS score, we found that only the ankle dorsiflexion and the foot progression angle were significantly changed. GGI of the affected leg showed improvement, but without statistical significance: 168.88–131.64 (p = 0.221). Total GPS of legs and the GPS, GDI, and GGI of the nonaffected leg showed improvement without statistical significance. At the 1-year follow-up, all patients expressed high satisfaction and continued to use the device.
Conclusions
Dorsiflexion functional electrical stimulation improves gait in hemiplegic patients, as reflected by GPS, GDI, and GGI.
Keywords
Introduction
Foot drop in patients with cerebral palsy (CP) hemiplegia was defined by Rodda [1] as a type 1 hemiplegia gait pattern, characterized by ankle plantarflexion during the swing phase due to an inability to selectively control the ankle without calf contracture, so that ankle dorsiflexion is relatively normal during the stance phase. This displays the greatest improvements in gait and functional mobility when using an ankle foot orthosis (AFOs) [2, 3]. Patients with type 2 hemiplegia may also benefit from other solutions, such as botulinum toxin type A injections, serial casting, or a combination of both, and may show significant improvement in parameters such as the range of passive dorsiflexion, selective motor control, and their observational gait score [1, 4].
The use of the functional electrical stimulation (FES) neuroprosthesis device was found to improve dorsiflexion in swing and initial contact as well as gait velocity, and to help with social integration [5–8]. Teenaged patients, especially ones with mild involvement, usually find braces or casts cumbersome and unappealing to their body image, and may seek a more discrete device.
Reports on the use of instrumentation that apply functional electrical stimulation to adolescents and young adults with spastic hemiplegia have shown favorable results, but the immediate effect of surface FES on gait kinematic, kinetic, and spatial–temporal parameters has not been well investigated [9].
The purpose of this study was to evaluate the efficacy of surface FES in patients with hemiplegic CP using three different gait indices as outcome measures: the Gillette Gait Index (GGI), the modified Gait Deviation Index (GDI), and the recently introduced Gait Profile Score (GPS).
Patients and methods
For our study, we used the NESS® L300™ neuroprosthesis (NESS Ltd, Ra'anana, Israel). This system delivers electrical stimulations to the common peroneal nerve during the swing phase of gait, causing the ankle to dorsiflex and thus preventing foot drop. As described in detail by Hausdorff and Ring [5], this system (Fig. 1) has three main components that communicate via radio frequency signals: (A) a remote unit giving the patient control over the system; (B) an integrated stimulation unit with electrodes placed around the upper leg, over the common peroneal nerve and the origin of the tibialis anterior muscle (modifying the positions of the electrodes during the fitting process may promote dorsiflexion with slight eversion); (C) a force-sensitive gait sensor placed under the foot to detect heel strike and toe off. Computed algorithms analyze the gait sensor's data and the information is transmitted to the stimulation unit to induce dorsiflexion at the appropriate time and for the exact duration. Necessary adjustments may be done by a clinician using a handheld computer (PDA) to set the stimulation intensity, pulse frequency, and gait parameters of the system (e.g., “extended” time—the time that the stimulation is continued after heel contact).
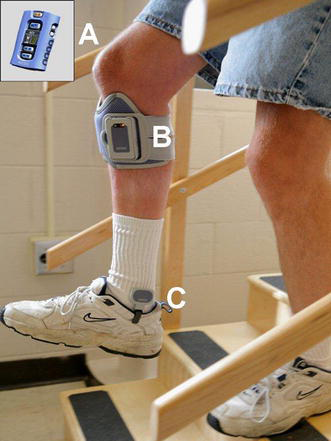
A Remote unit giving the patient control over the system. B An integrated stimulation unit. C A force-sensitive gait sensor
In this study, the FES neuroprosthesis was applied and calibrated individually for each patient and was then given for an adaptation period during which the daily use of the system was gradually increased. On the examination day, all patients underwent Gross Motor Function Classification Scale (GMFCS) [9] assessment (see Table 2), as well as a thorough physical assessment consisting of manual muscle testing, joint range of motion, and joint and muscle stiffness testing. A 3D gait analysis was performed using an 8-camera system (Vicon MX Giganet motion analysis system, Oxford Metrics, Oxford, UK). Retro-reflective markers were applied onto anatomical landmarks to capture gait performance. Kinetic data were collected using four AMTI force plates (Advanced Mechanical Technology Inc., Watertown, MA, USA). Gait was then assessed in two conditions, one with the neuroprosthesis worn but with the stimulation turned off, and one with the dorsiflexion stimulation turned on. The patients were then asked to walk along a 12-m walkway at a self-selected velocity.
The data collected from the representative trials were fed into an internet-based, anonymous data sharing program called Gaitabase, which calculated the three gait indices for each gait analysis examination. The indices were (see Table 1) as follows. (1) The Gillette Gait Index (GGI), based on 16 discrete variables calculated for each gait cycle as described by Schutte [10] in 1999 (the variables calculated for each index are summarized in Table 1). (2) The Gait Deviation Index (GDI), a methodology described by Schwartz in 2008 [11] that incorporates nine features, including three-dimensional rotation angles for the pelvis and the hip, the sagittal plane at the knee, and the sagittal plane at the ankle, as well as the foot progression angle. The interpretation of the GDI is based on the scaling, which has an average score of 100 for people without gait pathology and reduces by 10 units for every standard deviation away from this. (3) The Gait Profile Score (GPS), which is a single index measure introduced by Backer in 2009 [12] that summarizes the overall deviation of kinematic gait data from normative data. This single index can be decomposed to provide gait variable scores to the nine key component kinematic gait variables that make up the GPS score. These are presented by the movement analysis profile (MAP) which, on a graph, has ten columns: nine represent each gait variable, and the tenth is the GPS score. The GPS as well as the MAP are represented by the same units (°) as the kinematic variables, but as this score represents the distance from the normal average, the smaller values indicate less deviation from the norm or better results; for example, if limited dorsiflexion is increased by an intervention, the GPS score will represent this improvement by being lower than the initial score.
Parameters included in the different gait indices

GGI Gillette Gait Index, GDI Gait Deviation Index, GPS Gait Profile Score
Inclusion criteria were patients with spastic hemiplegic type gait 1 or 2, GMFCS levels of I or II, weak or spastic dorsiflexors that maintained an adequate passive dorsiflexion range of motion to above 0°, and sufficient cognitive and communication skills to follow the commands given during the gait analysis and comply with the FES apparatus and its electrical stimulation. Excluded were those who had had an operation on the lower extremities 1 year previously, or botulinum injections 6 months prior to the study, as well as those having active uncontrolled seizures.
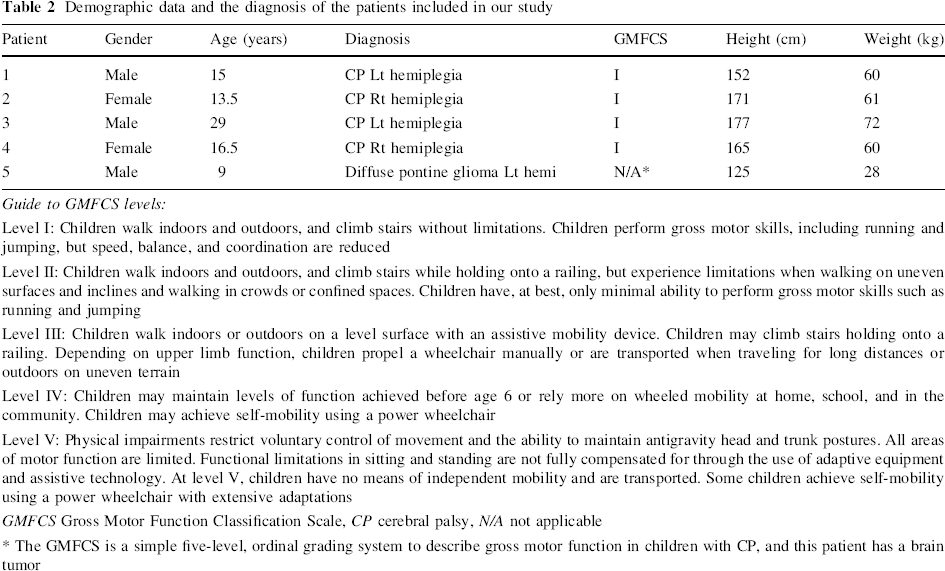
Five hemiplegic patients (four with cerebral palsy and one with diffuse pontine glioma) were recruited for this study. There were three males and two females. The mean age was 16.5 years; demographic data are shown in Table 2.
Demographic data and the diagnosis of the patients included in our study
Guide to GMFCS levels:
Level I: Children walk indoors and outdoors, and climb stairs without limitations. Children perform gross motor skills, including running and jumping, but speed, balance, and coordination are reduced
Level II: Children walk indoors and outdoors, and climb stairs while holding onto a railing, but experience limitations when walking on uneven surfaces and inclines and walking in crowds or confined spaces. Children have, at best, only minimal ability to perform gross motor skills such as running and jumping
Level III: Children walk indoors or outdoors on a level surface with an assistive mobility device. Children may climb stairs holding onto a railing. Depending on upper limb function, children propel a wheelchair manually or are transported when traveling for long distances or outdoors on uneven terrain
Level IV: Children may maintain levels of function achieved before age 6 or rely more on wheeled mobility at home, school, and in the community. Children may achieve self-mobility using a power wheelchair
Level V: Physical impairments restrict voluntary control of movement and the ability to maintain antigravity head and trunk postures. All areas of motor function are limited. Functional limitations in sitting and standing are not fully compensated for through the use of adaptive equipment and assistive technology. At level V, children have no means of independent mobility and are transported. Some children achieve self-mobility using a power wheelchair with extensive adaptations
GMFCS Gross Motor Function Classification Scale, CP cerebral palsy, N/A not applicable
The GMFCS is a simple five-level, ordinal grading system to describe gross motor function in children with CP, and this patient has a brain tumor
Patients were revaluated at a 1-year follow-up visit at the outpatient clinic. SPSS 11.5 for Windows (SPSS Inc., Chicago, IL, USA) was used for the statistical analyses, employing the paired t test.
The study was approved by the hospital's Institutional Review Board, and informed consent was obtained from all of the children's guardians.
Results
When comparing the gait with and without dorsiflexion stimulation, all scoring methods showed improvement (Table 3). GPS and GDI of the affected leg were significantly improved: 12.23–10.23° (p = 0.017) and 72.36–78.08 (p = 0.002), respectively. GGI of the affected leg showed improvement, albeit without statistical significance: 168.88–131.64 (p = 0.221). When the GPS score was decomposed into the nine gait variables, which are presented as the movement analysis profile (MAP), we found that only the ankle dorsiflexion and foot progression angle were significantly changed. Dorsiflexion was 13.6° without stimulation and 9.2° with the FES on (p = 0.042), while the foot progression angle changed from 17.6° to 11.8° (p = 0.043) with stimulation (Fig. 2, 3; Table 4). Knee flexion was not improved significantly (Table 5).
The results of the study as presented as the values obtained for the gait indices
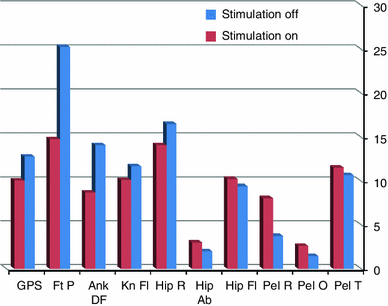
Movement analysis profile (MAP) of patient no. 4 and the total GPS score
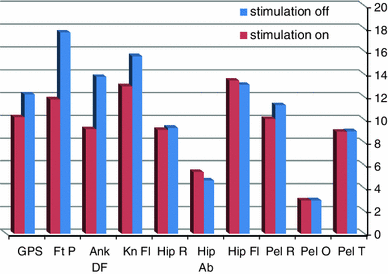
Average movement analysis profile (MAP) and the total GPS score. GPS Gait Profile Score, Ft P foot progression angle, Ank DF ankle dorsiflexion, Kn Fl knee flexion, Hip R hip rotation, Hip Ab hip abduction, Hip Fl hip flexion, Pel R pelvic rotation, Pel O pelvic obliquity, Pel T pelvic tilt (smaller values indicate less deviation from the norm)
Dorsiflexion during the gait cycle for each patient
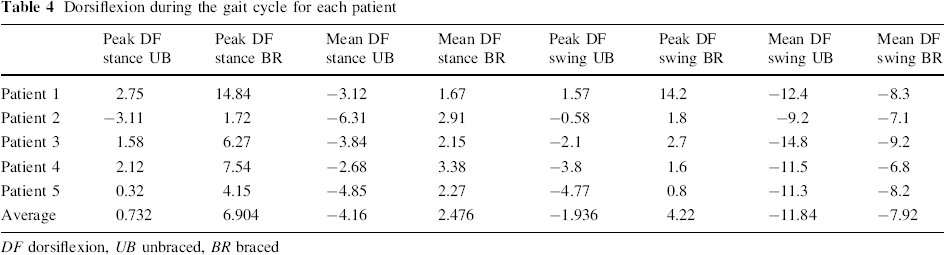
DF dorsiflexion, UB unbraced, BR braced
Knee flexion during the gait cycle for each patient
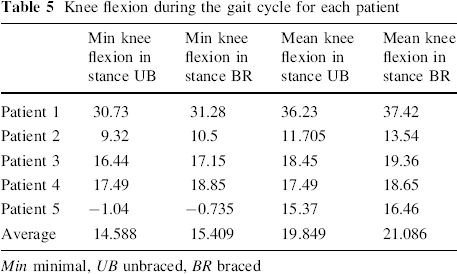
Min minimal, UB unbraced, BR braced
Total GPS (which includes both legs), GPS, GDI, and GGI of the less affected leg showed improvement, albeit without statistical significance.
Temporal parameters were all improved as well (Table 6).
Temporal parameters
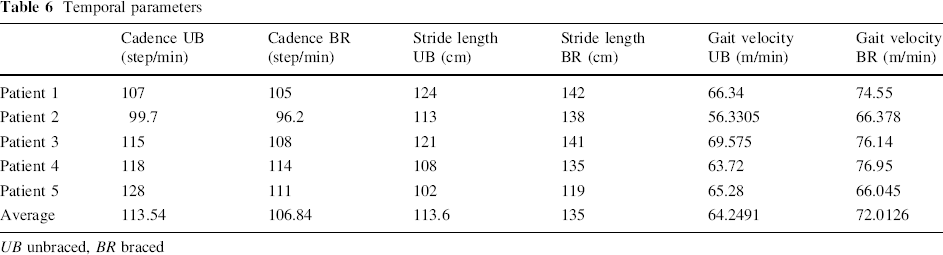
UB unbraced, BR braced
At the 1-year follow-up, all patients expressed high satisfaction and continued using the device.
Discussion
It has been our observation that adolescent patients and young adults with CP, especially the ones with mild involvement, tend not to use AFO type braces for reasons concerning body image, comfort, and social integration. One of the potential benefits of the FES neuroprosthesis device is its small proportions; its ease of use and high-tech image might also be more attractive to this population. The prospect of adding a new device to facilitate the gait of CP children led us to consider the FES neuroprosthesis and perform a preliminary investigation of its effect. Our study is distinguished by the use of gait indices as outcome measures to evaluate the effect of the device.
During the last decade, a few indices have been derived to calculate the amount that a subject's gait deviates from an average typical gait and present the deviation as a single number [10–12]: the Gillette Gait Index (GGI), the Gait Deviation Index (GDI), and the Gait Profile Score (GPS). These indices represent the overall gait pathology. In this study, we used all three indices in order to maximize the validity of the results.
Our results showing that functional electrical stimulation (FES) improves gait in hemiplegic patients correlate well with the results of previous studies. Laufer [7] found that FES significantly increased physical functioning and participation in community life as well as gait velocity in 16 adults with hemiplegia. Orlin [13] described the positive results of using percutaneously introduced indwelling electrode FES on 8 children with CP, 2 of whom had hemiplegia.
In our preliminary study, we have found that all patients were using this device at the 1-year follow-up and were satified with it—none had stopped using it.
However, even given our encouraging results, it is worth noting that the FES device is expensive and may not be the first-line choice for each patient. Age, level of comprehension, and compliance must be considered. It should be prescribed after a thorough individual investigation of its applicability. A reasonable time should be given for adaptation, which should be followed by an efficacy test, preferably based on gait analysis. Some patients have complained of initial discomfort arising from the electrical stimulation; that usually resolves after a short time. There are other patients that may not fully benefit from this device due to other reasons, such as short plantar flexor muscles or prolonged discomfort.
Our results are based on a statistically significant improvement in the gait of hemiplegic patients as reflected by the GPS and the GDI. Both of these indices, which are based on similar kinematic gait parameters, are closely related measures [12]. We have also shown that, as expected, the main components affected by the FES are the dorsiflexion of the ankle and the improvement of the foot progression angle. This decomposition of the GPS score into the nine movement analysis profile (MAP) scores is a helpful tool for evaluating gait studies, as it emphasizes the main components that are responsible for the overall change.
Although we studied a small number of patients, the GDI, GPS, and MAP were found to be sensitive to the changes in gait created by the use of FES, even to the degree of the component most affected.
The GGI did not detect significant improvement, although the trend was positive. This index, which was the first to be published [10], has been found to have some shortcomings, including the arbitrary and unbalanced nature of the 16 parameters that comprise the index as well as difficulties in its implementation [11]. For example, in a study of eight type 1 hemiplegic patients, the score ranged from 28.5 to 311.6 with an average of 98.8; this variability probably interferes with the attainment of meaningful results in small-population studies. Orlin [13] found that electrical stimulation did not alter ankle kinetics and spatiotemporal parameters in children with CP. This may also explain the results of the GGI, which is the only index to account for spatiotemporal parameters. This was a preliminary study, so the patient population was small.
Controlled investigations are warranted to determine the efficacy of functional electrical stimulation and to determine how to achieve a long-lasting benefit using this new modality in children with cerebral palsy. Studies should also aim at determining which CP patients are most likely to benefit from neuroprosthesis, possibly including diplegic patients too.
Footnotes
None.
