Abstract
Abstract
Purpose
Pamidronate, used for the treatment of paediatric osteoporosis, reduces the fracture rate and improves ambulatory status. Intravenous pamidronate therapy has known complications which have not been stratified based on its dose and distribution. This study aims to assess the early minor and major medical and late surgical complications and the effect of the dose and regimen of infusion on these events in paediatric osteoporosis.
Study design
Retrospective cohort.
Materials and methods
Three regimens for pamidronate infusion were followed in sequential periods in 10 years. Regimen A delivered 1.5 mg/kg/day as a single dose once in 3 months. Regimen B delivered 2 mg/kg/day for 3 days twice a year, while regimen C delivered 1 mg/kg/day for 3 days every 3–4 months. Adverse events were classified as early (major and minor) or late (surgical).
Results
Forty-eight children received 158 infusions using one of the three regimens. Twenty-nine complications occurred in 24 children. A significant difference in the complication rate was present among the three regimens (P = 0.005). Nineteen children had minor complications, mainly febrile reaction or asymptomatic hypocalcaemia. Four major complications consisting of one seizure, one respiratory distress and two hypocalcaemic tetany were encountered, all with regimen B. Intraoperative complication faced was loss of position due to splintering of the cortex while rush rodding. This was seen in 20% of the long bone segments operated in those who received pamidronate as compared to 4.4% of the segments which were operated prior to the initiation of pamidronate therapy; the odds of splintering were 5.4 times higher for those patients who were bone segment rodded after pamidronate therapy.
Discussion
Intravenous pamidronate is associated with complications in 50% of children with paediatric osteoporosis, with a dose-dependent significant difference. Major complications are not uncommon with higher doses and can be avoided by increasing the number of doses per year and decreasing the dose per cycle. Surgical difficulty, when possible, can be avoided by correcting any major deformities at presentation prior to the induction of pamidronate therapy.
Introduction
Pamidronate, a nitrogen-containing bisphosphonate, is being increasingly used in paediatric osteoporotic disorders following an initial case report for juvenile osteoporosis by Hoekman et al. and a series of 30 children with osteogenesis imperfecta published by Glorieux et al. [1, 2]. Major indications for pamidronate therapy are primary osteoporotic disorders, such as osteogenesis imperfecta and fibrous dysplasia, and some causes of secondary osteoporosis, like cerebral palsy and meningomyelocele [3]. Similar to other bisphosphonates, it acts by preventing osteoclastic resorption [1, 4]. In symptomatic patients, pamidronate decreases bone pain and the number of fractures per year, improves ambulatory status and increases the growth rate. In addition, an increase in bone mineral density and vertebral height has been recorded [2].
Dosages have varied widely from 0.25 to 3 mg/kg/cycle and yearly dosages have varied from 0.5 to 12 mg/kg/year [5–7]. Even with these schedules, there is further variation in the way each dose cycle is further administered over a period of days [7, 8]. Complications of bisphosphonate therapy in children have been sporadically reported, but their association with the dosage and administration protocol has not been evaluated in the literature [8–15]. Minor complications include febrile reactions, asymptomatic hypocalcaemia, body ache and metallic taste in the mouth. Major complications that have been reported in children with osteoporosis are symptomatic hypocalcaemia, seizures, respiratory distress and delayed healing of bone after fractures and surgical procedures [9–13, 16]. Pamidronate can be traced in urine samples for up to 8 years and some potential complications could be refractory [17].
In our unit, over the last 10 years, three different regimens of pamidronate administration have been used, mainly for the treatment of osteogenesis imperfecta. A number of minor and major complications were seen in patients who underwent treatment. This study focuses on the medical and surgical adverse outcomes of pamidronate therapy and their relation, if any, to the dose and schedule of administration.
The objectives of this study were:
To assess the total adverse event rate in children who receive pamidronate therapy for osteogenesis imperfecta or fibrous dysplasia. To assess the minor and major, medical and surgical, early and late adverse event rates. To assess the effect of infusion dose and regimen of administration on these events.
Materials and methods
A retrospective cohort study was carried out amongst all children up to 18 years of age who were seen by the paediatric orthopaedic unit of our institution between January 2001 and April 2011, and who received pamidronate therapy for osteogenesis imperfecta or fibrous dysplasia. Data collection was done from computerised inpatient and outpatient records.
Prior to therapy, all patients underwent counselling regarding admission, medical management, need for repeated cycles of therapy and associated medical risks. Pre-infusion serum calcium and phosphorus levels were checked for all patients and vitamin D levels from 2010 onwards. Post-infusion serum calcium was measured. All children received oral calcium and vitamin D supplements prior to therapy if deficient.
There were three different dosage schedules for pamidronate. Regimen A (January 2001 to December 2006) delivered 1.5 mg/kg/day as a single dose in 100 ml of saline over 8 h repeated once in 3 months [2]. Fifty infusions of this regimen were received by 13 children. Regimen B (January 2007 to July 2010) delivered 2 mg/kg/day in 250–500 ml of normal saline administered over 8 h for three consecutive days and repeated at 6-month intervals, providing a total dose of 12 mg/kg/year [5]. Eighty-three infusions of this regimen were received by 35 children. Regimen C (August 2010 to April 2011) delivered 1 mg/kg/day in 250–500 ml of normal saline administered over 8 h for three consecutive days and repeated at 3–4-monthly intervals [8]. Twenty-five infusions of this regimen were received by 17 children.
In some patients, there was an overlap of regimens; hence, adverse events were analysed in the short term based on the regimen followed at the time of complication.
Adverse events were defined as early or late, and early complications were sub-categorised as minor or major. Minor complications in the early stages were defined as those where infusion was continued after temporary suspension and included febrile reaction and asymptomatic hypocalcaemia, and major complications were those which caused a prolongation of hospital stay, stoppage of infusion, an intensive care unit admission or re-admission, such as symptomatic hypocalcaemia, seizures and respiratory distress. Late complications included non-union, delayed union, osteopetrosis and splintering of bone ends at the time of intramedullary rodding causing collapse.
The rate of intraoperative difficulty in the reaming and splintering of the cortex was compared in children who received pamidronate infusion before surgical intervention for deformity correction with the same in children who received pamidronate after surgical intervention in terms of the regimen followed, number of infusions and interval between pamidronate administration and surgery.
The data were analysed using a statistical online computational resource. The Chi-square test and Fisher's exact test were performed and odds ratios were calculated.
Results
Forty-eight children with a mean age of 7.7 years (range 1–17 years) received pamidronate therapy. There were 32 boys and 16 girls. Forty-seven children had osteogenesis imperfecta and one had McCune–Albright Syndrome with polyostotic fibrous dysplasia. The 47 children with osteogenesis imperfecta were classified following the Sillence classification, with 27 children being of type I and 10 each were type III and IV osteogenesis imperfecta [18].
There were 29 complications in 24 children, i.e. 50% of children had one or more complications. Fifteen children had minor complications, all during or in the immediate post-infusion period. Six children had one episode of fever, one child had fever twice, two children had asymptomatic hypocalcaemia and fever, one child had asymptomatic hypocalcaemia twice, and five children had one episode of asymptomatic hypocalcaemia. Febrile reactions were managed symptomatically with the temporary suspension of pamidronate infusion for a day. Asymptomatic hypocalcaemia was usually diagnosed after the infusion therapy was stopped. This was managed with calcium carbonate supplementation 500 mg daily.
Major complications of infusion therapy were tetany (two children), seizures (one child) and respiratory distress (one child). The early major complication rate was 2.53%.
Case 1
A 7-year-old boy with type IV osteogenesis imperfecta was started on his second cycle of regimen B pamidronate infusion. His pre-infusion serum calcium, phosphorus and vitamin D levels were 9.5 mg%, 4.5 mg% and 28.05 ng/ml, respectively. The first-day dose was uneventful. During his second day of infusion, the child developed fever of 101°F and a sudden onset of carpopedal spasm. The pamidronate infusion was stopped. His serum calcium was found to be 6.6 mg%. Following infusion of 10% calcium gluconate at 1ml/min, tetany disappeared in 5–6 min. Further pamidronate infusions were postponed. The patient recovered fully over 12 h with a serum calcium of 8.6 mg%.
Case 2
An 11-year-old boy with McCune–Albright Syndrome with polyostotic fibrous dysplasia, who had undergone corrective osteotomy 3 months before for proximal femur deformity, was started on regimen B infusion for the first time. His pre-infusion serum calcium, phosphorus and vitamin D values were 8.6 mg%, 3.6 mg% and 18.64 ng/ml, respectively. The first 2 days of infusion were uneventful. On the third day, the child developed sudden-onset carpopedal spasm associated with 102°F fever. Pamidronate infusion was immediately stopped. His immediate serum calcium was 7.3 mg%. Intravenous 10% calcium gluconate was infused at 1 ml/min. Over the next 12 h, the symptoms disappeared.
Case 3
A 6-year-old girl with type III osteogenesis imperfecta, non-ambulatory with multiple limb and spine deformities, was admitted for her second cycle of regimen B pamidronate infusion. Her pre-infusion serum calcium, phosphorus and vitamin D levels were 9.6 mg%, 4.6 mg% and 27.08 ng/ml, respectively. The child had no dyspnoea or chest infections or use of bronchodilators in the past. On examination, air entry was equal on both sides and there were no adventitious sounds. This child, on the second day of pamidronate infusion, developed fever (102°F) with acute dyspnoea and had bilateral reduced air entry on examination. Pamidronate infusion was immediately stopped and the child was moved to the intensive care unit. On investigation, her serum calcium was 8.2 mg%, phosphorus 4.5 mg%, sodium 136 mmol/l, potassium 4.0 mmol/l and creatinine was 0.5 mg%. Her total leucocytes count was 11,800/mm3 and blood culture was negative. Urine examination showed 2 WBCs/hpf. Peripheral smear for malarial parasite was negative and chest radiograph was normal. Intravenous 10% calcium gluconate infusion 1 ml/min was administered. The child maintained 100% saturation on O2 supplementation. All symptoms disappeared after 24 h and the child was moved from intensive care and further pamidronate infusions were postponed. Subsequent pamidronate infusions were given as regimen C uneventfully.
Case 4
A 1-year-old boy with type I osteogenesis imperfecta was started on regimen B pamidronate infusion. His pre-infusion serum calcium, phosphorus and vitamin D levels were 9.7 mg%, 4.6 mg% and 37.43 ng/ml, respectively. On the second day of infusion, the child had fever (101°F) and acute-onset generalised tonic–clonic seizures, which lasted for 5 min. Pamidronate infusion was immediately stopped. His immediate serum calcium was 7.1 mg %, sodium 138 mmol/l and potassium was 3.6 mmol/l. Intravenous 10% calcium gluconate was infused. Seizures did not recur and the child became completely asymptomatic after 24 h.
One of the major complications of surgery was splintering of the cortex of the bone ends during reaming and passing the rush rod (Fig. 1). This occurred in 5 out of 18 children who underwent surgery on 25 long bone segments. The operated segments included 14 femora, nine tibiae, one forearm and one humerus. These 18 children received mean 3.8 infusions of pamidronate, with a mean interval between infusion and surgical intervention of 10 months (standard deviation [SD] 7.8, range 0.5–24 months). Four segments each were operated after regimen A and A + B infusions. Sixteen segments were operated after regimen B and one segment was operated after regimen C infusion. All five patients with this complication had received regimen B infusion before surgery. The complications occurred in the femur (four patients) and forearm (one patient). The operative difficulty noted in these cases was in reaming the canal and negotiating the rush nail because of the increased stiffness and brittleness of the bone.
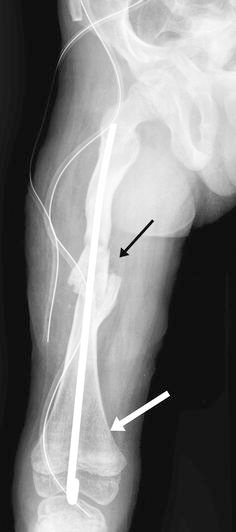
This post-operative radiograph shows splintering of the cortex of the femur shaft (black arrow) and resultant backing of the nail distally in a child with osteogenesis imperfecta who received prior pamidronate therapy. The white arrow indicates the metaphyseal sclerotic bands (zebra lines) showing the number of doses received
Five similar complications, all involving the femur, also occurred in the 17 children who did not receive pamidronate before surgery. They underwent surgery for 113 segments and, in all these children, the pamidronate administration was started after the surgery. The segments consisted of 60 femur, 43 tibia, six forearms and four humerus. The difference in the splintering rate was significant when the adverse rate per segment operated was compared using Fisher's exact test (P = 0.017). The odds ratio of developing an intraoperative complication was 5.4 times higher if the child had received pre-operative pamidronate therapy.
There was a single non-union of an osteotomy (0.7%) among children who underwent osteotomies for 138 segments. This was a child with non-union of the tibia who had received regimen A dosage.
Subgroup analysis of the three different regimens
Many children crossed over from regimen A to B and from B to C temporally. Medical complications were analysed as part of the specific regimen, as all medical complications were immediate.
Regimen A was given to 13 children who received 50 infusions, a mean of 3.4 per child.
In regimen B, there were 35 children and they received 83 infusions, a mean of 2.3 infusions.
Regimen C was delivered to 17 children. They received 25 infusions, a mean of 1.19 infusions.
Regimen A had one single minor event, a febrile reaction. In regimen B, there were 13 minor events (seven febrile reactions and six cases of asymptomatic hypocalcaemia). Regimen C patients had five minor reactions (two febrile reactions and three asymptomatic hypocalcaemia (Table 1). A statistically significant difference (P = 0.02) is present in the rate of early adverse events in the three regimens using the Chi-square test (Table 1).
Minor adverse events encountered under the three different regimens of pamidronate administration

All four major adverse events—two tetany, one seizure associated with severe hypocalcaemia and one respiratory distress—were seen with regimen B. All patients with these events recovered completely.
Type of osteogenesis imperfecta and complications
Twelve children with type I, six children with type III and five children with type IV osteogenesis imperfecta had complications following pamidronate infusion. No statistical significance was seen between the rate of complications and the type of osteogenesis imperfecta (P = 0.67).
Age and complications
The mean age of the children who developed complications was 7.1 years (SD 4.2). The mean age of the children who did not develop complications following infusion was 8.3 years (SD 4.4). There was no statistically significant difference in the mean age of children who developed complications and those who did not.
Discussion
There are many advantages of bisphosphonate therapy for paediatric osteoporosis, and it has become the mainstay of medical management in conditions like osteogenesis imperfecta [3, 8]. The regimens have varied based mainly on the ease of administration, convenience and age of the patient. Complications of bisphosphonate therapy are well reported in adults and children mainly for the treatment of malignancy and malignant hypercalcaemia, where high doses of up to 3 mg/kg/day are used [19–21]. These include fever, symptomatic hypocalcaemia, renal and ophthalmic toxicity [8, 20, 21].
In our study, three different regimens were used. The initial regimen A was based on the landmark publication by Glorieux et al. [2] reporting the results of cyclic pamidronate therapy with a mean pamidronate dose of 6.8 mg/kg/year given over 4–8 doses cyclically. In addition, single-dose infusions by Shaw et al. [5] were also considered to determine the dose. Based on this, a single dose of 1.5 mg/kg over a period of 8 h at 3-month intervals was selected. The clinical improvement was noted and, in 2006, after reviewing the literature, the dose was increased to 12/mg/kg/year in a 6-monthly cycle in a more intensive 2 mg/kg/day for 3 days. The reason for choosing the 6-monthly cycles was patient convenience. Pamidronate was not widely available in our country at that time, hence, many patients had to travel long distances. Reducing the frequency of drug delivery facilitated matters for patients.
A few major complications were noticed by 2010, causing us to review the complication rate and change to a lower dose per cycle of 1/mg/kg/day for 3–4 days and repeated every 3–4 months [8]. While minor complications were noted with the third regimen, no further major complications were seen.
This study was undertaken to highlight complications attributable to pamidronate therapy in our group of 48 children. A systematic review reports acute phase reaction (febrile), bone pain, non-specific gastrointestinal discomfort and asymptomatic hypocalcaemia as the frequent complications of such therapy for osteoporosis [8]. However, there is no major study which has focused on the complications of pamidronate therapy in children. In paediatric osteoporosis, the reported complications of pamidronate therapy have usually been minor. No adverse effects have been reported by a few series on pamidronate use for osteogenesis imperfecta in children [22, 23]. Many others have reported minor adverse effects without specifying the event rate [24–26]. Some have reported a fairly high frequency of minor adverse event rates, varying from 60 to 100% [2, 27–29]. In our study, 50% of children had minor complications, mainly febrile reactions and hypocalcaemia.
The most common immediate minor complication that has been reported with pamidronate is a febrile reaction that usually responds to paracetamol, as was seen in our patients [20]. While febrile reactions were noted in all three regimens, they were significantly higher in regimens B and C, suggesting that they may be dose-dependent within that cycle. We are not aware of any study in children where the relationship of febrile reactions has been found to be significantly dose-dependent in children with osteogenesis imperfecta.
The other immediate minor complication was hypocalcaemia. Hypocalcaemia is caused by the inhibition of osteoclastic activity that reduces the availability of calcium from this source [4, 20, 30, 31]. Minor asymptomatic hypocalcaemia in children on pamidronate therapy has been reported by many investigators [12, 32]. In our study, minor hypocalcaemia was significantly more common in regimens B and C. The monitoring of serum calcium at the end of each dose is recommended by almost all authors so as to avoid symptomatic hypocalcaemia. A risk factor for hypocalcaemia is hypovitaminosis D [20]. After we started assessing, we found many children with hypovitaminosis D probably due to limited sunlight exposure. It is important to highlight that hypovitaminosis needs to be corrected and parathyroid hormone levels restored to normal prior to starting pamidronate therapy, as is our practice now [20]. Hypocalcaemia is treated by replacement doses of oral calcium and vitamin D, and may need to be continued for several months [5, 20, 31].
The total minor adverse event rate was significantly lower in regimen A, which had a lower dose per cycle, as compared to B and C (P = 0.02). Most of the minor events were noticed on the second or third days. This suggests that, in those children prone to developing this side effect, lowering the dose per cycle and increasing the frequency may be a better tolerated strategy. Many regimens with low pamidronate dose have not reported any minor complications in their patients [22, 23].
Higher dosages have been used for patients with malignancy and there are many major side effects reported in these groups of patient, such as ophthalmic and renal complications, which have not been described in children with osteoporosis [8, 20, 33].
Though pamidronate in malignancies is well reviewed in the literature on adults and children, major reports on complications of pamidronate in paediatric osteoporosis are infrequent [20, 34]. Only three cases of symptomatic hypocalcaemia have been reported [12, 27, 35]. One report of respiratory distress in four infants and two reports of seizures in one child each have been published [6, 9, 11]. In this study, we had four patients with major medical complications, three with symptomatic hypocalcaemia and one with generalised seizures, giving an overall incidence of 2.5%. The three major hypocalcaemia-related complications (two cases of tetany and of seizures) occurred with regimen B. Hypocalcaemia in all these cases was managed with intravenous 10% calcium gluconate administered at the rate of 2 ml/min until symptoms ceased, followed by oral calcium supplement. Further pamidronate infusions in all these cases were postponed.
Munns et al. [11] reported on four children with respiratory distress following pamidronate therapy, mainly in children with pre-existing respiratory compromise. In our case, the symptoms were transient with no radiological changes and improved in 24 h, indicating that this may have been drug related. Subsequent treatment with regimen C, a lower dose per cycle, resulted in no recurrence.
We found that the medical complications analysed separately and together were significantly higher in regimen B, indicating that a dose of 2 mg/kg/day and 6 mg/kg/cycle led to an unacceptably high adverse event rate in these children. With regimen C, there were no major complications and the minor complication rate was not significantly different from that of regimen B (Table 2).
Total adverse events encountered under the three different regimens of pamidronate administration

Munns et al. [10] reported a delay in the healing of osteotomies in children receiving pamidronate therapy, with an odds ratio of 7.29 compared to controls not receiving pamidronate. They found fracture healing to be normal, but another study reported delayed fracture healing [16]. We had one case of non-union following tibial osteotomy—insufficient evidence of interference with osteotomy healing. The non-union rate of osteotomies depends on a number of factors, such as soft tissue handling technique, use of a power saw, method of fixation and external immobilisation. This may explain the difference in the non-union rates [36–38]. The effect of pamidronate on surgery has not been reported. We have noticed difficulty in passing rush rods through the canal and an increase in splintering of the cortex when reaming the canal. Splintering of the cortex was significantly more common in regimen B, where a single large dose is given once every 6 months. Remodelling is affected after treatment with pamidronate because of impaired osteoclastic action [7]. This has been shown for the metaphyseal regions and iliac crest biopsies [7]. While at diaphysis the histology may be different, remodelling occurs in the diaphysis following fractures in the region. We posit that, in children on pamidronate therapy, there may be more calcified cartilage rather than remodelled bone in areas of previous fractures and deformity. This and the increased stiffness of the bone may explain why splintering occurred significantly more often during rush rodding. If the child is older and requires rush rodding at the time of presentation, we now prefer to rod prior to starting pamidronate therapy. The rate of surgical complication was insignificant with regimen C, which administers 1 mg/kg/dose for 3 days, and regimen A, which administers 1.5 mg/kg/day for 1 day. The odds ratio of developing surgical complications is 5.4 with pamidronate infused prior to surgical intervention.
Limitations of this study are its retrospective nature and lack of objective scoring to assess surgical difficulty. No attempt was made to include efficacy measurement using instruments for assessment, such as dual-emission X-ray absorptiometry (DEXA) scan, fracture rate and quality of life questionnaires, as these are difficult to apply retrospectively. Even though our results are significant, a prospective study to evaluate efficacy along with adverse reactions will be of greater value.
Refractory secondary infection following osteonecrosis of the jaw is a serious complication recorded following bisphosphonate therapy in adults. However, we were unable to find any series with post-operative infections in children [8, 33]. On the contrary, pamidronate has been used for the treatment of chronic recurrent multifocal osteomyelitis with symptomatic relief [39]. There were no post-operative infections encountered following surgery or otherwise in our series.
While pamidronate therapy has been shown to have many advantages, there have been significant complications associated with this treatment in children. Some of the major side effects are dose-dependent, as highlighted in this paper, and we posit that increasing the number of doses per year and decreasing the dose per cycle can reduce major and minor complications. Hypocalcaemia appears to be related to a number of minor and major complications, and should be corrected by the supplementation of calcium and treatment of hypovitaminosis D before pamidronate therapy. Surgical difficulty, when possible, can be avoided by correcting any major deformities at presentation prior to the induction of pamidronate therapy.
