Abstract
Purpose
Studies on musculoskeletal manifestations (MSM) of childhood acute lymphoblastic leukemia (ALL) have yielded variable findings with regard to their clinical impact. We investigated the significance for differential diagnosis, treatment and outcome of musculoskeletal complaints as presenting symptoms of ALL, and their correlation with leukemia immunophenotypes, for which data is lacking.
Methods
Data on 783 children in the national study for childhood ALL between 1984 and 2003 were reviewed retrospectively. Statistical analysis examined possible relationships between MSM at the time of diagnosis and demographic and clinical data, biological features of leukemia (peripheral blood counts, immunophenotype and main cytogenetic aberration), response to initial prednisone treatment, and outcome.
Results
Of 765 children with data on orthopaedic complaints, 240 presented with MSM (31.4%). Among these children, B cell precursor (BCP) was much more common (209/576, 36.3%) than T cell ALL (25/176, 14.2%). Patients with MSM had lower white blood cell counts (WBC) (median of 9 vs. 20 × 109/L, P < 0.001) and percentage of blast cells in the peripheral blood at diagnosis compared to those without (median of 27 vs. 53%, P < 0.001). Hepatomegaly and splenomegaly were less common in MSM group (67 vs. 53% <3 cm, P < 0.001, and 63 vs. 50% <3 cm, P < 0.001, respectively). Poor response to initial treatment with prednisone was recorded in 7.1% of patients with MSM versus 11.5% of those without (P = 0.086). The analysis revealed no independent effect of MSM on event-free survival (EFS), after correcting for differences in EFS related to immunophenotype or initial WBC.
Conclusions
MSM occur mostly in children with BCP ALL who present with less involvement of extramedullary organs, low peripheral blood blasts and white blood cells counts. These findings highlight the importance of including ALL in the differential diagnosis of MSM even in the presence of an apparently normal peripheral blood count. Our study also suggests that MSM are caused by leukemic cells with enhanced biological propensity to remain relatively confined within the intramedullary bone-marrow space.
Keywords
Introduction
Musculoskeletal manifestations (MSM) associated with acute lymphoblastic leukemia (ALL) in children are common findings with a prevalence estimated between 11.6 and 50% [1–4]. The most common MSM are localized or diffuse bone pain, limp, arthritis, myalgia, failure to use an extremity, as well as other complaints mimicking orthopedic conditions such as osteomyelitis [5–7]. In addition, the affected individuals present an increased risk for fractures [2, 6, 8].
Of note that, during the course of the disease, between 70 and 90% of patients with ALL are reported to show abnormal skeletal findings on radiographs such as osteopenia, cortical lucencies, metaphyseal bands or combinations thereof [9–11]. Pathological changes in bone with subsequent skeletal morbidity are caused by the leukemic cells as well as by the chemotherapeutic agents and glucocorticoids used in leukemia treatment [2, 8, 12, 13].
The prognostic implications of the musculoskeletal expressions of malignancy in children with ALL have remained unclear. On one hand, some authors have suggested that patients with clinically severe MSM have a better prognosis [14, 15], whereas others found either a strong association between radiologically documented severe bone involvement and poor prognosis [16], or no relation at all [17]. On the other hand, the musculoskeletal symptoms can be so prominent or even the only presenting sign that sometimes the diagnosis of leukemia is delayed, and established only after subsequent detailed hematological workup [18, 19]. According to some reports, the presence of normal peripheral blood differential counts on routine laboratory tests, as well as the co-occurrence of commonly associated systemic symptoms such as fever, malaise, anemia, breathing problems or rash may contribute to a delay in diagnosis of the underlying malignancy [18, 20]. Based on nonspecific systemic and musculoskeletal complaints, the differential diagnosis is often complex and should take into account a wide range of pathologies such as juvenile rheumatoid arthritis, reactive arthritis, rheumatic fever, osteomyelitis or aplastic anemia [4, 18, 21, 22].
Considering the currently limited data on the distribution of MSM among the ALL subtypes, we studied whether there is an association between musculoskeletal symptoms at the time of diagnosis and specific leukemia phenotypes, and are attempted to investigate further the prognostic implications of MSM at presentation for pediatric patients with ALL in the context of modern intensive treatment.
Patients, methods and study design
From 1984 to 2003, 783 children were enrolled in the Israeli national studies (INS) for childhood ALL (INS 84 1984–1989, 134 pts; INS 89/98, 1989–2003; 649 pts). Informed consent was obtained from the parents or guardians for each patient prior to the child's inclusion in ALL studies. The treatment in INS 89/98 was based on the modified Berlin–Frankfurt–Munster 86/90/95 protocols for ALL [23]. An important change in treatment during this period was the substitution of extended triple intrathecal therapy for preventive cranial radiation to ensure safety of the central nervous system [24].
We conducted a thorough retrospective search into the records of all patients in the INS databases. For the subsequent analyses, we retrieved the data on musculoskeletal manifestations at the time of diagnosis, as well as the demographic and clinical data, biological features of leukemia (peripheral blood counts, immunophenotype, and main cytogenetic aberration), patients’ response to initial prednisone treatment, and outcome. Information on duration of symptoms was not included in the data analysis because it was incomplete.
Musculoskeletal manifestations were defined as subjective complaints of bone pain, arthralgia and back pain, clinical findings at physical examinations (arthritis, tendonitis, suspected osteomyelitis), or a limp, refusal to walk or to use the upper extremities. To avoid duplication, the major or most prominent symptom was counted for children who presented with more than one complaint such as both bone and joint problems (16 patients). In an attempt to elucidate their biological significance, the patients presenting with MSM were compared to patients without these symptoms on variables regarding leukemia characteristics, early response to treatment and outcome.
Statistical analysis
Using SPSS version 12.0 (SPSS Inc.), we performed chi-square tests, t tests and Wilcoxon tests to examine possible relationships between the presence of musculoskeletal complaints and medical and demographic variables. The Mantel–Haenszel test was applied to control for confounding variables in some of the analyses for possible relationships. In addition, logistic regression analysis was carried out to perform multivariate analysis of these variables. Kaplan–Meier curves, the log–rank test and the Cox regression model were used to assess the relevance of MSM as a prognostic factor for event-free survival (EFS); events included relapses, second malignancies and death from any cause. All the tests and summary statistics took into account all patients with complete data on the particular variables included in that analysis. Some analyses excluded patients in categories with very small numbers of subjects (e.g., comparisons by immunophenotype are limited to B cell precursor and T cell).
The sample size achieved 80% power, for a 5% two-sided test, for differences of 0.27 standard deviations for continuous outcomes and an absolute difference of 0.13 for dichotomous outcomes. For analyses that were restricted to the two most recent national studies, the corresponding differences are 0.30 standard deviations and 0.15 absolute difference, respectively. For analyses restricted to B cell (T cell) patients, the corresponding differences are 0.27 and 0.14, respectively, for B cell, and 1.31 and 0.66, respectively, for T cell.
Results
Data on MSM were available for 765 out of 783 children with ALL, of whom 240 patients (31.4%) reported orthopedic complaints upon diagnosis. Bone-related complaints were the most frequent MSM complaints with 124 out of 765 children (16.2%), followed by joint involvement in 72 patients (9.4%). Thirty-seven patients (4.8 %) were found to have walking complaints and seven children (0.9%) had other MSM findings. The MSM subgroups did not differ with respect to any of the medical, demographic or outcome variables (data not shown).
The most outstanding finding is the high frequency of orthopaedic complaints among the children with B cell precursor (BCP) leukaemia, 209 patients out of 576 (36.3%), compared to only 25 out of 176 children with T cell leukemia (14.2%) (P < 0.0001). The remaining 13 patients did not have sufficient immunophentypic data for classification. High-quality cytogenetic data were available only for the latest 312 patients in the INS-98 series, and the examination of the 237 children with B cell lineage leukemia in that study showed no relationships between MSM and major ALL cytogenetic aberrations. Among the patients with MSM we also found smaller liver and spleen sizes (P < 0.001), lower WBC (P < 0.001), lower peripheral blast percentage (P < 0.001), higher haemoglobin (Hb) levels (P < 0.001) and higher platelet counts (P = 0.006) (Table 1). Some of these differences, most notably the lower WBC count and less hepatosplenomegaly in the MSM group remained significant even after adjustment to the immunophenotype (Tables 2, 3).
Leukemia characteristics and outcome of patients with MSM compared to the group without MSM (765 patients)
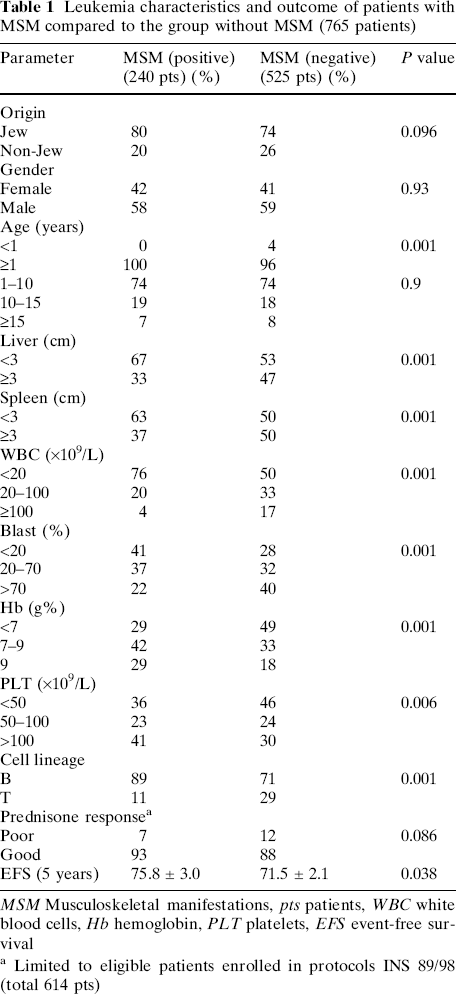
MSM Musculoskeletal manifestations, pts patients, WBC white blood cells, Hb hemoglobin, PLT platelets, EFS event-free survival
Limited to eligible patients enrolled in protocols INS 89/98 (total 614 pts)
B-lineage leukemia characteristics and outcome of patients with MSM compared to the group without MSM (576 patients)
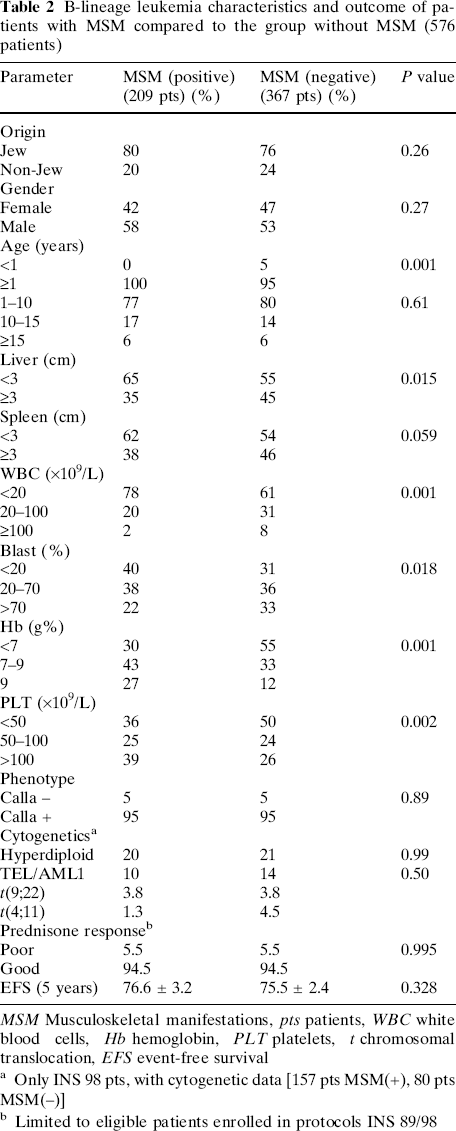
MSM Musculoskeletal manifestations, pts patients, WBC white blood cells, Hb hemoglobin, PLT platelets, t chromosomal translocation, EFS event-free survival
Only INS 98 pts, with cytogenetic data [157 pts MSM(+), 80 pts MSM(−)]
Limited to eligible patients enrolled in protocols INS 89/98
T cell leukemia characteristics and outcome of patients with MSM compared to the group without MSM (176 patients)
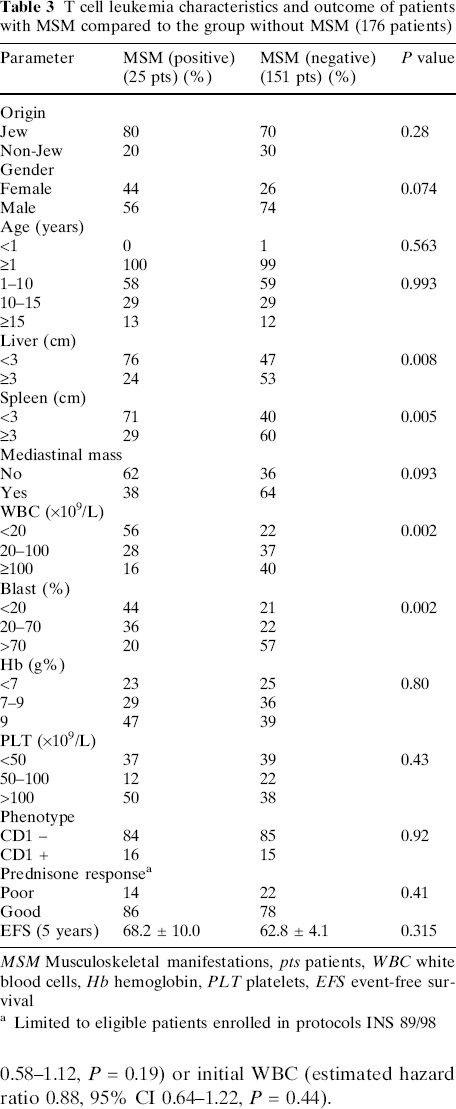
MSM Musculoskeletal manifestations, pts patients, WBC white blood cells, Hb hemoglobin, PLT platelets, EFS event-free survival
Limited to eligible patients enrolled in protocols INS 89/98
The early response to prednisone (Table 1) and event-free survival (EFS) were slightly better for patients with MSM (Fig. 1). The five-year EFS for patients with MSM was 75.8% (95% CI 69.8–81.8%) and for patients without MSM was 71.5% (95% CI 67.3–75.7%). However, much of these differences in outcome can be accounted for by the low proportion of T cell leukemia in the MSM group (Tables 2, 3) and the differences in initial WBC. No significant relationship of MSM to EFS is found after using the Cox regression model to adjust for either immunophenotype (estimated hazard ratio of 0.81, 95% CI 0.58–1.12, P = 0.19) or initial WBC (estimated hazard ratio 0.88, 95% CI 0.64–1.22, P = 0.44).
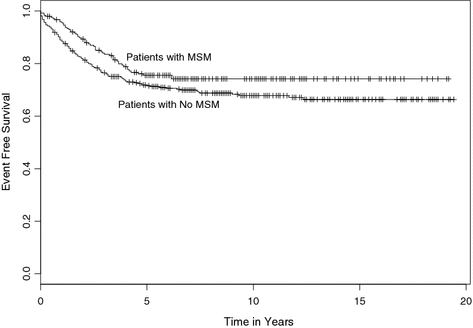
Kaplan–Meier event-free survival curves for children with (upper curve) and those without (lower curve) orthopaedic complaints at presentation. The low proportion of T cell leukemia and in the MSM group and the differences in initial WBC between the groups can account for the differences in outcome
Discussion
Our research is the first to assess the significance of musculoskeletal symptoms of leukemic origin for pediatric ALL patients by using summarized data from a prospectively conducted multicenter treatment protocol. The large database served as well as a national registry of all ALL diagnosed pediatric patients in Israel, and made possible a much more precise estimate of the proportion of affected children presenting with MSM (31.4% of 765 children). Furthermore, it allowed us to investigate the largest pediatric population for which both MSM data and immunophenotype analysis were available.
Of all childhood malignancies, excluding musculoskeletal tumors, acute lymphoblastic leukemia is the most frequently associated with musculoskeletal symptoms at presentation [19]. Several clinical and laboratory features at diagnosis were found to have prognostic value for the children with ALL, and have been grouped in risk-stratified clusters for treatment purposes. Beside the classical NCI consensus age and WBC count criteria, the outcome-focused groupings may include, according to various treatment protocols, involvement of central nervous system and liver, spleen, gender, race, as well as the biological makeup of leukemic cells and the early response to treatment [12, 22, 23, 25, 26]. The prognostic value of some of these outcome determinants was shown to weigh differently for B-precursor and T cell leukemia [26].
Our study investigated the hypothesis that the presentation of ALL with musculoskeletal complaints might be more common in patients in whom the leukemia has a slower progression with a tendency to affect primarily the intramedullary space and structures, such as BCP ALL. Musculoskeletal manifestations of leukemic origin can be regarded, therefore, as the overt expression of an intramedullary pattern of leukemic cells infiltration, which becomes obvious at a relatively early stage. Unlike the T cell ALL, which most probably stems from the thymus and only subsequently spreads to the bone marrow, the BCP ALL starts in the bone marrow itself. This distinction in origin may constitute a biological explanation for our finding a much higher frequency of MSM among the patients with B-precursor than in those with T cell ALL, in which the involvement of other internal structures (CNS, hepatosplenomegaly or mediastinal mass) is more likely to be found [12, 26, 27]. It seems, therefore, that the clinical picture of ALL at presentation reflects an intramedullary or extramedullary pattern of infiltration of leukemic cells. Our findings strongly suggest that MSM are likely to occur in leukemias with a greater biological affinity to bone marrow and consequently, with a tendency to affect primarily the adjacent structures and the intramedullary space.
MSM at diagnosis among the children with BCP ALL were associated with more-favorable laboratory findings, of which the most significant is a lower peripheral blast count. In addition, our results confirmed previous reports that small liver and spleen, and lower WBC are common findings in patients with MSM, regardless of cell lineage [18, 20].
Our study highlights the importance of including ALL in the differential diagnosis of MSM even in the presence of an apparently normal routine automated complete blood counts (CBC). It is thus recommended to obtain a differential WBC count in any child with vague or prolonged and unexplained musculoskeletal symptoms. Specifically we suggest the performance and examination of a Giemsa stained smear from any cellular joint aspiration in children. “Soft” signs in the WBC, for example mild thrombocytopenia, should also alert the orthopedist to the possibility of bone marrow infiltration by leukemia or other tumor. Most importantly, it is highly recommended to consider bone marrow examination before commencement of steroids or other drugs (e.g. methotrexate) that may further occult a missed or incipient intramedullary leukemia.
Footnotes
Acknowledgments
This study was partially funded by the Israel Cancer Association and The Goldberg Family Chair in Pediatric Surgery, Sackler Faculty of Medicine, Tel Aviv University—Incumbent: Prof. Shlomo Wientroub. The commitment of the principal investigators in The Israeli National Childhood ALL Study Group has been greatly appreciated: Dina Attias, M.D., Bnai Zion Medical Center, Haifa; Gali Avrahami, M.D., and Batia Stark, M.D., Schnieder Children's Medical Center, Petah Tiqva; Ami Ballin, M.D., Edith Wolfson Medical Center, Holon; Bella Bielorai, M.D., and Shai Izraeli, M.D., Chaim Sheba Medical Center, Tel Hashomer; Yoav Burstein, M.D., Dana Children's Hospital, Tel Aviv Sourasky Medical Center, Tel Aviv; Ronit Elhasid, M.D., Rambam Medical Center, Haifa; Gabriel Herzel, M.D., Ha'Emek Medical Center, Afula; Yosef Kapelushnik, M.D., Soroka Medical Center, Beer Sheva; Hagit Miskin, M.D., Shaare Zedek Medical Center, Jerusalem; Dalia Sthoeger, M.D., Kaplan Medical Center, Rehovot; Michael Weintraub, M.D., Hadassah University Hospital, Jerusalem. We thank Galli Pen and Dina Kugel for maintaining the INS database, and Irina Opincariu for editing the manuscript.
