Abstract
Stable, nontoxic, and aqueous Cu:ZnInSe quantum dots (QDs) have been synthesized using one-step method. Through controlling pH, the amount of 3-mercaptopropionic acid (MPA), and impurities Cu and In, high-quality Cu:ZnInSe QDs have been obtained. First, as-prepared Cu:ZnInSe QDs have better thermal stability. The photoluminescence (PL) intensity and wavelength are not changed during long-time heating reflux. Besides, the synthesizing method is simple, and raw materials are green. Cu:ZnInSe QDs are perfect fluorescent powder.
Keywords
1. Introduction
Due to the high cytotoxicity of Cd-contained QDs, more Zncontained QDs have attracted the attention of people, such as doped Cu:ZnSe [1, 2], Ag:ZnSe [3], and Mn:ZnSe [4, 5]. Pure ZnSe QDs, for their emission is in the ultraviolet range, could not be applied in light-emitting diode (LED) and quantum dots sensitized solar cell (QDSSC) fields. There are many problems for doped ZnSe QDs. The first one is that the stability of doped ZnSe is weak, which is caused by the lattice mismatch of the impurities and QDs. As we know, the lattice mismatch would result in metal impurities tending to present on the surface of QDs and make photoluminescence (PL) quenched easily when they are applied in various fields [6]. Recently, our group has reported internally doped Cu:ZnSe QDs in aqueous solution and improved the stability of Cu:ZnSe QDs with ZnS shell [7]. Besides, we have found that the excellent stability of as-prepared QDs is attributed to the simultaneous preparation of core-shell QDs and internally doped impurities. At the same time, the mechanism for the stability of internally doped Cu:ZnSe aqueous QDs has been investigated [8]. The second one is that the synthesizing method is complex, which is difficult to apply in mass production. Thus, it is important to develop a new simple method to bulk-producing stable doped ZnSe aqueous QDs. In this paper, Cu-doped ZnInSe QDs have been synthesized in aqueous. The synthesizing method is simple, which can be used for mass production. Besides, doped Cu:ZnInSe QDs have good stability for the intervention of In. Thus, the as-prepared Cu:ZnInSe QDs may be used as luminescent layer of LED, i.e., fluorescent powder.
2. Experimental Section
2.1 Synthesis of Cu and In co-doped ZnSe QDs
All materials used in this work were analytical reagents. Zn(NO3)2 and CuCl2 were purchased from Sinopharm Chemical Reagent Co., Ltd. NaBH4 was purchased from Guangdong Chemical Reagent Engineering Technological Research and Development Center. NaOH was purchased from Shanghai Zhongshi Chemical Company. 3-mercaptopropionic acid (MPA), In(NO3)3 6H2O, and Se powder were purchased from Aldrich. NaHSe solution was prepared using Se with NaBH4 according to the reference methods [9, 10].
Aqueous Cu and In co-doped ZnInSe QDs were synthesized according to our previous work [3]. Zn(NO3)2, AgNO3, In(NO3)3, and MPA mixture were adjusted to pH using NaOH solution (with concentration 5mol/L). Freshly prepared NaHSe solution was injected into the mixture after the solution was aerated with N2 for about 30 min. Then, the solution was refluxed at 100°C. The total concentration of Zn in the mixture is 0.01 mol/L.
2.2 Characterization
UV-vis absorption spectra (UV) were recorded with a Shimadzu 3600 UV-vis near-infrared spectrophotometer. Fluorescence experiments and PL decay experiments were performed with an Edinburg FLS 920 spectrofluorimeter. The excitation wavelength was 360 nm. All optical measurements were performed at room temperature under ambient conditions. X-ray photoelectron spectroscopy (XPS) was investigated using a PHI550 spectrometer with Mg Kα excitation (1253.6 eV). Binding energy calibration was based on C 1s at 284.6 eV. X-ray powder diffraction (XRD) investigation was carried out using the D/max-2500/PC diffractometer with CuKα radiation (λ=1.5418Å). For XPS and XRD measurement, Cu:ZnInSe QDs powder were used. To obtain the powder, freshly prepared QDs were precipitated from solution by addition of equal volume of isopropanol [10] and then dried at vacuum. Fluorescent images of cells were taken on an Olympus FluoView™ FV1000 fluorescence microscope.
3. Results and Discussion
3.1 The influence of pH, the amount of MPA, and the ratio of raw materials
Both the PL intensity and the range of wavelength of QDs are related to pH value, amount of ligand, and impurities. The following has obtained the optimal experimental data through comparing the change of PL and absorbance spectra at different condition.
First, the PL and absorbance spectra of Cu:ZnInSe QDs with different pH are shown in Figure 1. QDs solution with Cu:In:Zn:MPA:Se being 0.01:0.03:1:2:0.2 have grown for 2 hours at pH 7.0, 8.0, 9.0, 10.0, and 11.0. There is little influence of pH on absorption spectra, showing that pH does not influence the size of QDs. However, the influence of pH on the intensity of PL is obvious. For the intensity of PL, it is different at pH 7.0, 8.0, 9.0, 10.0, and 11.0. At pH 8.0, the intensity of PL of QDs is the strongest. The positions of PL peaks are almost at the same location, which is consistent with the results of the absorption spectra. For the samples at pH 7.0, 8.0, 9.0, 10.0, and 11.0, they have the same absorption spectra. Namely, pH of solution has little influence on the size of QDs.

UV-vis spectra (a) and PL spectra (b) of MPA-capped Cu:ZnInSe QDs with different pH values 7.0, 8.0, 9.0, 10.0, and 11.0. The intensity of PL is relative (RL) PL intensity.
Second, the PL and absorbance spectra of Cu:ZnInSe QDs with different amount of ligand (MPA 177, 354, 708, and 1416 uL) are shown in Figure 2. QDs solution with Cu:In:Zn:Se being 0.01:0.03:1:0.2 and pH8.0 have grown for 2 hours. Clearly, the amount of MPA has influence on the absorption and PL spectra. Seen from the absorbance spectra, with increasing of the amount of MPA from 354 to 1416 uL, the absorption spectra shift to blue. Blue shift of absorption spectra may be caused by quantum size effect. Namely, with increasing of the amount of MPA, the size of Cu:ZnInSe QDs becomes small. For the sample with MPA 177 uL, there is obvious aggregation in solution. In addition, the intensity of PL spectra increases from 177 to 354 uL and decreases from 354 to 1416 uL. Thus, the optimal amount of MPA for Cu:ZnInSe QDs is 354 uL.

UV-vis spectra (a) and PL spectra (b) of MPA-capped Cu:ZnInSe QDs with different amount of MPA 177, 354, 708, and 1416 uL
Third, the PL and absorbance spectra of Cu:ZnInSe QDs with different amount of impurity Cu (100, 200, 400, and 800 uL) are shown in Figure 3. QDs solution with In:Zn:MPA:Se being 0.03:1:2:0.2 and pH 8.0 have grown for 2 hours. Absorbance spectra shift to red with increasing of the amount of Cu. It is to say that the amount of Cu has influence on the size of ZnSe QDs. However, the difference of size is so small that could not be showed in transmission electron microscopy (TEM), high-resolution (HR) TEM. As for PL spectra, the intensity of PL is decreasing with increasing of the amount of Cu. When the amount of Cu is zero, there is no luminescence for ZnInSe QDs. Thus, impurity Cu takes important role in Cu:ZnInSe QDs. In fact, the energy bands of In and ZnSe are not matched. The energy band of Cu is matched with ZnSe and In. Thus, Cu is the bridge of energy bands of ZnSe and In (Scheme 1). At first, impurity Cu captures the hole of ZnSe. Then, the hole in energy band of Cu is captured by impurity In. The emission is from the conduction band of ZnSe to impurity band of In. Comprehensive consideration the PL intensity and function of impurity of Cu, the optimal amount of Cu is 200 uL.

UV-vis spectra (a) and PL spectra (b) of MPA-capped Cu:ZnInSe QDs with different amount of Cu

Schematic illustration of the possible relationship among the energy band of Cu:ZnInSe QDs
Fourth, the PL and absorbance spectra of Cu:ZnInSe QDs with different amount of impurity In (400, 600, 800, and 1000 uL) are shown in Figure 4. QDs solution with Cu:Zn:MPA:Se being 0.01:1:2:0.2 and pH 8.0 have grown for 2 hours. The amount of In has little influence on the absorbance spectra. Namely, the amount of In does not influence the growth of QDs. The shapes of PL spectra for Cu:ZnInSe QDs of different amount of In are the same but their PL intensity is different. The PL peaks of Cu:ZnInSe QDs are at 561 nm for Cu:ZnInSe QDs of the amount of In 400, 600, 800, and 1000 uL. However, when the amount of In is lower than 400 uL, the PL peak position of Cu:ZnInSe QDs has been shifted to blue, which is emission of Cu:ZnSe QDs (the dotted line and black line in Figure 4b). Therefore, the range of amount of In at Cu:ZnInSe QDs is from 400 to 600 uL.

UV-vis spectra (a) and PL spectra (b) of MPA-capped Cu:ZnInSe QDs with different amount of In. The dotted line and black line in (b) are PL spectra of Cu:ZnInSe QDs with amount of In 100 uL.
3.2 Stability an application of Cu:ZnInSe QDs
Cu:ZnInSe QDs synthesized under pH 8.0 and Cu:In:Zn:MPA:Se being 0.01:0.03:1:2:0.2 condition have good thermal stability and high quantum yields (7%). As we have known, QDs have quantum size effect. For aqueous QDs, their size increase with growth time. In this way, the PL wavelength of QDs has been changed when they are applied in LED and QDSSC. And even the emission color of QDs is changed under room temperature. For Cu:ZnInSe QDs, the wavelength of PL peaks has little change (from 558 to 547 nm) after refluxing lasted 17 hours (Figure 5). Moreover, PL intensity of Cu:ZnInSe QDs increased with the growth time from 1 to 19 hours. Then, the PL intensity keeps in this data. After refluxing for about 72 hours, PL intensity begins to reduce. Thus, Cu:ZnInSe QDs have good thermal stability. The PL peak position is stable, and PL intensity keeps stable under refluxing, which is important for application field of LED.
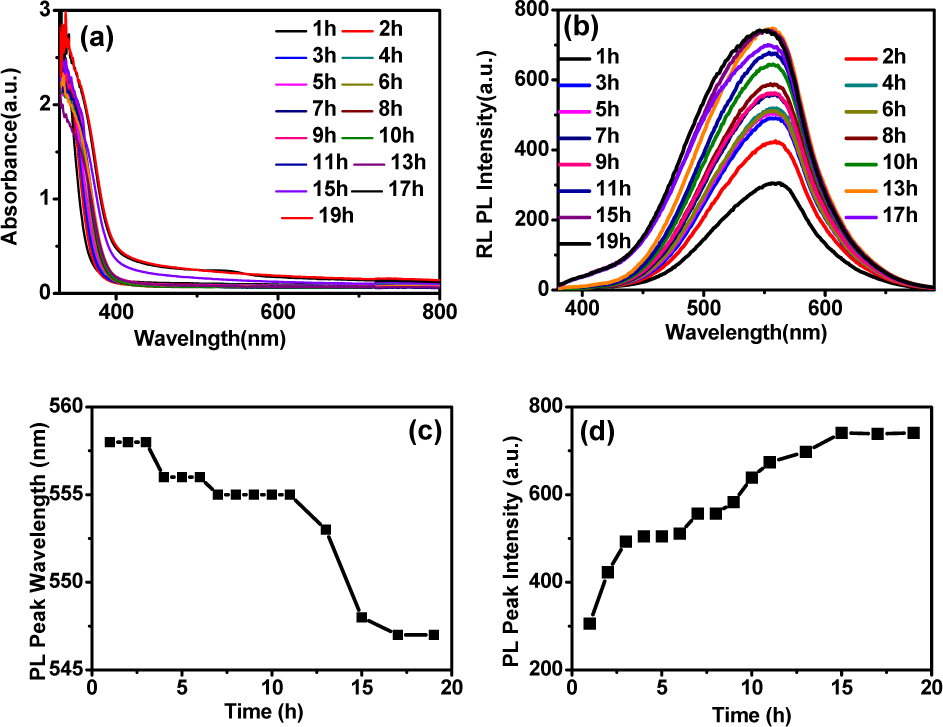
Time-dependent UV-vis spectra (a) and PL spectra (b) of MPA-capped Cu:ZnInSe QDs. Time-dependent normalized PL peak intensity (c) and PL peak position (d) of MPA-capped Cu:ZnInSe QDs.
In addition, Cu:ZnInSe QDs are easy to deposit to obtained powder (Figure 6, left). Under the optimal condition, synthesized Cu:ZnInSe QDs are deposited using centrifugal machine with lower rotation rate. Moreover, the productivity of Cu:ZnInSe QDs/QD-solution is 2 g/100 mL. Cu:ZnInSe QDs powder has good PL characters. The powder has good crystal type and atom mole rate. XRD data of Cu:ZnInSe QDs synthesized under pH 8.0 and Cu:In:Zn:MPA:Se being 0.01:0.03:1:2:0.2 condition are depicted in Figure 6. The peaks of Cu:ZnInSe QDs are the same with 110, 220, and 311 of ZnSe. The impurities have not changed the lattice of ZnSe, which may be the reason of Cu:ZnInSe QDs having good stability.
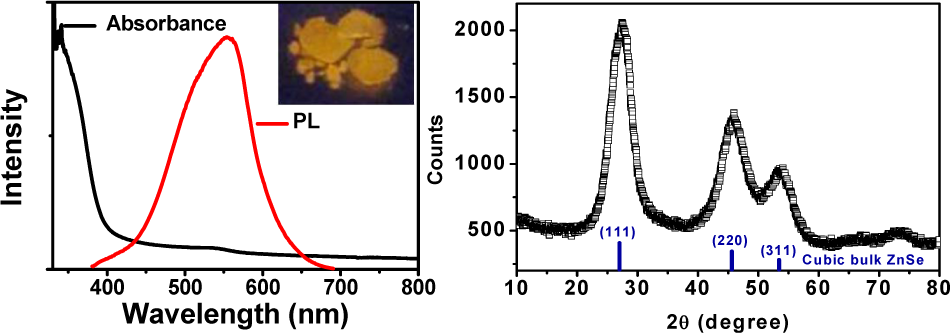
Left: UV-vis absorption and PL spectra of Cu:ZnInSe QDs synthesized under pH 8.0 and Cu:In:Zn:MPA:Se being 0.01:0.03:1:2:0.2 condition. Inset image is a photo of Cu:ZnInSe QDs powder under the irradiation of a UV lamp. Right: XRD of Cu:ZnInSe QDs powder under the above condition.
XPS data of Cu:ZnInSe QDs powder are depicted in Figure 7. The experimental data of Cu:In:Zn:Se:S are 0.01:0.03:1:0.52:0.74, which are similar to the raw atomic rate (0.01:0.03:1:2:0.2). It is to say that total Cu and In of raw is doped into ZnSe QDs. The amount of Cu and In in Cu:ZnInSe QDs is lower, but there is obvious peaks in XPS figure. The change of Zn:Se:S ratio between experimental data and raw atomic rate is caused by the excessive addition of Zn in raw. Seeing from the peak positions of XPS, the valent state of Cu and In is +2 and +3. In addition, TEM and HRTEM figures (Figure 8) show that Cu:ZnInSe QDs are about 3.5 nm.
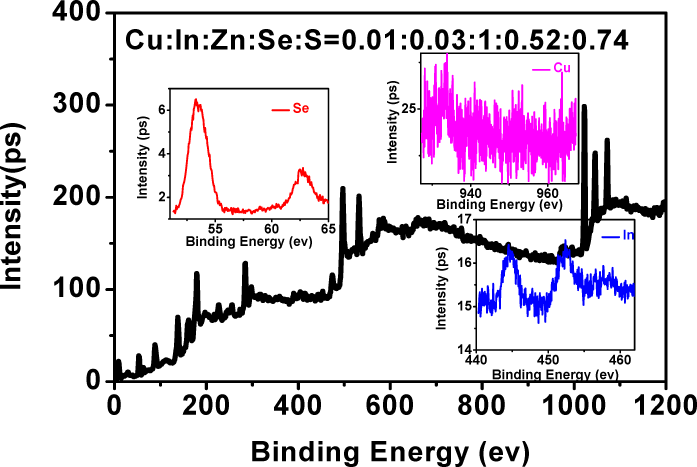
XPS (right) of Cu:ZnInSe QDs synthesized under pH 8.0 and Cu:In:Zn:MPA:Se being 0.01:0.03:1:2:0.2 condition

TEM and HRTEM (inset) of Cu:ZnInSe QDs under pH 8.0 and Cu:In:Zn:MPA:Se being 0.01:0.03:1:2:0.2 condition. The scale bars are 10 nm (TEM) and 4 nm (HRTEM)
4. Conclusions
Aqueous and nontoxic Cu:ZnInSe QDs powder with good stability has been synthesized using one-step method. First, influence of pH, the amount of MPA, and impurities has been investigated. On this basis, high-quality Cu:ZnInSe QDs have been obtained with good thermal stability. The PL intensity and wavelength are not changed during long-time heating reflux. Second, Cu:ZnInSe QDs powder has been obtained with simple method. The synthesizing process and purification method, raw materials are green. Thus, Cu:ZnInSe QDs powder has potential application in LED field as fluorescent powder.
Footnotes
5. Acknowledgements
This work is supported by the National Key Basic Research Program of China (Grant No. 2015CB352002), National Natural Science Foundation of China (Grant Nos. 61475034, 21403034, 61177033), the Fundamental Research Funds for the Central Universities (No. 2242014R30006), and the natural science foundation of Jiangsu Province Youth Fund (No. BK20140650), China Postdoctoral Science Foundation (No. 2014M560370, 2015T80477), Jiangsu Planned Projects for Postdoctoral Research Funds (No. 1401035B).
