Abstract
We reported a facile method for preparing self-assembled noble metal nanoparticle chains by using lysozyme amyloid fibrils as a biotemplate in an aqueous environment. The nanoparticle chains of gold (AuNPCs), palladium (PdNPCs), platinum (PtNPCs) and rhodium (RhNPCs), which are lysozyme fibrils coated by gold, palladium, platinum and rhodium nanoparticles, can be fabricated by simply reducing the corresponding metal salt precursors using NaBH4. Under the same molar ratio between salt precursors and fibrils, two types of morphologies of high-yield AuNPCs (thin- and thick- AuNPCs) were synthesized as a result of adjusting the fibrosis time and temperature in the final stage. Abundant PdNPCs with a length of several micrometres intertwisted with each other to form PdNPC networks. The growth of RhNPCs started from the inner surface of the fibrils and gradually spread to the whole fibre as superabundant rhodium nanoparticles (RhNPs) bound to the fibrils. Finally, PtNPCs at different growing periods were presented. The nanostructures were investigated by transmission electron microscope, UV-visible spectroscopy, fluorescence spectroscopy, energy-dispersive X-ray spectroscopy and atomic force microscope.
1. Introduction
Noble metal nanoparticles, with their unique electrical, optical and magnetic performance and various potential applications, have become one of the popular fields in nanoscience because of their interesting shape-dependent and size-dependent physical and chemical properties [1]. To optimize and extend the morphology of noble metal nanoparticles, biological self-assembled methods have been developed due to the following advantages of biomolecules, which are: (1) the charged and chemically reactive moieties with amine and carboxyl groups attracting with other chemical molecules, (2) their natural substrate-specific affinity assembling and aligning the biomolecule in a specific pattern, (3) transforming substrates into other forms for the enzymatic activity of biomolecules, which can be employed to synthesize nanostructured materials [2]. Up till now, numerous studies on the synthesis of metal nanoparticles have been achieved through the use of biological self-assembled methods. Mudalige Thilak Kumara and colleagues have effectively yielded metal nanoparticles and nanotubes on bioengineered Flagella scaffolds [3]. Tonggai Zhang and colleagues have synthesized ultrathin platinum nanowires using insulin fibrils as sacrificial templates [4]. In our study, we exploited self-assembled amyloid fibres from lysozyme, which exists extensively in nature, as biotemplates to synthesize noble metal nanoparticle chains.
To date, amyloid fibrils, which share a common β-sheet rich structure [5], have been extensively studied both theoretically and experimentally. They are known for their association with conformational diseases caused by protein misfolding, such as Alzheimer's disease, late onset diabetes and Parkinson's disease [6-9]. Nevertheless, amyloid fibrils have the outstanding advantages mentioned above, making them ideal materials and components for advanced nanotechnology. Therefore, the advantage of the spontaneous fibrillation mechanism of amyloid fibrils under conditions of low pH and high temperature [10] can be employed to control the nanostructure of metal nanoparticles.
Lysozyme, a type of amyloid protein used in this research, is composed of 129 amino acids. It is a monomeric globular protein with a high percentage of lysine and arginine [11], which was possibly the main action group of the predominantly electrostatic interactions [12]. Herein, noble metal nanoparticle chains were synthesized by a facile method in which lysozyme fibrils were exposed to gold, palladium, platinum and rhodium precursors separately. By adjusting the experimental conditions, different morphologies of gold nanoparticle chains (AuNPCs), palladium nanoparticle chains (PdNPCs), rhodium nanoparticle chains (RhNPCs) and platinum nanoparticle chains (PtNPCs) were presented.
2. Materials and Methods
2.1 Materials
Lysozyme from hen egg white (HEWL) was supplied by Beijing Biodee Biotechnology Co., Ltd. Potassium chloroplatinate (KPtCl6) and palladium chloride (PdCl2) were purchased from Tianjin Chemical Reagent plant (Tianjin, China). Rhodium chloride (RhCl3), chloroauric acid (HAuCl4) and sodium borohydride (NaBH4) were obtained from Chengdu West Chemical Co., Ltd. (Chengdu, China). Magnesium chloride (MgCl2) was purchased from Tianjin Fengchuan Chemical Reagent Technology Co., Ltd. (Tianjin, China). Hydrochloric acid (HCl) was bought from Gu'an Chemical plant (Langfang, China). Milli-Q reagentgrade water was home-prepared.
2.2 Methods
2.2.1 Fabrication of gold nanoparticle chains (AuNPCs)
HEWL powder was dissolved into 25 mM hydrochloric acid (HCl) to form 3 mg/ml HEWL-HCl solution. To induce the fibrosis of lysozyme, the sample was heated without disturbance in a thermostatic metal bath at 70 °C for 77 h. Then 5 mM chloroauric acid and 25 mM HCl mixture were added to the mature fibrils. After homogeneous mixture, it was incubated at ambient temperature for 24 h. Subsequently, the reducing agent of excess fresh NaBH4 (5 mM) solution was injected into the mixture, drop by drop. In the contrast experiment, we changed the experimental conditions and the fibrosis time (73 h), and heated the mixture after reducing it to improve the quantity of gold NPs coated on the HEWL fibrils.
2.2.2 Fabrication of palladium nanoparticle chains (PdNPCs)
HEWL powder was dissolved with 0.1 MMgCl2 and 25 mM HCl mixed solution. The target concentration was 3 mg/mL. It was then placed in a thermostatic metal bath at 70 °C for 24 h. The HEWL fibrils were not completely mature at that moment. Next, the 2.5 mM PdCl2 solution was added to the HEWL fibrils, followed by incubation for 48 h at room temperature. Subsequently, the reducing agent of excess fresh NaBH4 (5 mM) solution was injected into the mixture, drop by drop.
2.2.3 Fabrication of rhodium nanoparticle chains (RhNPCs)
HEWL powder was dissolved into 25 mM hydrochloric acid (HCl) to form 3 mg/ml HEWL-HCl solution. To induce the fibrosis of lysozyme, the sample was heated without disturbance in a thermostatic metal bath at 70 °C for 48 h. Then 2.5 mM rhodium chloride was added into the HEWL fibrils and incubated for 24 h. Finally, the sample was also reduced with excess fresh NaBH4 (15 mM) solution, drop by drop.
2.2.4 Fabrication of platinum nanoparticle chains (PtNPCs)
HEWL powder was dissolved with 0.1 MMgCl2 and 25 mM HCl mixed solution. The target concentration was 3 mg/mL. It was then placed in a thermostatic metal bath at a constant 70 °C for 60 h. Subsequently, the 2.5 mM K2PtCl6 solution was added to the HEWL fibrils, following incubation for 24 h at room temperature. Finally, the sample was also reduced with excess fresh NaBH4 (5 mM) solution, drop by drop.
A summary of these four samples and the conditions used for the synthesis are presented for reference in Table 1 below.
Synthesis conditions for four different metals

Transmission electron microscope (TEM) images of the metal nanoparticle chains were observed by using a Hitachi model HT-7700 instrument with an accelerating voltage of 100 kV. TEM samples were prepared by placing a droplet of the sample solution onto copper grids coated with amorphous carbon film and dried at room temperature [13]. The ultraviolet-visible (UV-vis) absorption spectra of AuNPCs were measured by a WFZ-26A UV–vis spectrophotometer. The fluorescence of AuNPCs under the Xe laser excitation was measured by a Hitachi model F-7000 instrument. The structure of metal nanoparticle chains was examined by selected area of electrical diffraction (SAED) and high-resolution transmission electron (HRTEM) on a JEM model 2010 instrument. To determine the component elements of RhNPCs, energy-dispersive X-ray spectroscopy (EDS) analysis was performed on the HT-7700 instrument.
3. Results and discussion
3.1 Fabrication of gold nanoparticle chains (AuNPCs)
Abundance of AuNPCs, which were determined by transmission electron microscopy (TEM), were highly yielded by a facile method, as shown in Figure 1a. The average length of AuNPCs was several micrometres and the longest reached to 12 μm. For further observation, a randomly selected AuNPC with a length of 6.8 μm is displayed in Figure 1b, which shows the mature fibrils coated with a few AuNPs. For convenience, the AuNPCs with a few AuNPs are called thin-AuNPCs for short. Figure 1c shows a higher magnification image of the randomly selected thin-AuNPC, which exhibits the double helix structure. The structure of thin-AuNPCs was investigated by means of SAED patterns (Figure 1e), whose continuous rings from inner to outer are accordant with the (111), (200), (220) and (311) planes of the expected face-centred cubic. Additional structural characterizations of thin-AuNPCs were performed using HRTEM, which indicates their crystalline nature. The HRTEM image showing different crystal planes is shown in Figure 1d. The smallest spacing between two crystal planes is 0.2348 nm, corresponding to the (111) lattice spacing of Au.

TEM images of (a) thin-AuNPCs and (f) thick-AuNPCs; (b) low and (c) high magnifications of a typical single thin-AuNPC; (g) low and (h) high magnifications of a typical single thick-AuNPC; high resolution images of (d) thin-AuNPCs and (i) thick-AuNPCs; SAED pattern of (e) thin-AuNPCs and (j) thick-AuNPCs.
To increase the quantity of AuNPs attaching to the HEWL fibrils, we changed the experimental conditions, fibrosis time and temperature in the final stage, instead of changing the molar ratio [14] between salt precursors and HEWL fibrils. The AuNPCs obtained in these conditions are called thick-AuNPCs, as exhibited in Figure 1f. The longest of the thick-AuNPCs was up to 15 μm. Similarly, we randomly selected one with a length of 9.3 μm, as indicated in Figure 1g, which was magnified (Figure 1h). From this, a large quantity of AuNPs bound to the mature fibrils were observed, compared with the thin-AuNPCs mentioned above. Figure 1h also indicated the helix structure of thick-AuNPCs. For comparison with the structure of thin-AuNPCs, thick-AuNPCs were also studied by means of SAED patterns (Figure 1j), the result of which corresponded to that of the thin-AuNPCs. Nevertheless, the lattice spacings in the HRTEM image of the thick-AuNPCs were 0.2031 nm and 0.2375 nm, which correspond with the (200) and (111) lattice planes respectively in the face-centred cubic Au.
Figure 2 shows the UV-visible absorption spectra recorded from thin-AuNPCs and thick-AuNPCs. It is known that the main feature of the absorption spectra for metallic nanoparticles is of the surface plasma resonance (SPR) band [15]. As can be seen, both thin-AuNPCs and thick-AuNPCs displayed only a single absorption band attributed to the collective dipole oscillation (surface plasma resonance, SPR) at 530 nm due to the elongated structure. Hence, it can be confirmed that the AuNPCs we prepared were spherical-like [16] nanoparticles arranged into elongated NPCs, corresponding to the TEM above. Furthermore, the peak of thick-AuNPCs was stronger in comparison with that of thin-AuNPCs, which might be attributed to the abundance of AuNPs bound to the HEWL fibrils. The colours of the thin-AuNPC and thick-AuNPC solutions were pink and burgundy (inset), respectively.
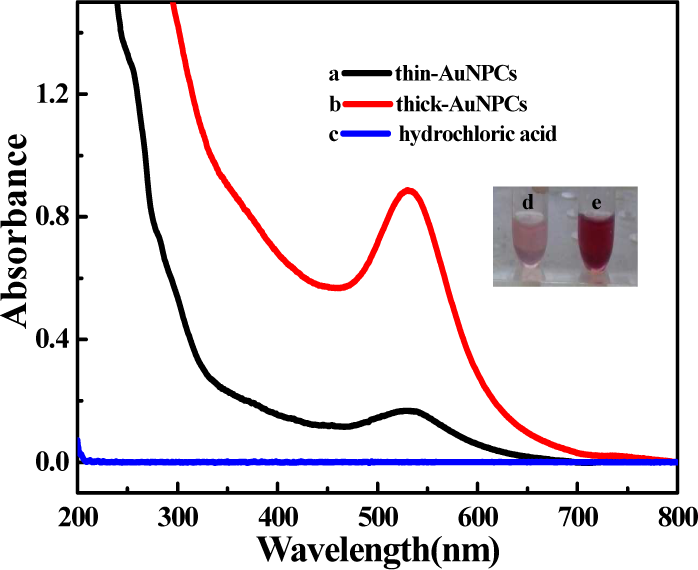
The UV-vis spectra of (a) thin-AuNPCs and (b) thick-AuNPCs. The insert shows the samples of (d) thin-AuNPCs and (e) thick-AuNPCs.
The fluorescence of thin-AuNPCs and thin-AuNPCs with the concentration of 0.17 mM was further investigated, as displayed in Figure 3. For nanometre-size or sub-nanometre-size gold particles, both small size effect and surface effect should be considered and the emission intensity and band position are sensitive to the morphology of the nanoparticles [17]. As can be seen in Figure 3, the fluorescence peak was displayed in the blue-violet wavelength region. This may be mainly attributed to some gold clusters formed in a certain percentage because of the HEWL fibril groups' (amino acid residues in HEWL fibril) confined space effect. These sub-nm Au nanoclusters [18–19] are too small to have a continuous density of states, but rather show quantum confined electronic transitions and thus are fluorescent. As indicated in Figure 3, the fluorescence spectra of thick-AuNPCs centred at 412 nm and 440 nm was blueshifted in comparison with that of thin-AuNPCs centred at 425 nm and 460 nm because of the smaller Au cluster size of thick-AuNPCs. The fluorescence intensity of thick-AuNPCs was stronger than that of thin-AuNPCs due to the higher load of gold nanoclusters on the HEWL fibrils.

The fluorescence spectra of thick-AuNPCs and thin-AuNPCs
3.2 Fabrication of palladium nanoparticle chains (PdNPCs)
TEM measurements were carried out on the palladium metallized HEWL fibrils. The TEM images in Figures 4a and b show the PdNPCs at different magnifications. The low-magnification TEM image (Figure 4a) reveals the presence of abundant PdNPCs with length of several micrometres intertwisted with each other to form PdNPC networks. The high-yield production of PdNPCs demonstrated that the observed features are indeed representative. A high-magnification image of a typical single PdNPC selected randomly is shown in Figure 4b. It indicates that the PdNPs coated on the HEWL fibrils grew along the double helix structure. In addition, the selected-area electron diffraction (SAED) pattern recorded from a few PdNPCs was performed (Figure 4c). The values in the SAED pattern correspond to the (220) and (311) planes of the expected face-centred cubic Pd structure, confirmed by the crystallinity of PdNPCs. A high-resolution TEM (HRTEM) image taken from an individual PdNPC elaborates a well-defined lattice spacing of 0.2011 nm (Figure 4d), which corresponds to the (200) lattice plane of palladium PdNPCs.

(a) low- and (b) high-magnification TEM images of PdNPCs; (c) SAED pattern of PdNPCs; (d) HRTEM image of PdNPCs.
3.3 Fabrication of rhodium nanoparticle chains (RhNPCs)
Different morphologies of RhNPCs are shown in Figure 5. The RhNPCs grow on the inner surface of HEWL fibrils (Figure 5a), which were heated for 96 h. The rhodium RhNPCs mainly germinated along a thread, as displayed in the inset of Figure 5a. However, when superabundant rhodium nanoparticles (RhNPs) bound to the HEWL fibrils, they overgrew the HEWL fibrils (Figure 5b), which can be clearly observed in the inset of Figure 5b. The fibrosis time was adjusted for 72 h to control the morphology of RhNPCs (Figure 5c). The rhodium RhNPCs became finer and more spindly compared with that in Figure 5b. Figure 5d shows the selected area electron diffraction pattern of the RhNPCs, which signifies the polycrystalline nature of the RhNPCs with the spacings corresponding to the (111), (200), (220) and (311) planes.
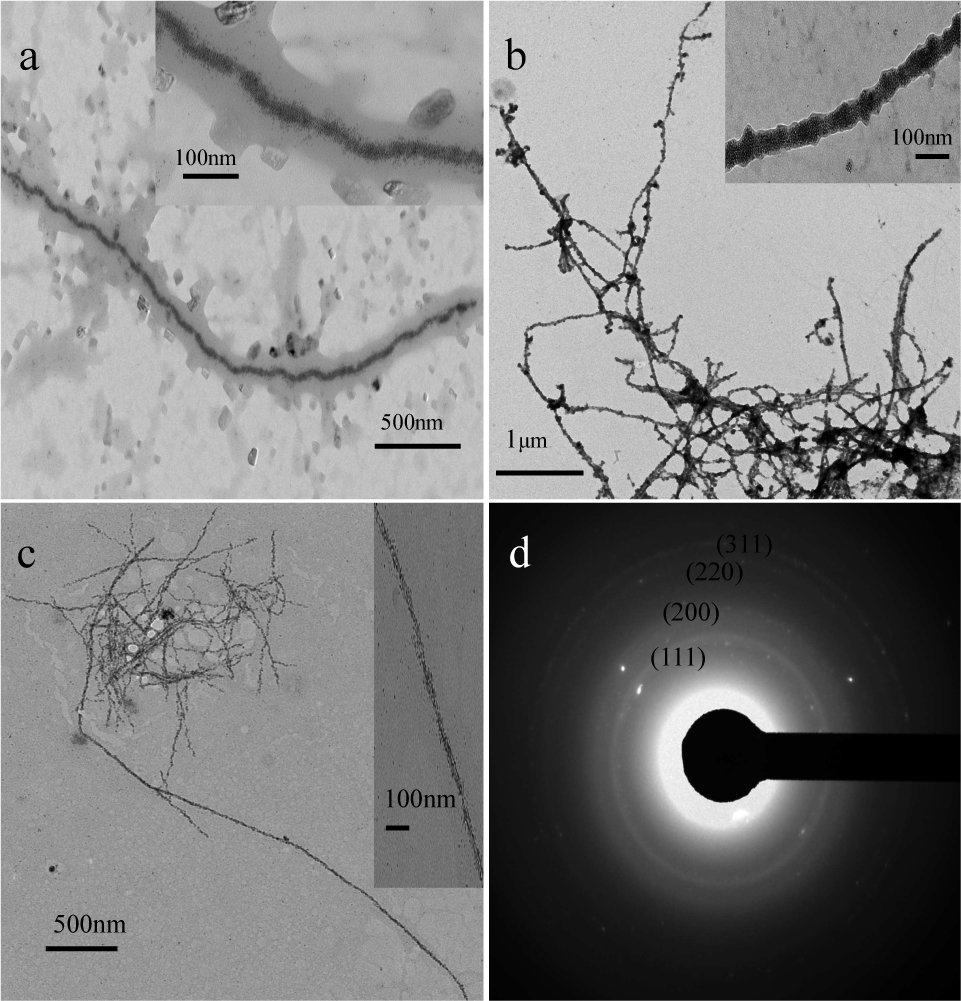
TEM images of (a) low-magnification RhNPCs grown on the inner surface of HEWL fibrils; (b) low-magnification RhNPCs with more RhNPs; (c) low-magnification RhNPCs with different experiment conditions; (d) SAED pattern of RhNPCs; (inset) high-magnification of counterpart RhNPCs.
The chemical composition of RhNPCs was determined by energy-dispersive X-ray spectroscopy (EDS), the result of which is shown in Figure 6. The EDS analysis shows that the sample contains Rh element with Cu and C signal peaks coming from the TEM grid. The presence of oxygen, nitrogen and sulphur should be attributed to the HEWL fibrils. The aluminium and chlorine peaks came from the aluminium sheet of TEM and hydrochloric acid, respectively.

EDS spectrum of RhNPCs
3.4 Fabrication of platinum nanoparticle chains (PtNPCs)
Similar procedures were followed to prepare PtNPCs, which are characterized by transmission electron microscopy and atomic force microscope, as shown in Figure 7. Figures 7a and b show the TEM images of PtNPCs at different growing periods. The length of the PtNPCs was apparently hundreds of nanometres in Figure 7a and it was immature. The length of the mature PtNPCs in Figure 7b was unable to be calculated owing to the reticular morphology. [PtCl6]2− nucleates along fibres and grows into chains after it is reduced. Initially, immature PtNPCs were mainlines, which absorb redundant platinum nanoparticles when they continue to grow. The AFE image in Figure 7c shows that the longest immature PtNPC was about 2 μm. The height of immature PtNPCs was 4.476 nm (Figure 7d). The SAED pattern of PtNPCs in Figure 7f was accordant with the (111), (200), (220) and (311) planes, which signified the polycrystalline structure of PtNPCs.

(a, b) TEM images of PtNPCs at different growing periods; (c) AFE image of PtNPCs; (d) the height of PtNPCs subjected to sectioning in positions in c; (e) 3D AFM image of PtNPCs; (f) SAED pattern of RhNPCs.
In the above discussion, we have demonstrated the wirelike assembled structures of gold, palladium, rhodium and platinum nanoparticles using lysozyme-templated amyloid fibrils. The formation mechanism of these four noble metals would be similar. Here, we give our best guess to elaborate the possible detailed mechanism of them based on HEWL as a template, hoping it would guide directions for future research. The 3D structure of a HEWL fibril is a helix of one or more pairs of protofilaments winding around a hollow core, which seems common to most and possibly all amyloid fibrils [20]. When HEWL fibrils co-incubated with the metal solutions, the charged metal ions could adsorb to the fibrils and form the HEWL-metal ion complexes through weak electrostatic interaction. After subsequently adding the NaBH4, the complexes are reduced to metal nuclei and, with time, the preformed metal seeds grow to form bigger metal nanoparticles on the HEWL fibrils, namely noble metal nanoparticle chains based on HEWL fibrils. Therefore, the presence of both HEWL and weak electrostatic interaction are crucial for the formation of metal NPCs. However, this weak electrostatic interaction between metal ions and HEWL fibrils varies with the type of metal. Moreover, when binding to the HEWL fibrils the metal ions are also affected by the fibrils' group space steric effect, which varies according to the fibril's maturity. Considering these aspects, we conducted the experiment of differentiation for synthesis of AuNPCs, PdNPCs, RhNPCs and PtNPCs above. We believe that most of the metal NPCs with varying morphologies can be synthesized by using HEWL fibrils at different growing stages.
4. Conclusion
In summary, we exhibited a simple route to yield gold nanoparticle chains (AuNPCs), palladium nanoparticle chains (PdNPCs), rhodium nanoparticle chains (RhNPCs) and platinum nanoparticle chains (PtNPCs) based on HEWL fibrils. Through changing the experimental conditions, fibrosis time and temperature in the final stage, the AuNPCs with two types of morphologies were fabricated. Their visible spectra and fluorescence were then discussed. Abundant PdNPCs with length of several micrometres intertwisted with each other to form PdNPC networks. Various morphologies of RhNPCs were also produced by adjusting the fibrosis time. To determine the elements of RhNPCs, energy-dispersive X-ray spectroscopy (EDS) of the sample was carried out. Finally, PtNPCs at different growing periods were presented. It is well known that one-dimensional AuNPCs, PdNPCs, RhNPCs and PtNPCs have many potential applications in the fields of catalysis, sensor, optical and so on. Furthermore, by combining protein engineering with inorganic nanostructures synthesis, this development will benefit the design and assembly of more multifunctional bio-nanomaterials.
Footnotes
5. Acknowledgements
This work was supported by the National Natural Science Foundation of China (Grants 21371149, 21101134) and Research Fund for the Doctoral Programme of Higher Education of China (Grant 20131333110010) and the Natural Science Foundation of Hebei (Grant 14961107D).
