Abstract
A series of cross-linked, pH responsive vesicles with tunable diameters were prepared by self-assembly of poly (ethyleneoxide)-b-poly [2-(diethylamino) ethyl methacrylate-co-glycidyl methacrylate, PEO-b-P(DEA-co-GMA)] in a THF/water mixture. The copolymers were synthesized by the random copolymerization of DEA and GMA, initiated by PEO-Br. With an epoxy group and a tertiary amine in its structure, self-catalysed cross-linking was triggered easily by mild heating. The vesicles showed pH responsiveness as characterized by DLS, and tunable permeability can be understood accordingly. As the pH of the solvent changed from 9.8 to 7.4, the diameters of the vesicles increased. It was observed that the diameters at pH = 6.0 were comparable to those at pH = 7.4. This alteration may have occurred because of the protonation of the PDEA segment. This particular property has great potential and many applications, since most drug-loading and controlled-release studies are conducted within this pH range.
1. Introduction
Block copolymers can self-assemble into a variety of morphologies, both in solvent and in bulk [1-10]. The vesicles and micelles prepared from the self-assembly of amphiphilic block copolymers are widely used as drug loading and delivery vehicles and have attracted much attention in the past decade [11-20].
As the most popular candidate for drug loading and delivery agent with excellent biocompatibility, poly(2-(diethylamino)ethyl methacrylate) (PDEA) with pH-stimuli responsiveness has attracted much attention [21-23]. However, the PDEA segment becomes protonated and soluble in an aqueous medium when its pH remains less than6.5, making it unfit for application as a hydrophobic block and as a drug-loading agent alone. To consider the vesicles as potential drug carriers and nanoreactors, the tunable permeability of membranes is greatly preferred. Using stimuli-responsive block copolymers as the building blocks of vesicles is one elegant way to modify the permeability. Later on, it becomes a simple matter to tune the releasing properties of the vesicles. In the past decade, the development of intelligent vesicles responding to pH, temperature, redox and light became one of the most important topics in the vesicle research. However, the stimulus often leads to the dissolving and disruption of the particles [14]. To avoid disruption, keeping the morphologies of the self-assemblies under stimulus and the cross-linking of vesicle membranes or micelle cores is necessary, although it was observed that polystyrene (PS) can also fix the morphological issues related to the self-assembly under proper stimulation in order to prepare breathing vesicles [24]. Previous research indicated that the dynamically stable morphology was attained as a new condition when the sample was appropriately heated [25]. Based on the gelation reaction of poly(3-(trimethoxysilyl)propylmethacrylate) (PTMPM), Du et al. produced pH-responsive stable vesicles by incorporating pH-sensitive PDEA into the PTMPM [26]. Peng et al. prepared thermo-responsive organic-inorganic hybrid vesicles from PEO-b-poly(N-Isopropyl acrylamide-r-TMPM) (NIPAM-r-TMPM)PEO-b-P (NIPAM-r-TMPM) [11]. Du et al. reported core cross-linked vesicles via UV exposure from 7-Hydroxy-4-methylcoumarin-based monomers [27].
As a versatile cross-linkable polymer, PGMA has been used in both cross-linking the morphologies of the self-assemblies [28, 29] and preparation of polymer brushes [10, 30]. In this paper, vesicles with cross-linked membrane were prepared from an amphiphilic block copolymer of PEO-b-P(DEA-co-GMA), as shown in Scheme 1. With both epoxy groups and a tertiary amine on the hydrophobic block, self-catalysed cross-linking was triggered by mild heating following Scheme 2. The vesicles showed pH responsiveness while keeping the morphologies of the vesicles intact. Moreover, the permeability of the vesicle membrane was tuned by tuning the molar ratio of the cross-linkable GMA unit and pH.

Schematic illustration for preparation of POE-b-P(DEA-co-GMA)

Self-triggered cross-linking of P(DEA-co-GMA) at the vesicle's membrane
2. Experimental Section
2.1 Materials
Poly (ethylene glycol) methyl ether(PEO; Mn ca. 2000 Da; Mw/Mn = 1.06) and 2-(diethylamino)ethyl methacrylate (DEA)were purchased from Maya Chemical Co. Glycidyl methacrylate (GMA) was supplied by TCI. PEO was dried under a vacuum overnight before being used. 2-Bromoisobutyryl Bromide and 2, 2′-Bipyridine (bpy; 99%) were obtained from Aladdin Industrial Co. DEA and GMA were passed through a column filled with basic alumina to remove the inhibitors. CuBr was washed with acetic acid and ethanol before use. Other chemicals and a reagent were purchased from Kermel Chemicals and used as received.
2.2 Preparation of PEO-b-P(DEA-co-GMA) by ATRP
Initially, the PEO-Br macroinitiator was prepared via the reaction of PEO with excess 2-bromoisobutyryl bromide according to the literature report [31], and was confirmed by 1H NMR. Later, the PEO-Br was used as a macroinitiator to prepare the block copolymer as illustrated below, and the reaction mechanism is shown in Scheme 1. The PEO-Br macroinitiator (0.20 g, 0.1mmol), PMDETA(21 uL, 0.1 mmol), DEA (2.22g, 12.0mmol), GMA (0.43 g, 3.0 mmol) were dissolved in 3 mL dioxane in a 25 mL reaction tube, before the tube was deoxidized via three freeze-evacuatethaw cycles. In the next stage of the experiment, 14.4 mg (0.1 mmol) CuBr was added to the flask in a solid state and the flask was deoxidized again. The flask was immersed in an oil bath at 50°C under continuous stirring. The reaction was polymerized for 3h and terminated by cooling to room temperature and exposure to air. The polymer solution was diluted with DCM and passed through a column filled with neutral alumina to remove the CuBr. The block copolymer was precipitated in petroleum ether three times and lyophilized in dioxane. The composition of the block copolymer was calculated by comparing the peak areas on 1H NMR, and the conversion of monomers was almost 100%.
2.3 Preparation of vesicles and the cross-linking of the membranes
The general procedure for the self-assembly of PEO45-b-P(DEA-co-GMA) and cross-linking is as follows. First, 10 mg of copolymer was dissolved in 2 mL THF in order to obtain a 5 mg/mL solution in a vial; then deionized water was added dropwise under continuous stirring until the water-volume ratio reached 80%. After stirring for 12h, 10 mg of ethylene diamine was added to the vesicle solution and it was stirred for another 12h in order to cross-link the membranes of the vesicles.
The hydrodynamic diameter and size distribution of the cross-linked membranes of vesicles prepared from PEO45-b-P(DEA-co-GMA) at various pHs were evaluated by dynamic light scattering (DLS). The pH responsiveness of the cross-linked vesicles was characterized by comparing the hydrodynamic diameter of the vesicles in different pHs along with aqueous dispersions by DLS. At the beginning, the THF was removed by dialyzing the vesicles against water, and then the vesicles were diluted into 0.1 mg/mL. The dynamic diameter of the vesicles at various pHs was characterized by DLS.
3. Results and Discussions
3.1 Synthesis of self-catalysed cross-linkable block copolymerPEO45-b-P(DEA-co-GMA)
During this study, the PEO45-Br was synthesized through the reaction of hydroxyl terminated PEO45 with an excess of 2-bromoisobutyryl bromide and confirmed by 1H NMR, as shown in Figure 1. By comparing the peak area of (a) (3.38 ppm) derived from PEO45 and (d) (1.94 ppm) derived from 2-bromoisobutyryl bromide, it was confirmed that all hydroxyl groups were consumed. The structure of PEO45-Br was also confirmed by FT-IR as shown in Figure 2. A peak appeared at 1730 cm−1, which represented the formation of the ester group; simultaneously, no absorbance between 3200−1 and 3700 cm−1 was detected, which confirmed that all hydroxyl groups were consumed.

1H NMR spectra of PEO-Br and PEO-b-P(DEA118-co-GMA33)
In the next stage, PEO45-Br was used as a macroinitiator to synthesize the PEO45-b-P(DEA-co-GMA) block copolymer through ATRP, as it is a good mediator of the controlled radical polymerization of DEA and GMA. After being purified by precipitation and dried under a vacuum, the structure of the block copolymer was characterized by 1H NMR and FT-IR. The 1H NMR of PEO45-b-P(DEA118-co-GMA33)as an example is shown in Figure 2 (top). The block ratio of PDEA and PGMA can be determined by comparing peak areas of (b) (3.76 ppm) with (e) (4.01 ppm) and (j) (3.20 ppm). A series of PEO45-b-P (DEA-co-GMA) block copolymers with different PDEA to PGMA ratios were synthesized in this work and their structural parameters are listed in Table 1. The structure of the block copolymer was also characterized by FT-IR, as shown in Figure 2 (bottom). The absorbance of carbonyl group at 1730 cm−1 increased, which indicated the polymerization of methyl acrylate. The wide absorbance at 3110 cm−1–2770 cm−1 was derived from C-H stretching on both PDEA and PGMA. At the same time, a small absorbance appeared at 907 cm−1, which derived from the epoxy groups of the block copolymer.
PEO45-b-P(DEA-co-GMA) block copolymers synthesized in this research and their characteristics

determined by 1H NMR
characterized by DLS at pH=7.4
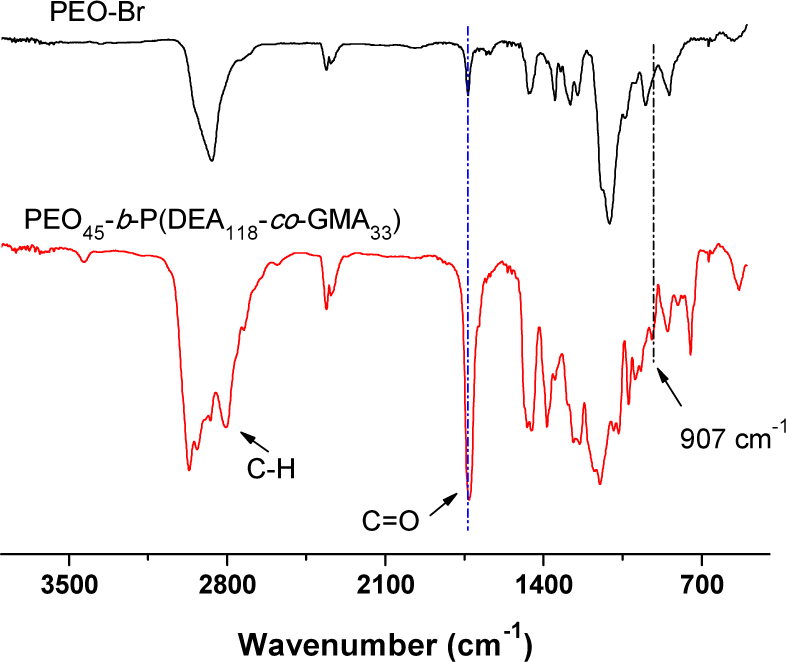
FT-IR spectra of PEO45-Br and PEO45-b-P(DEA118-co-GMA33)
The GPC characterizations were performed in order to determine the PDI of the block copolymer and characterize the stability of the block copolymer during ATRP. The GPC evolutions of PEO45-Br and PEO45-b-P(DEA118-co-GMA33) are illustrated in Figure 3 as an example. After ATRP, the peak moved to a lower elution time, indicated the increasing of molecular weight. More importantly, the GPC curve of PEO45-b-P(DEA118-co-GMA33) was also symmetrical, confirming the fact that the epoxy ring was stable during polymerization, although self-triggered cross-linking was noticed in the bulk state at 40 °C. This ensured good control of the block copolymer preparation and drug loading, and provided possible cross-linking without the addition of the cross-linker of amine.

GPC evolution of PEO45-Br and PEO45-b-P(DEA118-co-GMA33)
3.2 Self-assembly of PEO-b-P(DEA-co-GMA) copolymers ina THF/water solvent mixture
The self-assembly processes were conducted at room temperature and deionized water was added to the solution until the volume ratio of water reached 80%. After stirring for 24 hours, the ethylene diamine was added to cross-link the PGMA domains of the self-assemblies. The morphologies of the self-assemblies were observed via TEM after cross-linking. The TEM images of the vesicles prepared from PEO45-b-P(DEA133-co-GMA16)and PEO45-b-P(DEA105-co-GMA48)are shown in Figure 4. As is shown in Figure 4, the average diameter of the vesicles prepared from PEO45-b-P(DEA133-co-GMA16) is 110 nm, and the average diameter of vesicles prepared from PEO45-b-P(DEA105-co-GMA48) is 119 nm, values comparable to those of PEO45-b-P(DEA133-co-GMA16), possibly due to their similar hydrophilic/hydrophobic ratios.
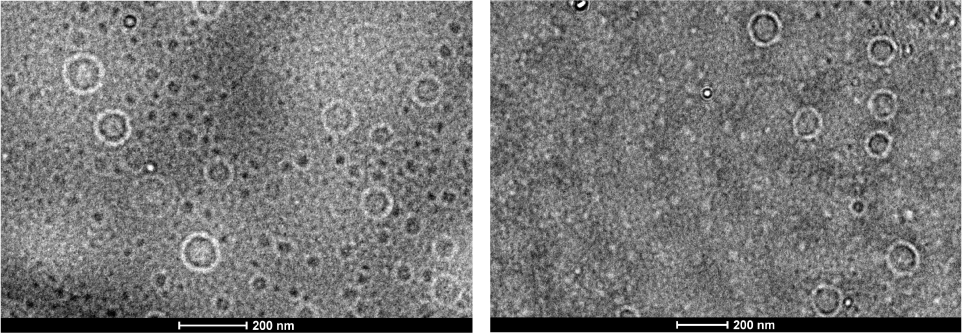
TEM images of vesicles self-assembled from (left) PEO45-b-P(DEA133-co-GMA16)and (right) PEO45-b-P(DEA105-co-GMA48)
To characterize the pH responsiveness of the vesicles, the hydrodynamic diameters dependent on the PGMA ratio were determined via DLS using various pH values in an aqueous dispersion. As shown in Figure 5, the diameter of the vesicles prepared from PEO45-b-P(DEA105-co-GMA48) is little larger than but comparable to TEM observations (166 nm at pH = 7.4). On the other hand, the diameters of the other two samples differed significantly; the diameter of the vesicles from PEO45-b-P(DEA118-co-GMA33) was found to be 314 nm at pH = 7.4, and the diameter of PEO45-b-P(DEA133-co-GMA16) vesicles was 352 nm at the same pH. The reason for this larger hydrodynamic was due to the hydrophilic properties of PDEA in the vesicle's membrane. Moreover, the diameters of the vesicles showed typical pH responsiveness in the pH range of 4.0–9.8. All three samples showed similar trends in terms of the variation of pH, as shown in Figure 6. At pH 9.8, the PDEA sample was insoluble in water and resulted in a smaller diameter. However, the vesicles with higher PDEA ratios showed much larger diameters, indicating that PDEA is more hydrophilic than the PGMA segment. With a decreased pH of 7.4, the diameters of the vesicles increased due to the increased hydrophilicity of the PDEA segment. The PEO45-b-P(DEA105-co-GMA48) vesicles' diameters increased by around 15% when the pH was changed from 9.8 to 7.4, whereas the PEO45-b-P(DEA118-co-GMA33) and PEO45-b-P(DEA133-co-GMA16)vesicles' diameters increased by38% and 44%, respectively, because of their higher PDEA ratios and lower cross-linking densities. When the pH changed further to 6.0, the diameters of all three vesicles were found to be almost intact.
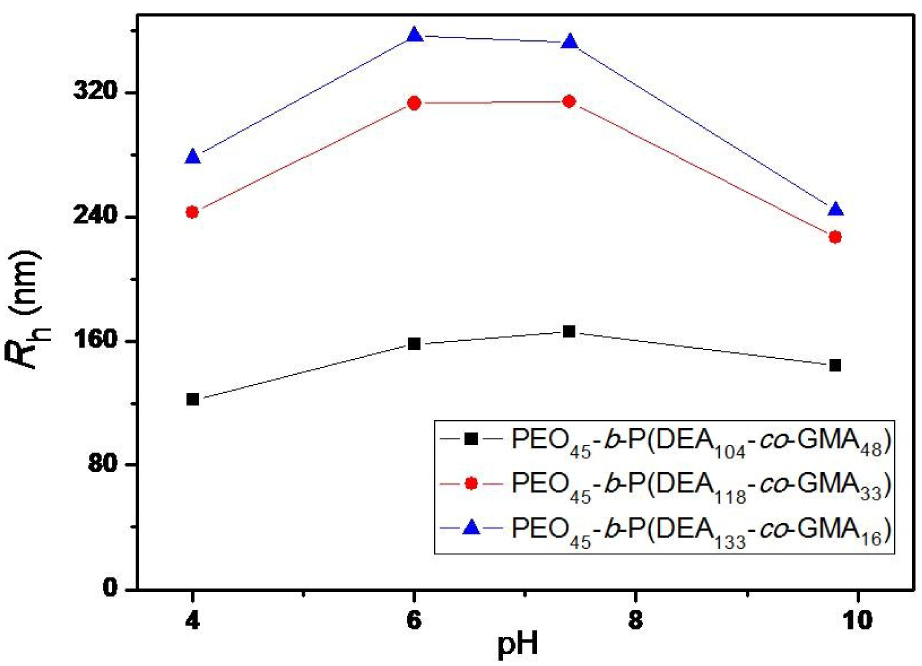
pH-responsive vesicles self-assembled from PEO45-b-P(DEA-co-GMA) with different DEA/GMA ratios

DLS curves of vesicles from the self-assembly of PEO45-b-P(DEA118-co-GMA33) at various pHs
The comparable vesicle diameters at a pH between 7.4 and 6.0 have important practical applications, since most drug delivery is conducted within this pH range.
Interestingly, a further decrease in pH to 4.0 resulted in an obvious reduction in the vesicles' diameter, rather than an incremental reduction, which is evident in Figure 6. The possible reason for this may be the alteration in the solubility of PDEA in water as a result of protonation at a low pH. It was observed that a reduction of the pH value to 4.0 allowed part of the PDEA segment belonging to the vesicle membranes to dissolve in water. In parallel, it was observed that the intensity of the dissolved PDEA segments was very low when compared to that of the cross-linked vesicle membranes. As a result, the diameters of the vesicles decreased because the hydrodynamic diameter of the vesicles did not take the dissolved polymer segment into account.
At the same time, the vesicles did not disaggregate, which proved that the PGMA segments had cross-linked the vesicle membranes successfully. The DLS curves of PEO45-b-P (DEA118-co-GMA33) vesicles at various pHs are shown in Figure 6 as an example. These kinds of responsive vesicles with pH-responsiveness have great advantages in anti-cancer drug loading and delivery, since the pH of a tumour is lower than that of other parts of the human body. However, the DOX deteriorated during the drug loading and release process; the most likely reason may be the reaction of the epoxy ring of the PGMA segment and the amino group on the DOX. Intensive study of the drug loading and release is already ongoing.
4. Conclusion
A series of amphiphilic block copolymers with pH responsiveness were synthesized via ATRP, and the self-assembly of these block copolymers was conducted in THF/H2O. The hydrophobic block of the copolymer was composed from cross-linkable PGMA and pH-responsive PDEA. The obtained results suggest that the block copolymers self-assembled into vesicles with easily regulated diameters by tuning the pH of the solvent and maintaining the stable morphologies of the vesicles. These kinds of polymer self-assemblies have great potential in the application of anti-cancer drug loading and delivery, and further study is underway.
Footnotes
5. Acknowledgements
This research was kindly supported by the Natural Science Foundation of Hebei (B2015201085); the Scientific Research Foundation for the Returned Overseas Chinese Scholars, State Education Ministry; The Plan of Science Technology Research and Development of Hebei Province (Grant No. 13211219) and Hebei University (2013–272).
