Abstract
Commercial surgical robots have been in clinical use since the mid-1990s, supporting surgeons in various tasks. In the past decades, many systems emerged as research platforms, and a few entered the global market. This paper summarizes the currently available surgical systems and research directions in the broader field of surgical robotics. The widely deployed teleoperated manipulators aim to enhance human cognitive and physical skills and provide smart tools for surgeons, while image-guided robotics focus on surpassing human limitations by introducing automated targeting and treatment delivery methods. Both concepts are discussed based on prototypes and commercial systems. Through concrete examples the possible future development paths of surgical robots are illustrated. While research efforts are taking different approaches to improve the capacity of such systems, the aim of this survey is to assess their maturity from the commercialization point of view.
Keywords
1. Introduction
The idea of telerobotic healthcare was most probably born in the early 1970s, proposed in a study for the National Aeronautics and Space Administration (NASA) to provide surgical care for astronauts with remote controlled robots [1]. The concept in Fig. 1 presents very well the fundamental characteristics of a complete teleoperational system, although the first prototypes were only built more than 15 years later.

NASA had elaborated advanced telerobotic concepts for in-space use in the early 1970s, well before the first military or commercial systems were developed (Image credit: NASA [1])
The era of surgical robotics—as they are available today—began by the mid-1980s. By that time dozens of medical robotic research projects have been launched, leading to the birth of instruments for remote and local robotic surgery. The idea of robotic Minimally Invasive Surgery (MIS) was conceived on the principle of employing technology in extending the surgeon's dexterity and capabilities. With the introduction of the first robotic devices in the 1990s (see subsection 4.1), Computer-Integrated Surgery (CIS) and telemedicine have become widely used around the world, with an estimated present total number of 5000 devices [2]. Surgeons and engineers created systems and networks for advanced patient care, and demonstrated their capabilities over a hundred different types of procedures. Extreme setups were also arranged, such as trans-continental surgery [3, 4], underwater trials [5], and even procedures in weightlessness [4, 6, 7].
Surgical robots form a subgroup of medical robotic devices, distinct from hospital care or rehabilitation robotics. The Medical Robotic Database (MeRoDa) [8] lists over 450 international surgical robotic projects; however, many of those did not result in more than a few scientific publications. This paper gives a comprehensive list of the most important devices, including commercial products and advanced prototypes, identified through a thorough literature review and a detailed survey of the internet. It is believed that this is the most complete enumeration currently available.
1.1 Surgical robots and robotic systems
Arguably, it is a challenging task to delimit the domain of surgical robotics. While the term ‘robotic surgery’ is widely used by the clinical community, there is a strong disagreement on what systems are to be called a ‘robot’. According to the latest ISO standard (ISO 8373:2012 Robots and robotic devices—Vocabulary), a ‘robot is an actuated mechanism programmable in two or more axes with a degree of autonomy, moving within its environment, to perform intended tasks.’ However, since key determining parts of the taxonomy, such as ‘Degree of Autonomy’, are not defined in the standard, it is up to the community to set the border between teleoperated systems and robotic devices. Since many groups of systems are usually referred to as robots, it will be used in a more generic sense.
Furthermore, based on the current standards, we can conclude that any system with a medical intended use should be seen as a medical device. In the case of robots, this includes all kinds of systems from psychological rehabilitation to natural orifice surgery [9]. The diversity of functions and appearance make the regulation and standardization of the domain extremely difficult.
This review mostly follows the practical definitions of the community, approaching the taxonomy problem from the application point of view. See section 2 for further details of the surveyed field.
1.2 Fundamental approaches in robotic surgery
There are two distinct concepts along which surgical robots are being developed:
Continuously controlled by a human operator on site or remotely connected.
A surgical plan is executed by the robot, typically guided by a navigation system, or other tracking method.
The most successful surgical robot, the da Vinci Surgical System (Intuitive Surgical Inc., Sunnyvale, CA)—following NASA's and other early concepts—only relies on the surgeon's direct input through the master controller, and therefore the liability for all actions stays with the human operator. The overall performance of the system is limited by the resolution of human fine-motion, reaction-time and cognitive skills. Many systems in orthopaedics and neurosurgery primarily chose to employ image guidance based on the industrial Computer-Aided Design and Manufacturing (CAD/CAM) paradigm, where the surgical plans and executable tasks are defined accurately on a pre-operative image (e.g., Computer Tomography (CT), Magnetic Resonance Imaging (MRI)) of the patient [10, 11]. This results in superior precision; however, these systems are less fault-tolerant, are typically not adaptive, and additional safety mechanisms are always required to protect the patient in the case of an adverse event [11].
Naturally, the mixture of the two concepts is also possible, where sensors are providing real-time updates to the robot regarding the environment, or where the robots can autonomously perform certain sub-tasks. Numerous systems are currently in the research-prototype phase (see Table 4). Fig. 2 presents the two most well-known examples of master–slave teleoperated and image-guided robots, and how their design evolved over time 1 . In the upper row, the da Vinci system (detailed description in subsection 3.1) is shown as an example of a teleoperated system, while the bottom row shows the ROBODOC (q.v. subsubsection 3.3.1) system, as an example of automated Image-Guided Surgery (IGS) systems.
Commercially available systems

Discontinued systems

Upcoming systems for commercial exploitation

Advanced research prototypes

Surgeon's operating force-feedback interface Eindhoven
Single-Port lapaRoscopy blmaNual roboT
Array of Robots Augmenting the KiNematics of Endoluminal Surgery

The two most famous surgical robots, Intuitive Surgical's da Vinci (top) and Curexo's ROBODOC (bottom) shown in their first commercial format (around 2000), current outfit and provisional future design. (Image credit: Intuitive Surgical Inc. (top row), ROBODOC, a Curexo Technology Company (bottom row)).
1.3 Current surgical robotic systems
The majority of the currently available systems in the field of surgical robotics and computer-assisted surgery are designed to imitate, copy and improve human capabilities. Systems aim for an enhancement of the skills of the surgeon to conduct medical procedures on the same scale, therefore not changing the existing paradigms but rather supporting them. However, the commercially available technology is still an early generation of robots. Therefore, they have their natural disadvantages, which are addressed by current research efforts.
This paper also deals with the next generation of robots, gradually becoming cognitive systems that have access to a knowledge base and are capable of adaptation. This would allow them to conduct certain parts of procedures automatically. Even in complex cases the systems could analyse the situation and provide a second opinion for the physician.
1.4 Methodology
This overview aims to help professionals on the field by gathering systems on a global scale, and providing an unbiased overview. Furthermore, the summary below and the detailed appendix provide a basic comparison of systems. For ease of understanding, the robot list is divided into four sections:
Systems approved for clinical use in at least one country.
Systems used to be commercially available in at least one country.
The research entity has achieved good progress in the development and announced the aim to acquire clearance from, e.g., the U.S. Food and Drug Administration (FDA). This is considered as a key act towards commercialization.
The project has left the first demonstrating stage and recent research progress has been published.
The survey in each of the categories is organized as a tabular comparison of the devices, and contains key facts announced. Together with the tables, a description of their content is given in section 10.
Medical implications and clinical usage of systems are not evaluated in this paper. The review focuses on the engineering and market aspects of surgical robots.
The survey starts with a discussion about the commercially available systems in section 3. This first overview of the market examines each robot in detail. If a system forms the foundation of future development projects, it might be presented in a later section.
2. Definition and differentiation of the surveyed field
Robotic surgery is defined by the Society of American Gastrointestinal and Endoscopic Surgeons and Minimally Invasive Robotic Association (SAGES–MIRA) Robotic Consensus Group as:
‘a surgical procedure or technology that adds a computer-technology-enhanced device to the interaction between surgeon and the patient during a surgical operation, and assumes some degree of freedom of control heretofore completely reserved for the surgeon. This definition encompasses micro-manipulators, remotely controlled endoscopes and console-manipulator devices. The key elements are enhancement of the surgeon's abilities—by vision, tissue manipulation or tissue sensing—and alteration of the traditional direct local contact between surgeon and patient.’[12]
Therefore, robotic surgery is usually an advancement of Minimally Invasive Surgery (MIS). Here, the tools of the surgeon are passed via small incisions (0.5–3 cm), e.g., into the abdominal cavity. This reduces the trauma and operation risk, but also requires a highly trained surgeon [13], since the orientation is quite challenging. Moreover, certain devices require the crossing of the tools, which increases the cognitive load even further (e.g., Covidien's Roticulator). Most teleoperated surgical robots are reducing this load, such as the da Vinci (see subsection 3.1), which fits the classical definition of a robot according to subsection 1.1, but is unable to perform any actions without the instructions of a surgeon. However, the research prototype version of the da Vinci allows unconstrained automated motion [14].
This review covers medical devices in a broader sense: systems supporting the physicians in the treatment of patients with a mechanical tool and computer assistance.
To further enhance such systems the use of artificial intelligence is considered, introducing cognitive surgical robotics which can be defined as:
‘A system that extends the capabilities of any surgical robot by cognitive functions. Today, these features may be very simple, but already extending human capabilities in certain ways. It is expected that future cognitive surgical robots will have access to ontology-based workflows of the surgical procedure, will be able to manage and control the workflows, and will be able to estimate the mental state of human collaborators, including that of the surgeons and the assistants. Cognitive robots could also provide information to the humans in various forms, including dialogue systems and augmented reality displays, quickly search large backing databases, for example based reasoning, and can recommend solutions based on fast risk estimations. It is also expected that such capabilities require different models, such as the model of the operating room, the model of the human body and good estimations of surgeons' and assistants’ behaviour. (András Lőrincz, 2013)
As stated in subsection 3.9, none of the currently available systems uses computer cognition or artificial intelligence to support the surgeon. The integration of those two software components is one of the major tasks for current and future research (q.v section 6 and 7).
3. Commercially available robotic systems
The initial development of robot-assisted surgery was driven by the aim to improve the efficacy of laparoscopic surgery.
The key features of the commercial systems are listed in Table 1. For completeness each of the commercial systems is introduced within the following subsections. Here, systems are grouped by their main target application 2 .
3.1 Master–slave telemanipulator systems
The da Vinci follows strictly the master–slave approach, as introduced in section 1 [10, 15], and is a direct successor of the project of that time [16, 17]. Figure 3 shows an example setup of the Si version of the robot family. It offers four arms for tool handling (including the camera) and dual console control for cooperative surgery or training. Their latest development iteration was the Xi version, offering an increased working space of the arms, more flexible tool positioning and an enhanced vision system.

An example setup of the da Vinci Si system. (Image credit: Intuitive Surgical Inc.)
The da Vinci is the only (as of March 2015) available system on the market for general MIS. Their domination on the market allowed Intuitive to sell more than 3200 units worldwide [18], despite a price of more than $1.5M [18].
But even with progress in the development of the robot, it still lacks the support of automated procedures and consumes a lot of space around the operating room (OR) table [19].
Only the da Vinci system has seen enough procedures to allow fine statistics regarding adverse events. In the recent years, much attention has been paid to FDA's Manufacturer and User Facility Device Experience (MAUDE 3 ), where all users are obliged to enter the details of every abnormal case, and the manufacturer must address these [20]. While the da Vinci proved to be a solid robot platform, naturally, there have been certain issues over the three million procedures performed so far [21]. Software-related problems have been reported in far less than 1% of the cases, and are typically solved by rebooting the system. Mechanical failure has been reported in 0.2–0.5% of the cases [22]. Altogether, including the medical complications, 0.9–4.7% have been measured (based on 2500 procedure), and the conversion ratio was 0.4–0.5% [23].
3.2 Computer-assisted radio-surgery
The general idea of radio-surgery is based on the lethal effect of high radiation doses to living cells. To prevent harmful doses for healthy tissues, the beams are focused within the tumour to be treated. The different approaches to the radiation treatment of tumours are listed in the following sections. All have in common that they require a pre-treatment scan of the targeted areas. This is required to plan the irradiation and dosage precisely.
3.2.1 CyberKnife
The CyberKnife (Fig. 4(a)) is composed of a six degree of freedom (DOF) industrial KUKA robot carrying a linear accelerator (LINAC) as the radiation source. Depending on the model chosen, the patient's bed has up to six additional DOF. The patient is placed on the actuated bed with markers on his/her bodies, and thus their movements can be tracked by the system. This allows fine adaptation to patient motion, e.g., to compensate breathing motion [24, p. 31].

Commercial systems in use for radio-therapy
CyberKnife is one of the few systems that have been thoroughly investigated regarding their inherent and application accuracy. The industrial robot base helps to target the tumour with 0.2 mm precision and the overall treatment precision is better than 0.95 mm [24].
3.2.2 Novalis
In the case of the Novalis system, the radiation source is constructed in an L-shape, and rotatable along a horizontal axis. Fig. 4(b) shows an example setup of the system, where the patient is placed on the articulated OR table. During the treatment, the radiation source rotates around the patient [25].
3.2.3 Gamma Knife
The Gamma Knife system shown in Fig. 4(c) has a different way of delivering the radiation source: it is basically a half sphere, which houses the fixed radiation source and moveable focusing mechanism. The patient's head is fixed by a stereotactic frame, and placed on a moveable bed in the focal centre of the sphere. Through the movement of the bed and the adjustment of the source, tumours can be targeted very precisely [24]. The architecture of the system only allows radiation treatment of the upper body, such as head, neck and shoulder.
3.3 Orthopaedic applications
3.3.1 ROBODOC
The first generation of ROBODOC was based on a five DOF IBM SCARA robot, which was holding a milling tool at the tip. This setup is shown in Fig. 2 in the bottom left. The robot's task is to prepare/sculpture the joint precisely in order to prepare it for implant insertion [10, 11, 15].
The ROBODOC's development began in 1986 at IBM T. J. Watson Center and U.C. Davis, aiming to create a bone milling robot for canine hip surgery. Integrated Surgical Systems Inc. (ISS) 4 took over the concept and designed a system for automated hole drilling in Total Hip Arthroplasty (THA) 5 surgeries. The first-ever completely robotic human trial was performed in 1992 [26, 27], and the system was later extended to Total Knee Artroplasty (TKA) procedures as well [28]. For both applications, the surgeon defines the position and dimension of the required hole, based on pre-operative data. Then s/he registers the robot with respect to the patient in various configurations, achieving a correlation between the coordinate systems [29].
Since 1994 ISS sold around 80 systems across Europe and Asia, and in 2008 it became the first FDA-approved automated bone milling robot, under Curexo Technology Corporation [29]. The Korean Hyundai Heavy Inc. invested into ROBODOC to develop a new generation of the system [30]. In late 2014 the original was put on the market under the brand name Think Surgical [31].
3.3.2 RIO system
The RIO system from MAKO Surgical (bought by Stryker Inc.) is shown in Fig. 5(a) is similar to that of ROBODOC. On a moveable cart, a robotic arm with a milling tool is mounted.
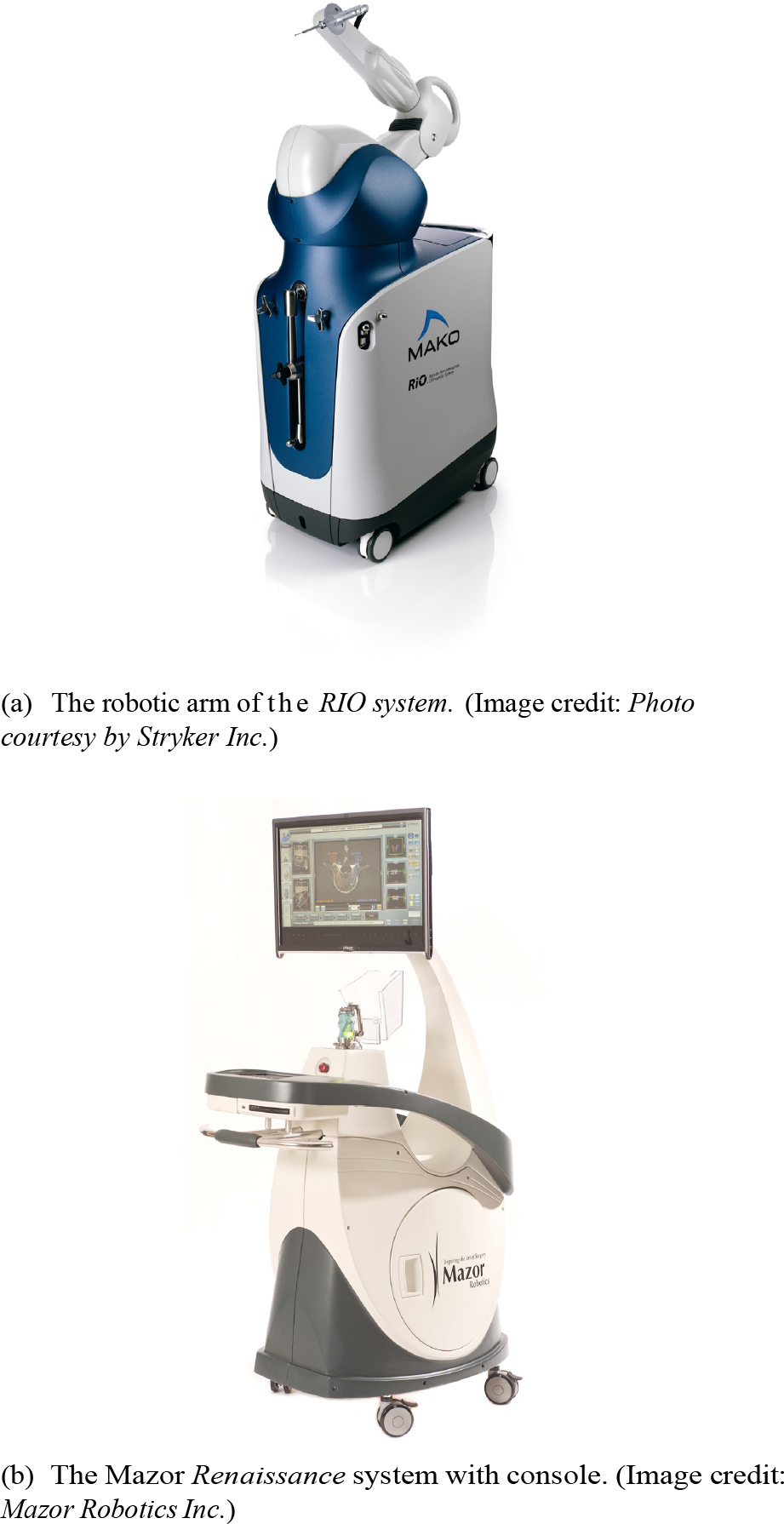
Surgery-support systems in orthopaedic applications
The RIO system aims for the same applications as the ROBODOC, but here the actions are not performed automatically, rather in a computer-guided manner, based on a pre-operative CT scan. The boundaries of the surgery, i.e., the limits (also called Virtual Fixtures) for the sculpturing of the bones are predefined. During the procedure, the surgeon guides the tool, while the system monitors the tip's position. If the tip reaches a limit, the robot generates resistance to prevent breaches of those. This gives the surgeon the control at every position with the benefits of computer-assisted supervision.
3.3.3 Renaissance
The Renaissance system (Mazor Robotics, Israel) is composed of two different parts: a guide rail and a tool positioning robot. The tool guide is mounted directly onto the patient's spine to prevent any relative movement between the robot and patient. For each implant, the positioning robot is placed on the guide rail and automatically moves into the desired position.
The Renaissance was developed for spinal surgery. Here its task is pre-operative planning, based on CT images and tool guidance during the surgery. The surgeon can use the software-generated 3D image of the patient's spine, and select the proper implants for each position. During the surgery, the guide rail is attached to the patient and registered via special markers and X-ray images. The markers are then matched with the pre-operative plan, and with the robot in place the system now can guide the tools onto the right trajectory. Mazor Robotics published a minimum positioning precision of 1.5 mm [32]. The system can also be used for neurosurgical procedures.
3.4 Organ inspection
Due to the length of the intestinal tract, it is not possible to inspect it entirely with traditional endoscopes, and flexible endoscopes also have major limitations. Preferably, swallowable capsules with cameras are used [33]. An example set of those systems is shown in Fig. 6. Each capsule is battery-powered and transmits the collected images wireless to a receiver carried by the patient. The battery lasts for up to 12 hours while the device takes between two and 35 images per second [34, 189–193]. The advancement of the capsule is provided by the natural motility of the bowels. To support the diagnosis, the patient is not allowed to eat or drink before the treatment. This empties the gastrointestinal tract, and allows a clear view of the structures [33]. According to subsection 1.1, these capsules are not robots, but mobile sensor systems. Yet they create the foundation for more complex miniaturised systems, as to be discussed in subsection 7.4.

Camera capsules used for the internal examination of the oesophagus, the small intestine and the large intestine—from left to right. (Image credit: Dr. H.H. Krause, CC-BY-SA 3.0.)
Another way to improve the classical inspection of the abdomen is via flexible, robotised endoscopes. These are highly redundant in their kinematics, and can be controlled through a complex Human–Machine Interface (HMI) [35]. An example system for this approach is the Endotics endoscope. Its single use head can be steered and equipped with a camera, water jets and insufflation outlets [36]. It propels itself forward with inch worm-like movement, and is retracted by pulling the umbilical end [36]. A similar approach is taken by the Invendoscope, that itself consists of two sheaths and is driven by a motor unit outside the patient. One main difference to the Endotics systems is that here, an additional tool can be passed through a channel, e.g., to treat a tumour in-situ [35].
3.5 Catheter operations
MIS catheter operations on the heart are usually conducted by accessing the femoral or brachial artery or vein, and pushing the catheter all the way up to the heart. This is usually achieved with X-ray fluoroscopy to assist the surgeon in the navigation. Due to the passive structure of the catheter, it may be difficult to reach certain parts, and it also lengthens the duration of the surgery [37].
With emerging robotic tools for catheter operations, complex trajectories can be followed. Just like conventional systems, the robotic systems also use X-ray visualization for navigation assistance. Two different approaches have been implemented: robotic push and magnetic pull catheters.
3.5.1 Mechanically steered robots
All three catheter systems in Fig. 7 (from top to bottom: CorPath 200 (Corindus Vascular Robotics, USA), Sensei X (Hansen Medical Inc., USA) and Amigo (Catheter Robotics Inc., USA)) use mechanical controls to steer the tip. Each control unit is placed outside the patient in a custom designed container. The catheter is then pushed through the blood vessel to the targeted region. This requires a flexible, yet stiff sheath, in order not to deviate along the access pass due to the exerted pushing force [38–40].
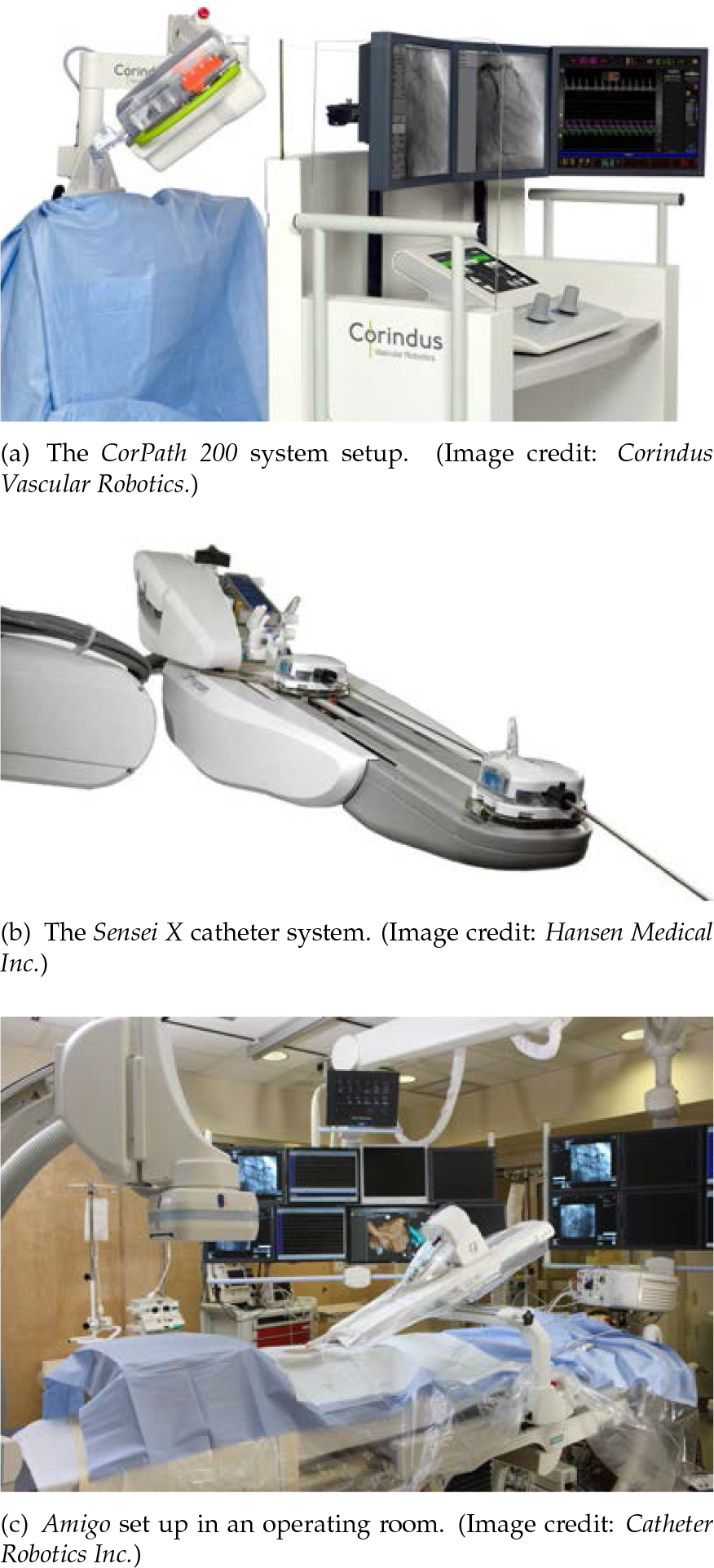
Robotic assisted systems for catheter interventions
3.5.2 Magnetically guided robots
Magnetically guided catheters have the advantage that they do not need to be stiff to resist the pushing force during insertion. This allows a finer and more flexible sheath.
The Niobe ES (Stereotaxis, USA) system follows this approach. With two magnetic field generators, one on each side of the patient, the tip is computer-controlled to be pulled through the veins. To support the surgeon's situational awareness, a force feedback system translates the resisting forces of a collision with surrounding tissues to the human interface [41].
3.6 Sensor and diagnosis systems
MIS offers the advantage of reduced trauma to the patient, but also has the difficulties of visualizing the operation area. Two types of robotised sensor systems are examined: endoscopic camera systems and X-ray imaging devices.
3.6.1 Endoscopic camera systems
Conventionally, the endoscope is controlled by the surgeon or nurse. With the surgeon at the control, s/he is forced to switch at least one hand between the endoscope and a tool. Furthermore the second approach requires good communication and coordination between nurse and surgeon.
Robotic systems provide the advantage for freeing up staff and reducing the surgeon's workload. Two endoscope systems are examined here: Freehand 1.2 (Freehand Ltd., UK) and ViKY (EndoControl, France). Both systems offer a robotised endoscope, which can be controlled through an HMI. Freehand 1.2 comes with a robot arm holding the endoscope mounted on a moveable cart. The surgeon activates the control of the endoscope by a foot pedal. A marker placed on the surgeon's head then allows the system to register head movements and to translate those into the corresponding camera motions [42]. The ViKY, on the other hand is directly placed on the port for MIS. This offers a minimal footprint of the system within the OR [43]. To accompany different needs in the various surgical interventions, the systems comes in two different sizes [43, 44]. A dual control interface is provided by foot switches or voice control, and both interfaces can also be used at the same time [43].
3.6.2 X-ray systems
X-ray imaging devices are usually heavy and large. This makes moving around the device difficult, and positioning the patient to the device during a surgery in also not a good option either. As an application in the OR, Artis Zeego (Siemens AG, Germany) was developed. As shown in Fig. 8, it provides a 6 DOF robotic arm practically carrying a C-arm. While the robot carries the weight, the surgeon can easily move the device around [45].
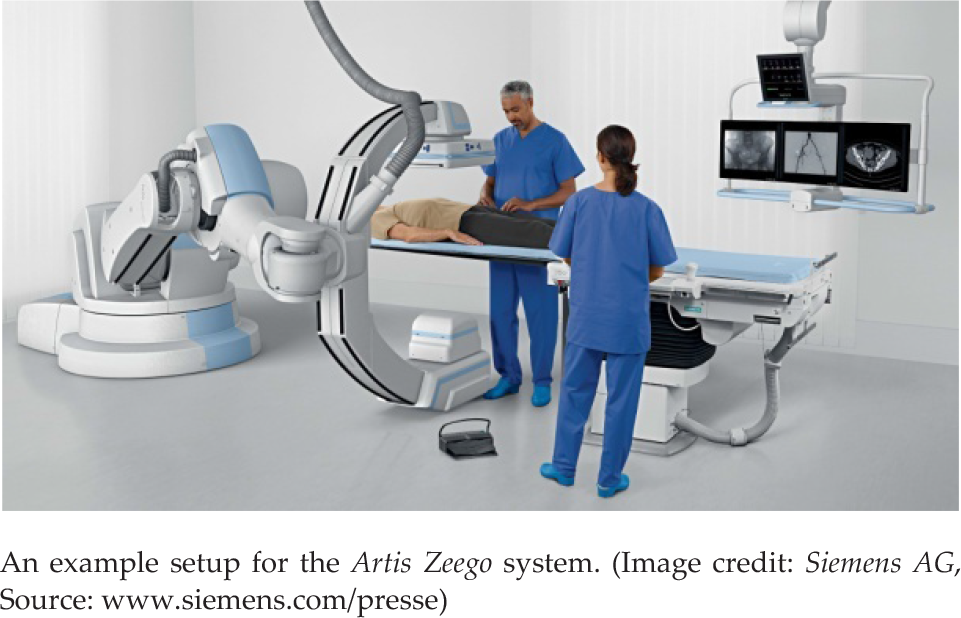
Sensor systems to support MIS and other procedures
Targeted procedures for the Artis Zeego include cardiac, vascular and angiography. Since it is mounted on a robotic arm, repeated positions can easily be targeted [45].
3.7 Hair transplantation systems
The ARTAS (Restoration Robotics Inc., USA) for hair restoration shown in Fig. 9 provides a robotic tool follicle harvesting. This contains a camera vision system and extraction needles for the harvesting [46]. The surgeon places the tool over the harvesting area, where the camera captures the hair positions and software calculates which can be extracted in evenly thin portions. The extraction process of the selected hairs is then conducted automatically. To provide maximum accuracy, the system adapts to patients' movements during the procedure [46]. Each harvesting sessions lasts between six to ten hours and up to 1600 hair follicles are harvested [47]. The harvested hairs need to be implanted by the surgeon afterwards by hand [46, 47].

The ARTAS hair restoration system. (Image credit: Restoration Robotics Inc.)
3.8 Active tool guidance systems
Many surgical tasks demand high situation awareness and navigation capabilities on behalf of the surgeon. The visual limitations of MIS have raised the challenge to accurately pinpoint the tools' actual position within the patient. This task, which requires exact insertion of needles, is difficult to achieve when there are only a few landmarks outside the body to orientate along [11].
To ease this task, robotic assistance systems have been developed. This new generation of intra-operative navigation systems provide some form of active guidance well beyond the classical registration-based visualization and tracking [11].
3.8.1 APEX Robotic Technology (ART)
The ART (OMNI life science Inc., USA) system simplifies the bone cutting operations during orthopaedic surgeries. The surgeon digitizes the bone after the skin is removed. The software then calculates the optimal positioning of the cuts to ensure the proper function of the prosthesis. The tool itself is basically a stabilizer with a guide rail, which positions itself to help the surgeon to cut the bone precisely [48].
3.8.2 NavioPFS
The NavioPFS 6 (Blue Belt Technologies, USA) systems provides a 3D motion tracking of the tools. Fig. 10(a) shows the hand piece of the system, the tracking sensor is visible at the top. The tool itself is hand held by the surgeon and offers a milling tool at the tip [49].

Systems to support the navigation and position of surgical tools and applications
After preparation of the patient, s/he is registered to the system, and the software calculates the limits for the milling operation based on pre-operative data. The surgeon performs the milling similar to the conventional approach, but the system monitors and visualize its progress. In case the tool approaches the limits of the planned carving, the systems slow down the milling tip to prevent unwanted limit breaches [49]. It can even retract the milling part of the tool to enforce safety at the defined limits [50].
3.8.3 Robotised Stereotactic Assistant System (ROSA)
The ROSA (Medtech, France) is a 6 DOF robot mounted on a wheeled chart. As pictured in Fig. 10(b), the arm carries a cooperatively controlled needle guide.
Targeted domain for this system include neurosurgical procedures that require high position accuracy to prevent unwanted damage to the patient's brain.
The cooperative control approach allows the surgeon to use his/her usual movements to place the guide rails. Based on pre-operative data, the system can limit the positioning of the needles around defined limits (Virtual Fixtures) and therefore increase the precision of the placement [51]. With the mandatory registration of the patient's position to the system, it is also capable of positioning itself for the planned intervention. Registration can be achieved with or without the use of a stereotactic frame [51, 52]. ROSA has also been used for orthopaedic procedures recently.
3.8.4 iSYS robot
The iSYS 7 mobile rack robot (Fig. 11) and its original version (B-ROBs) were developed for CT- and US-guided biopsies by the robotics laboratory of ARC Seibersdorf Research (Austria) and the ACMIT 8 center in coperation with iSYS Medizintechnik Gmbh [143, 144, 145]. Besides being used for different needle-based procedures in clinical routine since 2011, a recent clinical study was validating the potential of the robot in neurosurgery [283]. The various systems were thoroughly tested on needle-penetrable phantoms, where the application accuracy was 1.1 ± 0.8 mm, which was shown to be better than the traditional freehand technique [279].

The iSYS mobile rack robot. The iSYS1 is a compact robotic targeting system to assist interventional radiology and neurosurgery procedures with 3D image-based needle path planning. (Image credit: iSYS Medizintechnik GmbH [143])

The discontinued systems of Computer Motion Inc. (acquired by Intuitive Surgical Inc.)
3.8.5 Robotic scrub nurse
Another scenario to employ automation in the OR is a robotic scrub nurse (although it is not a surgical robot). The nurse's task is to provide the surgeon with the tools required, and also to be responsible for tracking the tools in use to prevent leaving any behind accidentally. The tracking task is nowadays supported by certain protocols and even technologies, such as the SmartSponge 9 (Clear-Count, USA) system. Current practice relies on the proper use of checklists, registering in/out the tools required. To circumvent this, an automated tool handling nurse may be used. The system is fed with the planned procedure, and could identify parts of the procedure during the surgery. The communication between the surgeon and robot may be done with hand gestures to prevent any transmission of bacteria [53, 54]. A camera-based system could also track the supplied tools and disposables, and if something is missing in the end the surgeon would receive an alarm. Such a system could be extended by mounting an antenna and RFID chips on tool and the surgical table. Then, every tool could be recognized (assuming they are tagged properly). This would eliminate the need for an extra registration step.
The scrub nurse robot Penelope (Robotic Systems and Technologies Inc., USA) was already introduced to clinics over six years ago. It is an automated robot for hospitals' sterile supply department, where used/dirty surgical instruments are cleaned, sorted, inspected, and repacked into containers, to be sterilized for the next procedure. The robot counts, sorts, inspects, scans, arranges and packs tools. Penelope assisted its first human procedure in 2008, and it also worked together with SRI's 10 M7 (see Table 4) robot [55].
3.9 Summary of the commercial systems
Many of today's systems are built following the master–salve approach, such as the da Vinci (see subsection 3.1). This allows the surgeon to control every move of the robots, but also prevents the advantages of computerized motion control. Other systems execute automatically pre-planned procedures with minor adaptations to, e.g., the breathing motion of the patient (see subsubsection 3.2.1). Autonomous or automated procedures are not used (except for tissue harvesting or orthopaedic procedures), but they might come into regular use within the next years. The examined systems of sections 5 and 6 describe approaches to tackle the disadvantages of today's systems and enable new capabilities in computer-assisted surgery.
4. Discontinued robotic systems
Even though the development of robotic systems in the operation room only began 30 years ago, several systems have appeared and then vanished from the market. The full list of their basic features can be found in Table 2.
The reasons for their discontinuation were not published in many cases; public information is cited in the respective section Table 2.
4.1 AESOP and Zeus
The Automated Endoscopic System for Optical Positioning (AESOP) system was an automated positioning system that also featured voice control for the endoscope according to the needs of the surgeon [15, 56–58]. At the time it received FDA approval in 1994, and was one-of-a-kind MIS surgical system.
Based on the AESOP technology Computer Motion Inc. built the Zeus system [57] approved by the FDA in 2001 [56]. The surgeons had two tool arms and an AESOP endoscope at their disposal. One feature unique to the Zeus system was the possibility for remote control in telesurgical MIS, since it employed the generic UDP 11 over the Internet. Its potentials were clearly demonstrated in transatlantic surgery (the Lindberg operation) in September 2011 [59]. Jacques Marescaux (Institut de Recherche contre les Cancers de L'Appareil Digestif—IRCAD) performed the operation from New York. The patient and the slave robotic arms were in Strasbourg, France, approximately 7,000 km from the surgeon's site [60].
The AESOP and the Zeus were aimed for general MIS, however, following the acquisition by Intuitive Surgical Inc. the both systems were discontinued in 2003 [61].
4.2 Pathfinder
The Pathfinder (Prosurgis Ltd., UK), as shown in Fig. 13, is a small 6 DOF robot mounted on a rolling cart. It carried a needle guide rail, and the procedures of the Pathfinder were supported by immobilizing the patient's head with a Mayfield head clamp. Targeted applications were needle insertions into the patients head. Therefore, the system registered the head of the patient with the help of reflective markers and a camera mounted on the tool holder. Then, the system aligned the needle guide according to the preplanned trajectories. The fixed guide rail allowed the surgeon to insert the needle with an increased precision compared to the conventional approach [62, p. 47–48].

The Pathfinder system. (Image credit: Prosurgics Ltd.)
The system was discontinued due to a lack of commercial interest, along with the company's other technology, EndoAssist.
4.3 Computer Assisted Surgery, Planning and Robotics (CASPAR)
The CASPAR (OrthoMaquet, Germany) was a milling robot composed of a 6 DOF robot arm, with a bonedrill, as a tool, as shown in Fig. 14

The CASPAR system. (Image credit: OrthMaquet GmbH)
The main applications for the system were knee and hip surgery [63] and Anterior Cruciate Ligament (ACL) reconstruction [64]. The surgical plan execution was based on CT data, the robot executed the drilling of the required holes automatically after registration [63].
4.4 Probot
The Probot (Imperial College, UK), as shown in Fig. 15, was intended to support the surgeon with cutting out the prostate through the urethra [65]. Conventional treatments are done with long tools which are inserted through the natural orifice [66]. It offered 3 DOF actuation aligned in such a way, that the cutter at the tip of the tool could carve out cuts concentric cones, beginning at the most inserted point to the outside [67, 68]. With the system, the surgeon only measured and pre-set the required cutting, and programmed the robot. After manual alignment of the robot to the patient and insertion of the tools, the process was executed automatically [67, 68]. The technology was later acquired by Acrobot Inc. and no further updates on it were published (technically, it was discontinued).

The Probot mechanical system setup. (Image credit: Imperial College)
5. Systems under development for commercial exploitation
In contrast to previous sections, this section focuses on the prototypes that may create the foundation of future development paths. The full list of systems is featured in Table 3.
5.1 Improvements in robotic assisted MIS
The next step in the development of MIS might be the transition to single site surgery (some forms of it are called Natural Orifice Transluminal Endoscopic Surgery—NOTES) Here, the operation area is accessed through only one incision in the patient's skin. This approach is also designed to reduce trauma for the patient even further, but it makes the tools and control more complex.
Intuitive Surgical Inc. adapted its system, and has provided tools for single incision surgery since 2011. The distal half of the tools is flexible, allowing them to pass through the trocar (a five-lumen port) along a curvature, and cross over in the abdomen. Unlike in the case of manual single port devices (such as Covidien's Roticluator), the computer control mirrors back the arms when linked to the master controller. Nevertheless, the employment of semi-rigid tools means a loss of dexterity, and requires guidance tubes to ensure the right position of the tools above the target [69].
In mid-2014, Intuitive announced the FDA approval of its new da Vinci Sp single port robot system that had been first shown to the public as a research prototype five years before. The tool delivers an articulated 3D HD camera, along with three fully articulated instruments over a 25 mm port. Its introduction to the market is currently projected for the second half of 2015, as an extension to the da Vinci Xi system [70].
In Fig. 17(a) the ALF-X (Sofar S.p.A, Italy) is shown, which aims to address primarily the large footprint problem of the da Vinci. (Although, the latest generation Xi alleviates much of these problems.) This was solved in the case of ALF-X by extending the arms and only mounting a single arm per cart [71], clearing space for the nurses. Nevertheless, extra safety features must still be implemented to ensure collision of the arms to humans [280]. Force reflection and eye-tracking control have also been added to the system. Although the system received CE mark, it is currently only employed in a teaching hospital in Rome.

The da Vinci tool for single site surgery (image credit: Intuitive Surgical Inc.)
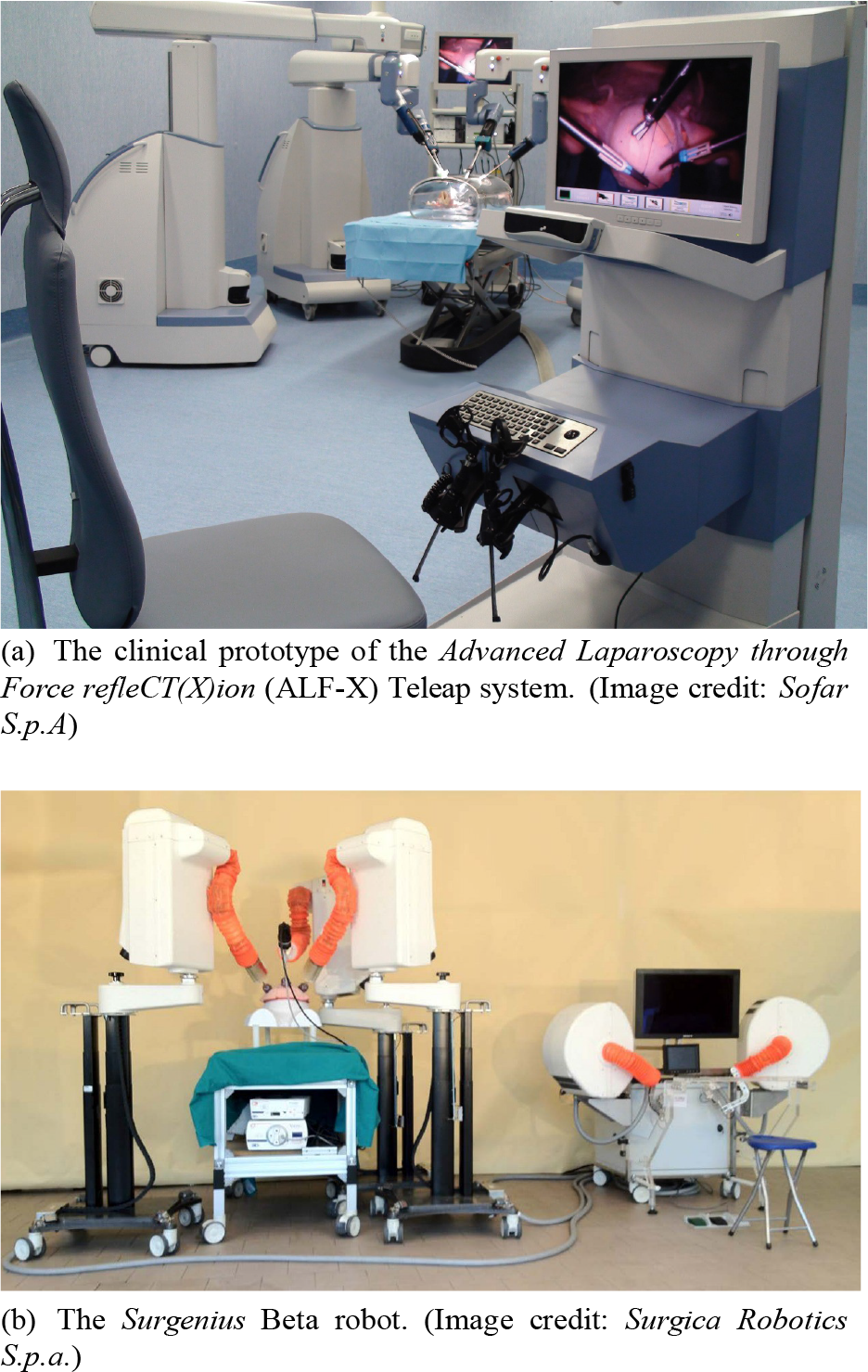
New robots for MIS on the verge of commercialization
Another Italian system, aiming for commercialization in the near future, is the Surgenius (Surgica Robotica Sp.A, Italy), as shown in Fig. 17(b). The development of Surgenius technically dates back to the mid-1990s, when NASA JPL developed the RAMS system. The Robot-Assisted Micro-Surgery (RAMS) system consisted of two 6 DOF arms, equipped with 6 DOF tip-force sensors, providing haptic feedback to the operator. It used a kinematically identical master controller and the operator was seated right next to the slave arms. The robot was originally aimed at ophthalmic procedures, especially for laser retina surgery. It was capable of 1:100 scaling (achieving 10 micron accuracy), tremor filtering (8–14 Hz) and eye tracking. Almost ten years later, the Italian Surgica Robotica S.p.a. company together with the Verona University decided to create a newer version of the RAMS, called Surgenius. The first fully functional ALPHA prototype was built in four months, and has been tested both in vitro and in vivo. One and a half years later, a BETA version was designed and built. Usability and performance evaluation has been carried out with renowned surgeons with extensive experience in robotic surgery. The robot received the CE mark in March 2012, and got through some limited in vivo trials. Currently it waits for further funding before commercialization [72].
5.2 Single site technology platforms
One of the future robots in this field is the Single Port Orifice Robotic Technology (SPORT) from Titan Medical Inc. (Canada). The two arms carry tools and a stereo camera with a light source [73]. Fig. 18 shows the extended configuration of the system. The insertion trocar itself is rigid and provides a fixed base for the flexible end effectors of the tool.

The SPORT system's end effector. (Image credit: Titan Medical Inc.)
Fig. 19 shows the i-Snake robot (Imperial College, UK), which is an entirely flexible robot. It is controlled by commands sent for the tip of the snake. The body would follow the head on the same trajectory by using the patented principle of ‘follow the leader’. This grants access to difficult-to-reach areas of the body. After the targeted area has been reached, the hollow robot provides a guide tube for tools to perform the treatment [74, 75]. Medrobotics Corp. (USA) has developed this principle into a near commercial product, know called Flex. They achieved approval in Europe in 2014, and aim for a first limited commercial launch on selected European markets [75].
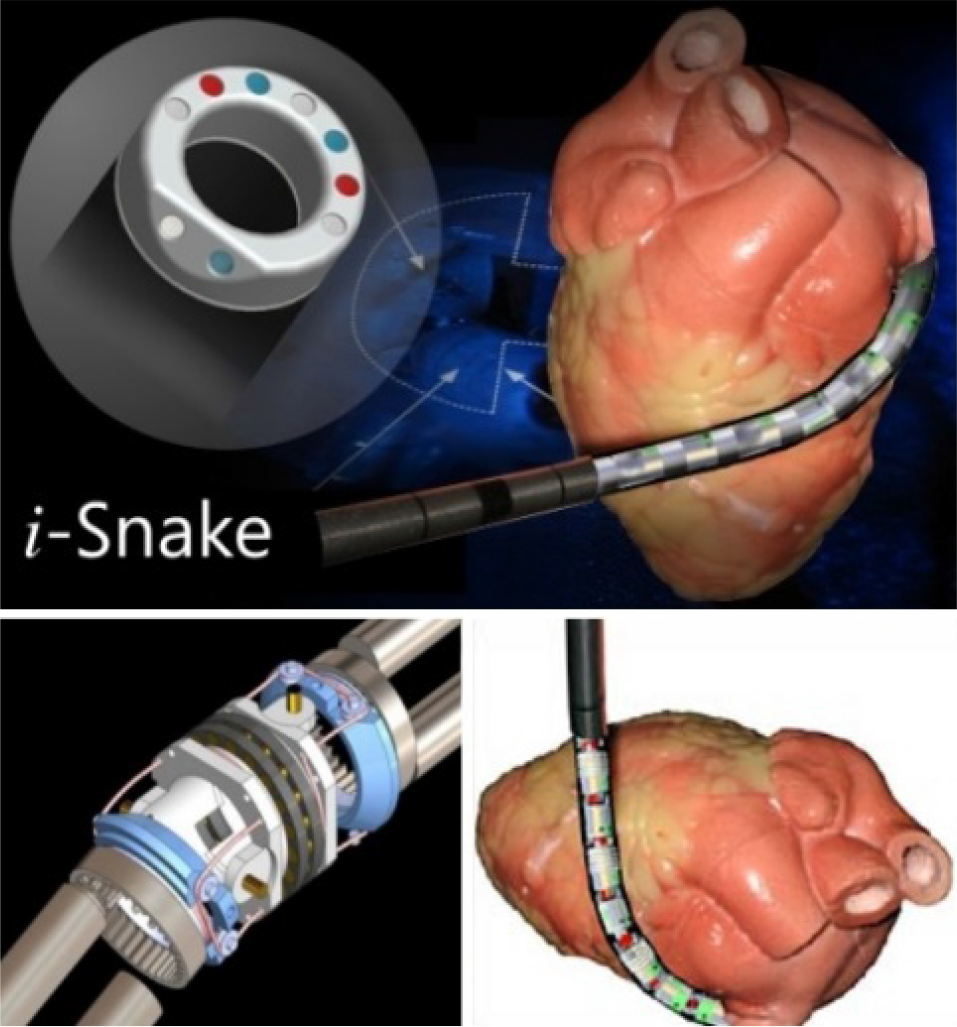
The i-Snake robot in an example use case. (Image credit: Imperial College.)
5.3 Development in organ inspection
Fig. 20 shows the Sayaka capsule endoscope. This is the next step in the development of the conventional capsule endoscopy. The Sayaka system replaces the battery with receiver coils. These coils allow an external source to provide the power for the capsule. It also replaces the forward-looking camera with a rotatable, side facing one. The capsule is propelled forward by the natural movements of the colon. During its way through the bowel, the rotating camera creates a spiral and complete image of the inner surface of the colon.

The inner structure of the Sayaka capsule endoscope. (Image credit: RF SYSTEM lab., RF Co., Ltd.)
6. Advanced research prototypes
6.1 Proposed improvements for robotic assisted MIS
The MiroSurge (DLR, Germany) system follows a similar approach to the ALF-X, as shown in Fig. 21(a) Each arm holds a single instrument, yet the arms are smaller in size and can be mounted directly to the operating table. This prevents any relative movement between the OR table and the robotic cart. The system is designed to be able to perform complex procedures, e.g., beating heart operations, where the tools and camera are moved synchronously with the heart, to give the surgeon at the remote master the impression that the heart stands still. The virtual stopping of the heart allows the omission of the heart lung machine, and therefore reduces the trauma to the patient.

Systems with the aim to improve the current state of robotic assisted MIS
Large distance teleoperation has also been in the focus of many of these development projects [4]. Nevertheless, communication latencies pose a hard limit to master–slave teleoperation scenarios [281, 282], which may only be overcome with the automation of certain functions.
The down-scaling is also enforced in the case of the Surgeon's operating force-feedback interface Eindhoven (SOFIE) 12 system, where the arms are mounted on a single frame which is also attached to the OR table. However, the main feature of the system is the haptic feedback, which is provided through a custom-built master interface.
The RAVEN 2 represents the second most common system in research. The BioRobotics Laboratory at the University of Washington (USA) and its collaborators have designed the lightweight, cable-driven arm, which allows all the movements a surgeon needs for MIS [76]. With the support of the National Institutes of Health (NIH), eight robots have been built and distributed, and the system was also made available as a commercial research platform. To facilitate development and research with the RAVEN platform, Applied Dexterity Inc. was founded. This markets the robot and helps with building a research network, based on the RAVEN platform. They also made their control software open, which is based on ROS 13 . The open software architecture allows researchers to easily prototype custom control algorithms on the system [76].
6.2 Partial automation of instruments
One of the first computer-assisted systems used in surgery was the AESOP, described in subsection 4.1. A more sophisticated approach is an automated tool to support the surgeon. This was demonstrated by E. Bauzano et al.: they monitored the position of the surgeon's tools, and on a command, the robotic tool was moved to the point of the surgeon's tool. During the movement, it avoided obstacles and collisions [77].
Other attempts focused on the increased time consumption of knotting under laparoscopic view. To improve this, a robot has been trained in the use of a neural network to tie knots autonomously [78].
To demonstrate that the combination of many partially automatized surgery setups can lead to a fully automatized surgery, Garcia et al. built a demonstrator of a fully automatized operation room [79]. It featured a da Vinci robot (see subsection 3.1) and scrub nurse robots, serving as the tool handlers. A human physician was only located outside the room to conduct remote surgery on a human phantom without any direct line of sight [79, 80]. They could already prove the basic function with the current technology, but the integration and introduction of such systems to the market is not expected in the near future.
The standardization of basic safety and essential performance requirements of surgical robots have started under the ISO/IEC TC 184/SC 2/JWG 9 Joint Work Group on Standard for Medical Robots [9]
6.2.1 Cognition in surgical robotics
A further step in surgical automation will be the addition of cognitive capabilities to the robot, i.e., the awareness of the current medical situation, and the ability to react in an appropriate way. This capability will initially address simple tasks, such as preparation, suturing and basic cutting and puncturing, thus freeing the OR personnel for the more demanding tasks. This topic has been addressed in a number of meetings held in the past few years, e.g., the IFR Medical Robots Workshop in Heidelberg in January 2012, Milan in July 2012, Karlsruhe in May 2013 and in London in 2013. These workshops effectively established a community of researchers in this area, as demonstrated also by the EU-funded projects with related topics, e.g., SAFROS, STIFFFLOP, EUROSURGE, I-SUR, ACTIVE [81]. This last project, for example, addresses specifically the development of automatic puncturing and suturing. The I-SUR first prototype has 8 DOF, and has been developed to automatize the needle insertion during a kidney tumour cryoablation procedure. Since this task requires a large workspace to properly position and orient the needle over the tumour area, the I-SUR robot has a micro-macro unit structure, with a macro part consisting of a linear delta robot, providing the rigid support to the micro unit, that is a 4 DOF robotic arm, with a serial kinematics, holding the cryoablation needle.
The needle holder incorporates a 6 DOF force/torque sensor (ATI Nano 17, ATI Industrial Automation Inc., NC, USA) to measure interaction forces and torques during needle insertion. The robot controller has a hierarchical architecture: a low-level and a high-level control. The low-level control controls robot position and velocity and is implemented in real-time LabVIEW 2013 (National Instruments, USA) for speed of development. In addition to position and velocity control, software safety routines monitor position, velocity and motor current. The high-level controller defines the end-effector trajectory for the needle insertion based on pre-operative data. This control level uses a passivity-based control method that allows to implement a stable interaction with the soft anatomical environment and a glitch-free transition between autonomous and teleoperated control modes. Novel EU projects, such as the EurEye-Case 14 aims to translate these cognitive functions into clinical applications.
6.3 Flexible robots
In subsection 5.2, the first flexible robot developments for commercialization were presented. Since non-rigid tools and robots offer significant advantages in small spaces over conventional robots, new variations are being developed all the time. One of those is the HeartLander. Its forward motion is supported by two independent suction areas. While one holds the robot in place, the second one is pushed/pulled forward to a new location. It sucks itself to the tissue and the first one releases itself, and then the process starts again, forming an inch worm-like movement. Naturally, it follows any surface it is placed on [82, 83]. The fixation to the surface that is to be treated allows handling with the impression of a stopped heart.
6.4 Improved capsule endoscopy
While the standard capsule endoscopes are not steerable (see subsection 3.4), and some current developments aim for a better observation of the colon (e.g., the Sayaka capsule, see subsection 5.3), some researchers are going another step forward. They are aiming for a magnetically controllable capsule that can be moved by the physician to a desired position. Therefore, the capsule is modified to be magnetic, and the physician controls the motion and position of it with a hand-held or robot-articulated device. This allows for the detailed inspection of, e.g., the stomach [84, 85]. A water-filled stomach can support the diagnosis [86].
6.5 Hand-held robotic devices
According to Payne and Yang [50], five categories of handheld robotic devices have been examined and/or used for surgery:
(For example, the Micron, as listed in Tab. 4.)
(The NavioPFS system, as described in sec. 3.8.2 is a current implementation of this category.)
Inclusion of haptic feedback [89];
Improvement of robustness to tracking occlusion;
Multi DOF tool actuation;
Integration of tremor suppression for applications requiring high accuracy.
Many of the devices require a miniaturization and clinical validation to see a wider adoption. This assumption is based on the facts, that the hand-held devices are smaller, and usually easier to use than the conventional systems.
Generally speaking, a hand-held device does not constrain the surgeon's classical non-robotic access to the patient's body, yet it enhances their capabilities with computer-assistance.
7. Future research directions
Current commercial systems, as already summarized in subsection 3.9, are either directly controlled by a human operator or strictly follow a pre-defined paths. Automated systems are allowed to compensate (to a certain degree) instinctive motions of the patient, such as the breathing motion. Future systems might use artificial intelligence (AI) or cognition to come up with their own decisions. These systems then might take over specific parts of procedures, which are difficult to handle, or require repeated similar motions. The following sections describe a possible development direction of robotic surgery. Predictions are based on the information of the previous sections, and may only represent the authors' anticipation towards the future.
7.1 Anticipated future development
In section 3, a broader overview was given on the existing medical robotic systems. These have been gradually improved over the past years, and probably will continue so. This section aims to sketch the next steps ahead after the implementation of the development directions shown in section 5.
Generally speaking, the introduction of medical robots to the mass market relies on cost reduction. This might either be the shrinking direct costs of the systems or reduced patient care at the hospital. Without significant reduction of the costs, any system will only remain available to the richest institutions and most developed countries.
7.1.1 MIS and Single-Site Surgery
In MIS, as the newest version of the da Vinci also shows, there is still tremendous opportunity for perfection, to gradually improve robotic concepts and systems. While Intuitive Surgical Inc. remains the dominant player in the field, other companies are willing to copy its success and fix minor flaws and drawbacks with their systems.
In general, many of the teleoperated surgical robot companies are aiming to reduce the size and weight of their systems. A smaller OR footprint, especially of the arms, allows closer placement, and thus frees space for movement. Improvements in path planning algorithms could avoid the problem of manual rearrangement to prevent collisions [97]. This can be enabled by the addition of redundant DOF, which allow the robot to keep the position of the end effector stable, while moving the arm out of the way. Such advanced capabilities have been demonstrated by e.g., the new generation of KUKA arms, the LBR4 15 and the intelligent industrial work assistant's (iiwa) new medical version [98].
The implied development direction from MIS (subsection 5.1) to single site surgery (q.v. subsection 5.2) could lead to the development of NOTES-capable robotic systems [99]. An application of the NOTES technique on a routine basis would most likely require more complex mechanics to allow the same degree of freedom as today's system through a single incision. First development bases are shown with e.g., the SPORT system of subsection 5.2. NOTES robots are believed to break the market dominance of da Vinci type surgery one day.
7.1.2 Renaissance of image-guided robots
In orthopaedics, we foresee the renaissance of robotics, Stryker, Google, Ethicon (Johnson and Johnson), Covidien (Medtronic) and other companies are all having a strong bet on the emergence of image-guided robotic surgery. Here again, 20-year-old concepts are re-emerging, since the advancement of technology made it more affordable and easier to achieve engineering soundness. Furthermore, with the rise of alternative tracking modalities, practically all tools can be followed in the operating environment, providing a virtual verification and guidance solution for major surgeries (given an accurate registration).
A current way of development is to integrate the robots with advanced imaging devices to increase their utility by allowing intra-operative imaging. MRI is a non-invasive, high-quality imaging modality, allowing continuous image guidance [29]. MR-compatible robotics has been in the focus of research interest since the mid-1990s, and numerous research prototypes are on the rise beyond the ones already listed in the paper. Johns Hopkins University (Baltimore, USA), with Queens University (Kingston, Canada) and the Automation and Interventional Medicine Laboratory at the Worcester Polytechnic Institute (Worcester, USA) are considered to be the leading centres in MR-compatible neurosurgery and brachytherapy system development [100–102].
7.1.3 Sensor systems
Current sensor systems (as described in subsection 3.4 and 3.6) offer new ways and more information for the surgeon. One main challenge for integration of robotic systems in this area will be the development of reliable and robust algorithms to pre-process the data for the surgeon. This would eventually reduce the workload and increase the concentration on the automatically selected abnormal areas. Main requirement for such computer systems must be the prevention of false negative results, since a missed medical condition would threaten the patient.
Another development area for sensory networks in MIS might be patient registration for currently used systems like the da Vinci (see subsection 3.1). A registration of the patient to the robot can employ an intra-operative navigation system based on pre-operative data it would enable advanced safety features and might also support innovative patient positioning. Currently, the patient is either placed horizontally or fixed very firmly to the table, even though many conventional fixation devices could be compatible with current robots. This is done due to the fear of any patient motion during robotic surgery. Any unwanted and abrupt changes can lead to serious complications and damage for the patient [103, 104]. With knowledge of the patient's movements, the system could adapt to the new situation and prevent such adverse events.
7.1.4 HMI improvements
The layout and working principle of HMI for different systems significantly affects the way a system can be used. A good HMI is intuitive and can be used after a minimum of training. Touch interfaces are currently replacing conventional input devices, such as keyboards, in many areas and the touchless interfaces are also on the horizon, such as ADORA, spin-off from UMC Maribor 16 . A more radical approach would be to create a system that provides surgeons the impression s/he is within the patient's body. With such a Virtual Reality (VR) system, the surgeon's movements could be tracked and translated to directly control the robotic tools within the patient. This VR could be enhanced with additional information from multiple sources and numerous units could be join in a collaborative scenario [105, 106]. An external imaging device, such as an Ultra Sound (US) or a C-arm (maybe a robotic one, such as the Artis Zeego, subsection 3.6) could be merged with the visual image displayed on the surgeon's screen. This could then allow see-through capabilities for surgeons.
Arguably, proper training has become one of the most crucial elements of the operation of complex, modern robotic devices. Recently, the advantages in HMI and computer generated environments led to new methods in surgical training [13, 107, 108]. Different surgery scenarios can be simulated on an high fidelty virtual patient to properly skill the surgeon and improve the outcome of real interventions.
7.1.5 Summary
Fundamentally, pure technological improvements may already lead to reduced patient trauma and new patient care possibilities in various domains of surgery. Furthermore, new technologies, such as the pre-processing of data (indicated in subsubsection 7.1.3) may reduce the workload for the surgeon during the operation. There are certain indications for single-site and NOTES surgery, and these are already applied to various treatment types.
7.2 Assisted surgery planning and execution
An advanced cognitive surgical planning system may provide assistance in navigated surgery. For that, a generalized model of the patient is needed, and the entrance point (port) has to be defined. First steps have been taken in this direction [109], and tool tracking has become possible even under ultrasonic guidance. The information, where the camera and tools are located with respect to the entrance point might support the orientation of the surgeon significantly. If the system registers the patient body as a whole, the absolute position within the body can be calculated. This could be displayed on the vision system of the surgeon. Further iterations of the system might even help in the identification of tissue types. Since the position would be known, the search space for the anatomical structures could be reduced drastically. The extracted possible tissues can then be displayed in an overlay on the surgeons' vision. This knowledge can also be used to compare the collected data to a database of healthy human subjects. If a difference is detected, it could be directly pointed out. This might further help to identify any pathologies, even before they are recognized by the surgeon.
3D printers showed a great progress in the recent years in surgery training and preparation. For the latter cases, scaled 3D printed copies of the real situation of the patient can help to visualize difficult situations before the surgery has begun. This might reduce the patients trauma and therefore support the recovery after the surgery. On the other hand, 3D printing (i.e., stereo lithography) has a great potential even in surgical robotics. The flexibility of the technology may allow one day custom disposable tools and instruments [110].
7.2.1 Knowledge-based assistance systems
Furthermore, a knowledge-based system could be used to identify rare illnesses that the medical staff is not aware of. The input could be the complete medical record of the patient, including the current condition, and the diagnosis would be based on a large dataset, personal history, etc. matched against the collection of known diseases. Then, the team can concentrate on the actual treatment, while the AI sorts out the conditions to be treated. IBM's Watson supercomputer is now working on similar research topics, analysing big data, millions of electronic patient records, which are saved in unstructured natural language format [111]. Microsoft's HealthVault is also seen as a similar system, while Google has given up on its Health project 4 years ago. 17 .
7.3 Partially or fully automated execution
It is believed that certain applications of robotic surgery or computer-assisted surgery might become automated, as first trials have been described in subsection 6.2. Some tasks are easier to automate due to their repetitiveness, such as suturing. Semi-automated and autonomous approaches have been investigated by various groups. On the cognitive side, a long thought-after feature is to estimate what the surgeon would like to do next. This could be taken into account when planning and executing the next movement of the robotised tool or camera [112].
Another possible field of application for partially automatised surgery might be microsurgery of e.g., the eye. Here, the precision of the surgery needs to be high in order to achieve a benefit for the patient with the procedure. One approach to increase the surgical outcome is an automated tool for lens replacements. It could be actuated by magnetic fields and piezoelectric actuators. Even in the early stages of development, such a system allows precise micro movements [113].
7.3.1 Tool tracking
One of the emerging modalities for intra-operative navigation is electromagnetic tracking (EMT) [114]. Its main advantage is that it does not require the continuous line-of-sight between the generator and the receiver (as it is the case with the NavioPFS system, sec. 3.8.2), while this remains a serious limitation for optical tracking, for example. EMT makes it possible to assist complex interventions, such as bronchoscopy, radiosurgery, heart ablation and beyond [114].
In the future, tracking technology could get embedded in the entire operating theatre. The addition of identification marks, e.g., RFID tags, to the tools then also enables tracking and identification of the used tools and the analysis of the procedures and work flows during a surgery without interfering with the workflow. The potential offered by the tracking data includes improvements in surgery support technologies and data gathering for surgical simulation tools. These could be used for training medical staff based on real examples and tracking down sources of error.
Beyond the pure tracking of the used tools, the position data could also be used to support the surgeon. Since the system also knows position, orientation and type of the tools currently in use, the surgeon's vision could be enhanced with augmented reality. Here, an overlay of the conventional image provided by an endoscope may be added to show e.g., where a new tool will appear on screen, or where the surgeon is exactly positioned with the body of the patient. The further development of tracking systems will include the support of multi-sensor applications, significant extension of the work space and innovative types of sensors.
7.3.2 Highly automated surgical procedure
A highly automated surgery could provide medical support for people in remote areas, for example, winter expeditions in Antarctica or extreme situations, such as during the aftermath of natural disasters or on the battle field. To achieve this goal, many technologies are to be developed. Some of them were explained in the previous subsubsections 6.2.1, 3.8.4 and 7.3.1.
These technologies combined with a remote controllable version of e.g., the da Vinci, or one of its competitors might allow the construction of such a Trauma Pod. This partially autonomous patient care unit was proposed by the Defence Advanced Research Projects Agency (DARPA). DARPA's targets for this pod was to improve the early treatment of wounded soldiers on the battlefield. If taken care by a teleoperated robot, the army does not need to risk the lives of highly trained medical staff (c.f. subsection 6.2).
7.4 Free moving micro-robotic systems
As already discussed in subsection 6.4, there are efforts to manipulate the movements of capsules through magnetic fields. An improvement on motion scale and resolution could be achieved by using a computer-assisted field generator. Such magnetic fields are used in the Niobe system as guidance, described in subsection 3.5. Further development in relying on magnetic levitation could allow a free floating capsule with the ability to move in any direction [115], and first experiments have been conducted successfully [116]. Even finer control step sizes could be achieved by using a capsule that is able to manipulate its own magnetic field [117]. But the control, localization and data transmission of micro robots within the human body is not limited to magnetic fields. Nelson et al. [115] discussed in details different options on control and application of different types of micro robots, such as CT or US for localization or different locomotion options.
A step further might be a two-part robot as shown in Fig. 22(a). The main section is magnetic and provides the controllable basement of the robot. It is steered by a system outside the patients body, and it also provides the energy for the payload, which is located in the adjacent nonmagnetic part. Driven by e.g., ultrasonic motors, small surgeries could be conducted or biopsies taken. Without the need of a umbilical cord, the system may include used with high patient comfort and in difficult-to-reach parts of the body.

Setup of the RAVEN in an underwater scenario, the NEEMO 12 (NASA Exterme Environment Mission Operations). (Image credit: NASA.)

Micro-Robotics development concepts and prototypes
7.5 Nanorobots
Considering the miniaturization over the last two decades, it might only be a question of time, when robotic systems will transverse through our cardiovascular system. Nanoscale devices may be used for transporting, delivering and targeting drugs. This means the assembling and control of individual modules, such as nano-actuators and nano-sensors through molecular computer-aided design.
First applications may be a nano capsule, carrying a drug release mechanism that can be controlled from outside the body. This would allow to insert the drug at some point of the patient's body with the activation signal given at the actual treatment area. This could reduce the amount of medication needed to achieve a certain level of medicine at desired positions within the body. The capsule could also be used to store the drug over a long period, to be only activated on demand.
Further system aim to affect various biological procedures, e.g., repairing damaged cells or the DNA itself. Artificial red blood-cells could be affected to absorb a hundred times more oxygen, and nano-robot swarms could be directed around the body with external magnetic fields.
One of the most prominent research group in this area is at the Swiss Federal Institute of Technology Zürich (ETH). They are also involved in the Nano-Actuators and Nano-Sensors for Medical Applications (NANOMA) project, aiming at the development of a drug delivery microrobotic systems. They plan to use ferromagnetic microcapsules in the cardiovascular system, navigated via the induction of force from magnetic gradients generated by a magnetic resonance machine, similar to those used for MRI [118]. To enable such systems self propelled movements through human fluids, researchers at the Max Planck Institute for Intelligent Systems in Stuttgart have developed a small propulsion system, based on external magnetic fields. They copied the scallops pulsating movements, with fast closing and slow opening movements. As we have non-Newtonian fluids in the body, it can achieve forward motion [119].
7.6 Future development roadmap
With the anticipated compound growth of 12.6% per year from 2012 to 2018, the market for medical robots is expected to reach a total volume of $13.6 billion in 2018 [120], and $20 billion by 2020 [277]. To maximize the benefit and market share for their economies, government bodies and NGOs aim to steer and coordinate the future development of medical robots. They try to predict the future development directions and work out areas of cooperation between the different developing bodies. The public interest is to develop fundamental technologies in a cooperative way. Core elements of the future development in medical robotics is aiming at: cost reduction, safety and accuracy [103, 109].
In Europe, the euRobotics aisbl, Europe's highest-level international robotics organization has prepared a Strategic Research Agenda (SRA) [121] followed by a Multi-Annual Roadmap (MAR) [122], laying out the near future of robotic research, including medical application. In the USA, the Government has long been emphasizing the growing significance of robotics, and a key domain within is medical and surgical robotics. Already in 2009, the team of invited US experts created the US Robotics Roadmap, outlining the development of the domain for the next 5, 10 and 15 years, respectively. In due time, the updated version of the US Robotics Roadmap was released in 2013, prepared by the Congressional Robotics Caucus Advisory Committee [123, 124].
8. Conclusion
Over the last three decades of development in the area of computer-assisted medicine and robotic surgery, many new technologies emerged. These laid the foundation of new surgical techniques, supported by capable engineering systems. Not all of these reached the market, and many of the initially successful systems failed to attract sufficient customers. We foresee a steady rise of the field, while the market is also realizing its potential from the business point of view.
The first challenge for the near future will be to adapt the conventional treatment forms to the integrated, computer-assisted alternatives. This requires new training plans for the medical staff, and changes in the layout of the hospitals to accommodate the new requirements. Without a team that can exploit these opportunities to the fullest, the gained benefit of technology will stay small. The other challenge will be to ensure interoperability of systems, and to achieve wider adoption. Today's robot manufacturers are keen on keeping their systems closed in order to ensure customer loyalty and future sales for themselves. To facilitate interoperability, standards in interfaces need to be defined, similar to the ones used in the manufacturing industry. This would allow a more direct competition, which eventually enables new use cases and treatment possibilities. Furthermore, essential objective safety and performance metrics are needed for the entire field, that can only be realized via international standards.
In the age of ICT, engineering supports the medical staff to treat diseases in novel ways. The society is gradually understanding and accepting surgical robotic technology. The development will eventually lead to an increased rate of success with better patient outcome, for the benefit of all.
9. System key-features overview tables
As already introduced in subsection 1.4, the survey incorporates a list of surgical robot system, organized in tabular form. The tables contain the following columns 18 :
Full name of the reviewed system and version number, where applicable. If the system was commercially available, yet discontinued, it is marked, or similarly, if a competitor took over the system and sells it now under a different name.
The name of the manufacturer or developer, including the domicile (city and country).
Primary form of use, e.g., automated treatment delivery, or teleoperated.
E.g., number of tools, availability of haptic feedback, etc.
Foreseen surgical treatment types targeted, even if only a theoretical application domain.
Clearance efforts, focusing on the USA and Europe. For the US, an approval by the FDA and in Europe, the Confirmité Européenne (CE) mark is required.
Estimated cost of a single system is given, if available. Installation costs and maintenance/running costs are indicated, where applicable.
The pages containing the tables are in landscape format for readability and to allow maximum space usage for the tables. All data represents the current best knowledge of the authors, as of April 2015.
Footnotes
10. Acknowledgements
This review was partially funded through the EuRoSurge project (FP7-ICT-2011-7). Further information about it is online available under: http://www.eurosurge.eu/. T. Haidegger is a Hungarian Eötvös Fellow. His work is supported by the Hungarian Academy of Sciences' Bolyai Scholarship. Updates to this article will appear on SurgRob blog (![]() ).
).
1
The evolution of the system is shown in both rows from the left to the right.
2
It is difficult to fit absolute numbers of the precision of many systems. Many reports state only a ‘high precision positioning’, without exact numbers. There are only a few comparisons between the systems based on the mechanical properties. If the numbers are available for a system, those will be indicated within the respective section.
4
Based in Sacramento, USA.
5
Surgical shaping or alteration of the joint.
6
Precision Freehand Sculpturing
7
Interventional Systems, iSYS GmbH, isys.co.at
10
Standfort Research Institute
11
User Datagram Protocol
12
Eindhoven University of Technology, Netherlands
15
LBR stands for ‘Leichtbauroboter’, German for lightweight robot.
18
Research systems are not quoted for approval and costs for obvious reasons.
