Abstract
A series of well-crystallized praseodymium oxide (Pr6O11) nanoparticles are synthesized via a highly scalable process from praseodymium nitrate. Praseodymium hydroxide is synthesized through a modified polyol process and subsequent calcination in air at 600°C for 2 h to transform the precursor material into Pr6O11 nanoparticles. A complete characterization of a specific sample by SEM, HRTEM, and X-ray diffraction (XRD) demonstrates that Pr6O11 nanoparticles form elementary blocks with a calculated size of 118 nm, containing slightly agglomerated nanocrystalline particles with a polycrystalline face-centred cubic phase and an individual size of around 10 nm. Particles can be easily dispersed in water through ultrasonification. Particles display a BET Specific Surface Area (SSA; 8.75 m2 g−1) with a nitrogen adsorption-desorption isotherm of type II and a helium density of powders (d = 5.66 g cm−3).
1. Introduction
Over the past few years, praseodymium oxide (Pr6O11) has been applied in different application domains such as sensors [1], high-temperature pigments [2], catalysts [3,4] and oxygen storage components of three-way automotive catalysts [5]. For such applications, nanosized materials are attractive because of the possibility to tune their physico-chemical properties in terms of shape, size and surface-to-volume ratio.
Pr6O11 is conventionally prepared by solid-state reactions [6–8], a molten salt method [9], template-free precipitation [10], or sol-gel coupled with a shaping process such as electro-spinning [11]. A relatively recent paper has illustrated the possibility of preparing nanostructured Pr6O11 via two traditional methods (calcination of the nitrate and a sol-gel method) and two more sophisticated, modern techniques (citric method and polymerized precursor route/modified Pechini method) [12]. The praseodymium precursor used as a starting material for the synthesis in all cases was praseodymium nitrate Pr(NO3)3.6H2O. As far as we know, there is a very limited number of methods that have been reported in the literature for the synthesis of Pr6O11 nanopowders, although such nanomaterials are expected to provide access to a wider range of applications in set-up devices such as sensors or dispersed in porous ceramic supports to act as a catalyst. A major reason for this is that a reliable shape-, size- and phase-controlled synthesis process leading to the synthesis of nanosized Pr6O11 powders is still not available. Usually, direct heat-treatment of praseodymium nitrate hexahydrate Pr(NO3)3·6H2O leads to micron-sized Pr6O11 powders. In addition, both direct precipitation of Pr(NO3)3·6H2O into praseodymium hydroxide Pr(OH)3 nanoparticles and synthesis of Pr(OH)3 nanoparticles in reverse micelles followed by heat-treatments lead to loss of nanoparticle cohesion, i.e., formation of nanorods. Within this context, developing a new and simple method for synthesis of Pr6O11 particles on the nanoscale could thus open up new avenues for uncovering their remarkable properties and for achieving useful applications.
Inorganic nanomaterials can, in principle, be synthesized using both top-down and bottom-up techniques. The bottom-up strategy uses the techniques of molecular synthesis [13], colloid chemistry [14], polymer science [15], and related areas to make structures with nanometre dimensions. The top-down strategy [16] uses miniaturizing techniques such as machining, templating or lithography to pattern materials. Self-assembly [17–19] bridges these two techniques and allows materials to be designed with hierarchical order and complexity. The use of organic surfactants and polymers in self-assembled nanoparticle synthesis has been a popular method of achieving morphological control, where the polymer or foreign ions typically work to regulate and stabilize the surface of nanoparticles. One of the most common and easy approaches that utilize this strategy is the polyol method [20].
Polyol synthesis is a chemical method involving forced hydrolysis of transition metal salts, which allows metal or metal oxide nanoparticles to be prepared with controlled size, texture and shaping [21–26]. Initial work on elemental metals or alloys exploited the reducing properties of a high-boiling alcohol (glycerol, glycol) towards a suitable metal precursor. Owing to their high dielectric constants, the alcohol acted as a solvent able to dissolve inorganic compounds, a growth medium and a complexing agent in some cases. Furthermore, owing to relatively high boiling points, such alcohol offered a wide operating-temperature range (from 25°C to the boiling point) for preparing inorganic compounds. It also often acted as a stabilizing agent that effectively limited particle growth and hindered inter-particle aggregation.
The polyol method holds promise as a low-cost, high-yield technique with a wide range of scientific and technological applications, which is ideal for potential industrial scale-up [27,28]. In the case of oxide synthesis, the polyol method can be considered as a sol-gel process carried out at relatively elevated temperature with accurate control of particle growth. It has been studied and reported for the synthesis of a wide range of oxide sub-micrometre particles, including Y2O3, ZnO, Mn3O4, CoTiO3, VxOy, TiO2, PbO and SnO2 [29–36].
Ethylene glycol is among the most widely used solvents for the polyol synthesis of metal oxide nanoparticles due to its strong reducing capability, relatively high boiling point and high dielectric constant, which increases the solubility of inorganic salts [37]. Ethylene glycol is commonly used as a crosslinking reagent because of its propensity to coordinate with the central metal ion and form a metal glycolate leading to subsequent oligomerization [38]. It has also been demonstrated that as-synthesized glycolate precursors can later be converted to their more common metal oxide derivatives when calcined in air, while maintaining the original precursor morphology [35–39].
In this paper, we describe a fast, convenient and green synthetic route for the large-scale synthesis of praseodymium oxide nanoparticles with an individual size of approximately of 10 nm by polyol synthesis from praseodymium nitrate in diethylene glycol (DEG), and a subsequent addition of sodium hydroxide followed by calcination in air at 600°C. Thermogravimetric analysis and X-ray diffraction are combined to discuss the effect of both the nature of the Pr6O11 precursor and the quantity of sodium hydroxide added to the praseodymium nitrate-DEG mixture; electron microscopy, EDS, and BET studies were used to characterize the structure, crystallinity, morphology, chemical composition and texture of the Pr6O11 nanoparticles. Also, we note that our method provides nanoparticles that are stable and easily dispersed in water, and could be adapted to the preparation of coatings or porous materials.
2. Experimental procedure
All chemicals were purchase from Sigma Aldrich and used as received. The polyol method involves the reduction of a metal salt precursor by a polyol, a compound containing multiple hydroxyl groups. The polyol used in this synthesis, diethylene glycol ((HOCH2CH2)2O-DEG, 99%), served as both reducing agent and solvent. 2.5 g of praseodymium nitrate (5.75 mmol, Pr(NO3)3.xH2O, 99 %) was stirred in 50 mL of diethylene glycol for 30 min in a Schlenk flask (in air). The as-prepared solution was then treated at 140°C (in air). Sodium hydroxide (NaOH, with a controlled concentration in water) was added after a dwelling time of 1 h, and then the mixture was directly heated up to 180°C and kept at this temperature for 4 h, after which it was cooled naturally.
Following centrifugation, precipitation isolation, washing several times with ethanol, acetone then distilled water, and drying, as-obtained samples labelled
Concentration and quantity of NaOH solution added during the synthesis

Thermogravimetric analysis (Hi-Res TGA 2950, TA Instruments,) of Pr(OH)3 was performed with the sample
3. Results and discussion
The reduction ability of reagents is critical in the control of the size and morphology of nanostructures. Herein, a modified polyol process was designed to synthesize Pr6O11 nanoparticles. The Pr6O11 precursor (Pr(NO3)3 or PrCl3) was mixed with DEG ((HOCH2CH2)2O) to be heated at 140°C before addition of controlled quantities and concentrations of NaOH and subsequent heating to 180°C. After a dwelling time of 4 h at 180°C, the mixture was cooled down to be calcined at 600°C in air.
Firstly, we have compared the effect of the nature of the Pr6O11 precursor on the production of nanoparticles. In both cases, nanoparticles are obtained. However, whereas 2.5 g (5.75 mmol) of Pr(NO3)3 could be easily dissolved in 50 mL of DEG leading to samples
The as-synthesized suspension from Pr(NO3)3 can be used as-is for coating-based processes such as spray-/dip- or spin-coating, or to infiltrate inorganic templates such as ordered mesoporous powders or materials with hierarchical porosity such as those we prepared in our group [40–46]. In the present paper, we obtained pre-calcinated powders by centrifugation, precipitation isolation, washing several times with ethanol, acetone and distilled water and drying, then additional heating to 600°C to obtain the required oxide nanoparticles (Figure 1).

Overall synthetic path employed to generate Pr6O11 nanoparticles
In our process, the polarity of polyol offers the ability to dissolve praseodymium nitrate. As a high-boiling-point solvent, polyol provides a high-temperature environment and improves the reactivity of various reactants. In this work, DEG acted both as the solvent, as a reducing agent and as a growth medium. NaOH provided the hydroxyl ion to form Pr(OH)3, as reported in Figure 2, which presents the XRD patterns of samples
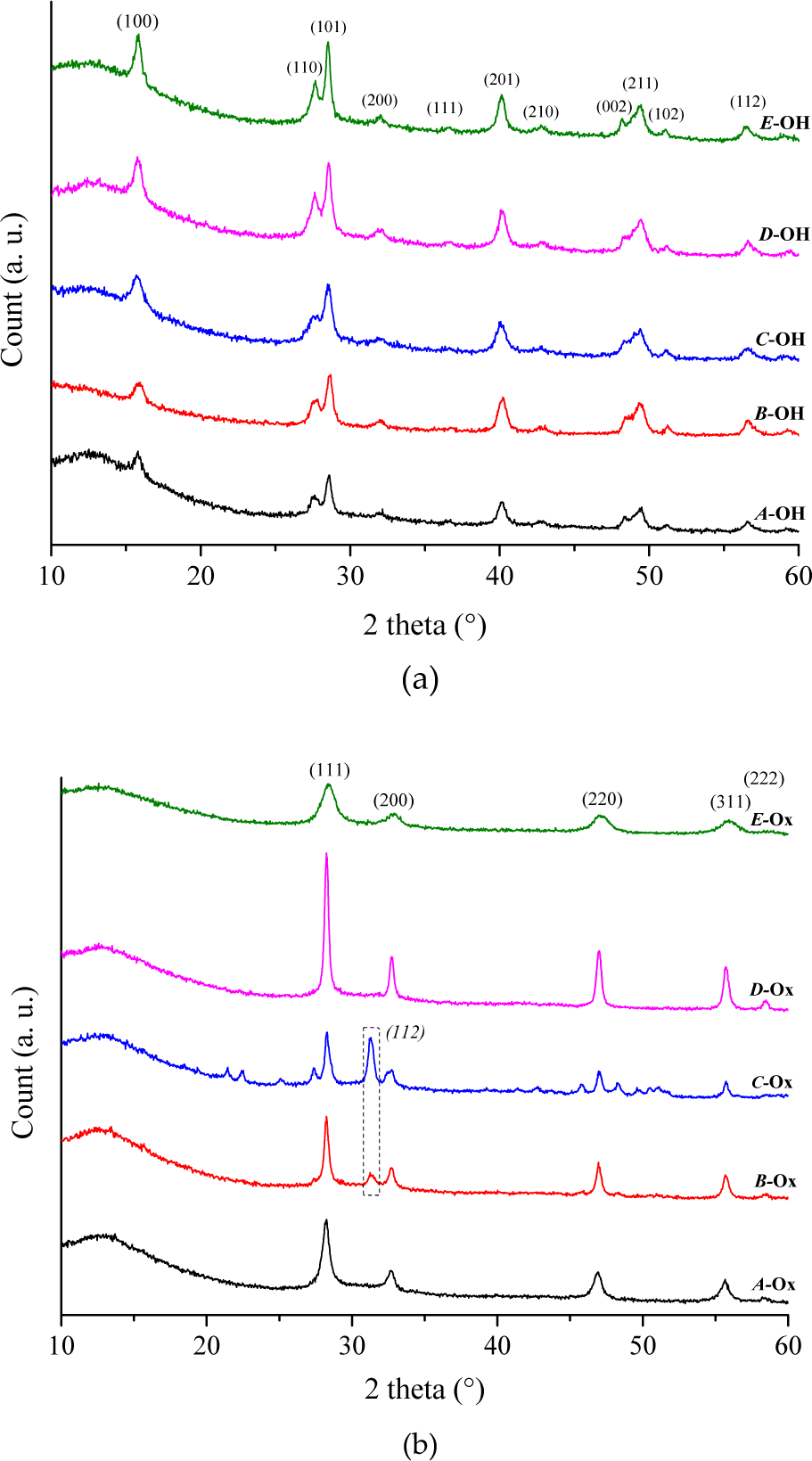
XRD patterns of the pre- (
During the subsequent calcination step, the removal of gaseous by-products induced a weight loss of 75% after thermal decomposition at 600°C (see supporting information, Figure. 1SI). The important weight loss is attributed to the presence of residual DEG trapped in the Pr(OH)3 powders. This is clearly observed in the FTIR spectra of samples
Calculated crystallite size in samples

This clearly indicates the increase in crystallite size along with the concentration of NaOH used during the synthesis. There are no impurities detected by energy-dispersive X-ray spectroscopy (EDS) analysis (see supporting information, Figure 3SI) except for carbon elements arising from the carbon film in SEM observations.
Figure 3 shows the scanning electron microscopy (SEM) images of the selected pre- and post-calcined samples

SEM micrographs of selected pre- and post-calcined samples

SEM micrographs of the sample
We focused our study of the sample
A typical bright-field TEM image of the sample

TEM micrographs of the sample
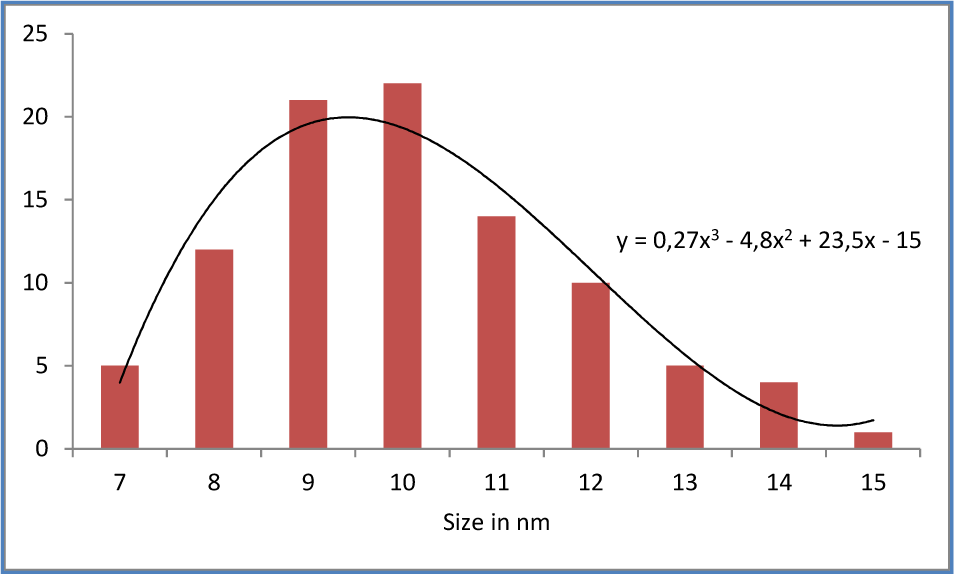
Histogram of the particle size deduced from TEM observations of the sample
The sample

BET nitrogen adsorption-desorption isotherm the Pr6O11 nanoparticles
The form of the hysteresis loop suggests that the sample is basically non-porous and agglomerated. Assuming that the Pr6O11 nanoparticles are almost spherical, as confirmed by TEM, equation (1) shows how the SSA and density (d) can be used to calculate the Average Particle Size (APS in nm) of powders:

An APS of 118 nm is calculated for Pr6O11 nanoparticles.
4. Conclusions
In conclusion, this is a highly scalable and reproducible process for the synthesis of a series of high-purity and well-crystallized Pr6O11 nanoparticles. We have described polyol synthesis from praseodymium nitrate in diethylene glycol (DEG) and the subsequent addition of sodium hydroxide, followed by calcination in air at 600°C to convert into praseodymium oxide (Pr6O11). The morphological and textural analyses of a specific sample showed that the sample formed elementary blocks with a calculated average aggregate size of 118 nm based on the BET Specific Surface Area (SSA; 8.75 m2 g−1) and the helium density of powders (d = 5.66 g cm−3). These elementary blocks are composed of spherical-shaped nanoparticles connected to each other with individual sizes of around 10 nm. The Pr6O11 nanoparticles possess a polycrystalline face-centred cubic phase.
