Abstract
In this paper we report the development of the robot EcoBot-II, which exhibits a primitive form of artificial symbiosis. Microbial Fuel Cells (MFCs) were used as the onboard energy supply, which consisted of bacterial cultures from sewage sludge and employed oxygen from free air for oxidation at the cathode. EcoBot-II was able to perform sensing, information processing, communication and actuation when fed (amongst other substrates) with flies. This is the first robot in the world, to utilise unrefined substrate, oxygen from free air and exhibit four different types of behaviour.
Introduction
Over recent years the notion of autonomy has been amply used in artificial intelligence. In very broad terms, it describes the ability of a robot to operate with minimum human intervention. More emphasis, however, has been given to the ability of a robot to perform computational tasks with minimum human intervention (computational autonomy) rather than to the collection and management of energy from the environment (McFarland, D. & Spier, E., 1999). Autonomous robots, apart from carrying out a pre-assigned mission with minimum human intervention, should also be self-sustainable. Thus, it may be considered that energetic autonomy should be given the same emphasis as computational autonomy in the design process of such systems. When energy collection and management become fundamental for developing autonomous robots, then potentially novel implications emerge for roboticists. For example, the onboard energy supply may not be enough for continuous operation but instead only suffice for periodic actuation. This opens a ‘waiting’ window, of variable time length, which could be used to incorporate other behavioural aspects that may not be very energy expensive, but still important for the agent's operational viability. In the case of raw food substrate (plants, insects) being the energy source, the robot may be designed to be able to extract and utilise this from its environment and remove the waste produced. These are issues that have not been addressed in the past and thus may form a paradigm shift in the way autonomous robots would be designed. This paper describes EcoBot-II which exhibits some partial form of energetic autonomy through the integration with Microbial Fuel Cells (MFCs). EcoBot II is the first robot in the world to be powered by MFCs containing bacteria from sludge fed with unrefined organic matter such as insects (flies), crustacean organisms (prawn shells,) or fruits (peaches, pears, apples, plums) and also employed an O2 cathode. The main aims of this work were to investigate the MFC robustness and capability of going through a feed/exhaust cycle for powering a mobile robot. This was investigated using short distance (50cm) experiments for different substrates. The longevity of the batch culture MFCs in producing energy was investigated using long distance endurance runs in which the same bacterial culture was left to operate continuously, on a single feed at the beginning of the experiments. In both short distance and endurance runs the MFCs were individually exhausted prior to connection with EcoBot-II. This was done to ensure the depletion of any endogenous substrates. The work presented is from the set of experiments in which bacteria in the MFCs were fed with flies and the cathode used was based on the gas (O2)-diffusion system.
Microbial Fuel Cell (MFC)
A Microbial Fuel Cell (MFC) is a two-compartment bio-electrochemical transducer. In the anode compartment, bacteria in an aqueous solution metabolise a given substrate and produce biochemical energy used for maintenance and routine tasks for survival. A small portion of this energy (in the form of electrons) is extracted from the bacterial metabolic cycle and then transferred onto the electrode surface, also found in the anode compartment. At this electron extraction stage, protons flow through the bacterial lipid wall into the liquid solution. Electrons and protons flow into the second compartment of the fuel cell, known as the cathode, through different routes. Electrons flow through an external circuit which is connected to the electrode terminals of both the anode and cathode and protons flow through a proton-exchange membrane (PEM) that physically separates the two compartments. The MFCs used for these experiments together with their physical size and electrode materials have been previously described (Ieropoulos et al., 2005a). A schematic diagram of the redox reactions taking place in a typical mediator-based MFC are shown in Fig. 1.

Redox reactions taking place in the anode (left) and cathode (right) of a typical mediator-based MFC
Many single or mixed bacterial cultures with their unique properties have been used over the years as bio-catalysts in MFCs. For example, pure cultures of E. coli combined with synthetic electron shuttling mediators, were used in the MFCs powering Gastrobot and EcoBot-I (Wilkinson, S., 2000; Ieropoulos et al., 2003). EcoBot-II, on the other hand employed mixed cultures from activated sewage sludge. This strategy offered the advantages of higher power outputs, better stability and the potential to utilise a wide variety of substrates due to the diversity of the microbial community. Although activated sludge samples were exposed to air at the collection stage, anaerobic bacterial groups were revived after the addition of the necessary nutrients and the removal of oxygen. The anaerobic cultures were then injected into the MFCs, where they were subject to exhaustion (discharging) through a resistor load, before being fed and connected to EcoBot-II. Two systems are known to be used in the cathode half-cell of MFCs; the first, which is most commonly used, works with ferricyanide (K3Fe[CN]6) and the other with oxygen (O2). Ferricyanide is a good laboratory standard used in analytical studies that is highly efficient, at the early stages of experiments, but degrades with operation time. This is mainly due to the continuous reduction of the oxidising agent (in the cathode) by the incoming electrons and also the taking up of the hydrogen ions (protons or H+), which result in the need for replenishment. Oxygen, for the gas (O2)-diffusion cathode on the other hand, is available from free air and offers a stable and in some cases improved performance over operation time. Electrons combine with H+ ions and O2 molecules to produce water. Thus it does not suffer from H+ ion accumulation (decrease in pH), which could have adverse effects on the overall MFC performance. The only initial condition required is that the electrode must be kept moist in order for the H+ ions to flow. In this study the cathode electrode was in an open compartment, (see Fig. 2) and the moistening to initiate the reactions was performed manually. Apart from the comparative experiments with ferricyanide, artificial seawater (ASW) was used for moistening the cathode electrodes. ASW was chosen over distilled water and natural rain water, due to its high conductivity, which enhanced the oxidation process. The water production from the chemical reactions was subsequently sufficient to make the cathode half-cell self sustainable. This type of cathode was also ~20g lighter than the ones operating on liquid ferricyanide. Clearly, in terms of autonomous mobile robots, the O2 cathode system was more favourable.
MFCs may be categorized in 3 different types, known as generations, which are distinguished by the electron transfer mechanism in the anode and also by their chronological appearance in the scientific literature (Ieropoulos et al., 2005a). The sludge MFC system initially reported by Habermann and Pommer in 1991 has recently received increased attention from the scientific community but has not yet been fully characterized [Habermann, W. & Pommer, E-H., 1991; Park, D., H. & Zeikus J., G., 2003; Gil et al., 2003; Liu, H. & Logan, B., E., 2004]. Recently it has been reported however that the dominant species in the microbial flora may be a combination of electrochemically active (Gen-II) and anodophilic (Gen-III) (Ieropoulos et al., 2005b). Due to this diversity in the sludge ecosystem, the power output levels recorded were higher than from any of the other three generations MFCs, when compared under identical conditions. Fig. 2 below illustrates a typical current and power output profile from an individual batch-mode (closed system) 50mL sludge MFC. The cathode employed in this experiment was 25mL liquid ferricyanide.
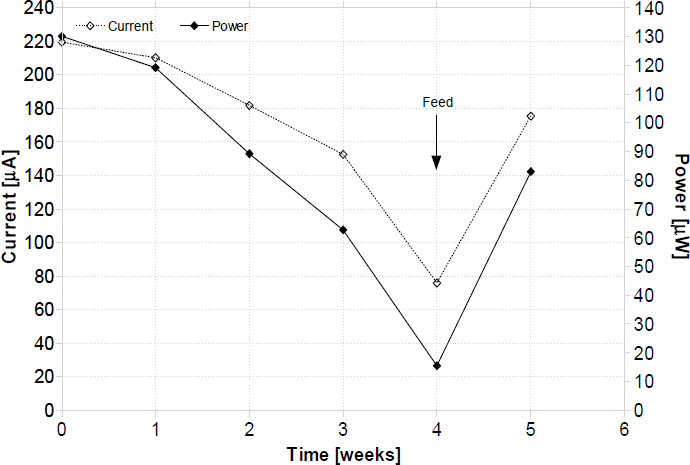
Current and power output profile from a typical single sludge MFC over 5 weeks. Arrow indicates the point of feeding the bacteria in the anode with wastewater. Ferricyanide catholyte replenishment was performed weekly.
Batch-mode MFCs are non-continuous flow systems, in which the feeding of nutrients and disposing of waste products are manually performed. This was the type of MFC employed in the EcoBot-II experiments. As it can be seen from the figure above, the performance of these devices degrades with operation time, due to the depletion of vital nutrients and the accumulation of waste products. In addition, power output decreases as a result of the ferricyanide catholyte degradation. In the experiment above, the ferricyanide catholyte was replenished once every week.
The experiments reported in this paper were carried out using a simple, low-cost platform (Fig. 3). This consisted of light-weight styrene as the chassis, the electronic circuitry and two high torque motors. EcoBot-II was constructed with carved ‘pockets’ on the upper chassis to accommodate and allow the exposure to free air of the O2 cathode MFCs. When fully assembled, it weighed 780g which was ~180g lighter than EcoBot-I.

EcoBot-II with O2 MFCs onboard. Fuel cell units were placed with the open cathode facing outwards, so that the electrode could be exposed to air.
As was the case with EcoBot-I, EcoBot-II operated in bursts of energy. This has been previously described as ‘pulsed behaviour’ and it introduced the waiting window during which energy is accumulated to reach a pre-set threshold (Ieropoulos et al., 2003). None of the two versions of EcoBot incorporated any other behavioural aspect/task apart from energy accumulation during this waiting window. Instead, they remained ‘idle’ for the required time to charge the onboard accumulator.
One of the major differences between the EcoBot-II and its predecessor EcoBot-I, was that the former incorporated additional token tasks. On top of the actuation (phototaxis), EcoBot-II performed temperature sensing, information processing and transmission of the sensed information. For temperature sensing, a 1-wire® low power sensor was used, which was connected to the onboard wireless microprocessor (rfPIC12F675) for data transmission. This resulted in the energy being distributed between the four tasks, instead of being solely used for phototactic locomotion and therefore it was slower than EcoBot-I.
The MFCs that were employed in the EcoBot-II runs were first exhausted by individually discharging them through a resistor load. The exhaustion experiments (Fig. 4) were of sufficient time-length for the MFC power output to reach a pre-set baseline, but not to the extent that would prove lethal for the microbes. This was to ensure that the amount of residual nutrients and carbon energy (CE) sources were kept to a minimum and that the energy to drive the robot was derived solely by the added substrate.
During the exhaustion cycle, which would typically last 24 hours, the O2 cathodes were moistened once with artificial seawater (ASW) to prime the cathodic system (arrow pointing downwards). The arrow pointing upwards shows the feeding point with 0.1% w/v (final concentration) flies, which was equivalent to 1 fly/MFC and the baseline shown as the horizontal dotted line was set to 35μA.
A different set of experiments was carried out to investigate the effect of modifying the O2 electrodes with ferricyanide fixation. In this case, the cathode electrode was soaked in 0.5M ferricyanide made with 0.1M dipotassium orthophosphate (K2HPO4) pH 7.5 for 24 hours and then air-dried for 48 hours. The modified electrode was then directly inserted in the MFC cathode assembly and moistened with artificial sea water. This is shown as data set (b) in Fig. 4 below, which is the average current output from 2 individual MFCs (2 repeats). Only two repeats were carried out for these experiments since these were enough to establish the concept of electrode modification.
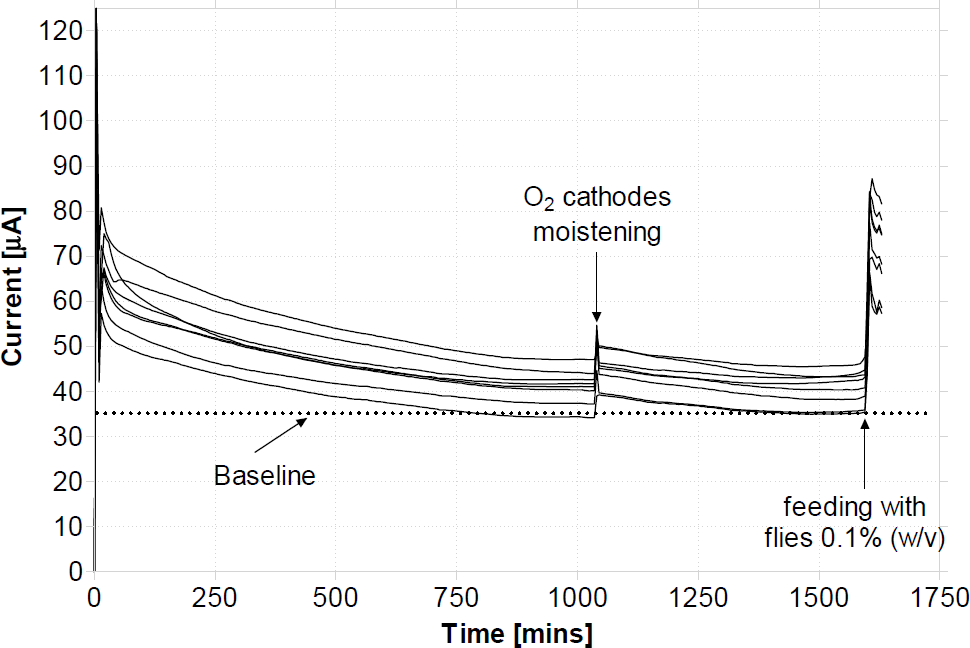
(a) MFC exhaustion cycle for eight different MFCs prior to connecting with EcoBot-II.
Arrows indicate points of feed and cathode moistening. For contrast data-set (b) illustrates the mean current output (2 repeats) from a single batch-mode MFC employing the open O2 cathode of which the electrode had been modified with ferricyanide fixation.
As it can be seen from the data-set (b) in Fig. 4 above the average current output from the modified O2 cathodes was 3-fold (200%) higher than that produced from the plain O2 electrodes that were employed in the EcoBot-II runs (data set (a)).
The main objectives of these runs were to investigate the capability of MFCs as the onboard energy supply and its effects on the performance and energetics of EcoBot-II. In this case the robot was carrying out token tasks, over a certain distance and its performance was compared for different substrates. The runs (Fig. 5) were carried out over 50cm and the duration of each was variable depending on the charge/discharge cycle of the robot. Three exemplar substrates were investigated in this series of experiments; refined sugar, rotten peach portions and dead flies – the latter being one of the most significant milestones in this work and therefore only these data are presented.

Experimental setup for the EcoBot-II short distance runs. In order to perform light seeking, the robot was always placed at a 90° angle with respect to the light source.
The end of the run was signified by the robot reaching the light source whilst performing phototaxis. In order for EcoBot-II to perform light seeking, the start point was (for all three repeats) set at a 90° angle with respect to the light source.
Initial experiments (for comparative purposes only) were carried out with the normal closed cathode system containing liquid ferricyanide (0.5M) in 0.1M K2HPO4, pH 7.5). MFCs were fed with 0.1% w/v flies and the 50cm distance was covered in 2 hours and 10 minutes (data not shown).
Repeat experiments with the open O2 cathode were carried out on separate days, starting at the same time of the day when the ambient temperature conditions were similar. At the start of these experiments each MFC was given 1 fly of identical mass, which was equivalent to 0.1% w/v. During each run, the O2 cathodes were moistened once with 3mL of ASW. Data from these experiments are shown in Fig. 6. below.

Time over distance for the 3 runs.
Fig. 6 illustrates the average data from the three repeat experiments. On average the robot was moving every 14 minutes for ~2-3 seconds before stopping again to recharge. The distance covered for every move was ~2-3 centimeters. The open square symbols shown are the mean from the three repeats. As it can be seen, for the fly-insects substrate, EcoBot-II took an average of 6 hours to cover the 50cm distance, which means that it was traveling at a speed of 8.3cm/h. This was 3-fold slower than the performance recorded when the ferricyanide cathode was employed (initial experiments) in which the mass of the robots was 160g heavier.
The temperature transmitted by EcoBot-II was recorded by the base-station receiver. This was to test the temperature sensing capabilities of the robot. A temperature gradient from the start of the run towards the finish line was created by employing two halogen lamps as the light source. Typically the ambient temperature at the beginning of the experiments was 23°C, increasing to about 30°C at the end of the run.
The objective of these continuous runs was to investigate the endurance and viability of the onboard MFCs from a single feed of 1 fly/MFC. This would give a rough indication of the length of time within which the robot could operate continuously, with minimum human intervention. In these runs, the same experimental procedure was adhered for the individual exhaustion of the MFCs, prior to feeding and connecting them onboard EcoBot-II. Bacteria in the anodes were fed with 0.1% w/v flies, which was in line with the objectives of this work. The robot was left in an open arena, moving towards a light source, sensing, processing and transmitting temperature information to a base station, which was located 20m away from the arena. The light source was placed 6 meters away from the start point. This distance was within the detectable range of the photodiodes so as to allow for phototaxis, but outside the temperature sensor range. EcoBot-II was therefore transmitting the ambient temperature, since there was no gradient produced. In the case of the O2 cathodes moistened with ASW, the end of the experiment was marked by the charge period being in excess of 24 hours. This was long enough to assume that the onboard bacterial cultures were weakened to the extent of their death phase and thus would be unable to contribute to the artificial symbiosis. In the case of the O2 cathodes moistened with liquid ferricyanide the end of the experiment was marked by the robot reaching the light source. Moistening of the open cathode electrodes was performed manually on a once a day basis. For the two repeat experiments in which the O2 cathodes were moistened with ASW, this was done over a period of 5 days which was the required time for the cathode system to become self-sustainable. The robot operated for a mean of 11 days (max. 11.5 days) and the average distance covered was 2 meters (max. 2.15 meters) (data not shown). Time constraints restricted the amount of repeats to only two for these long-term continuous runs. However, the recorded data were within the 5% variation margin (data not shown), which showed consistency and repeatability in the EcoBot-II performance. Hence the need for further time-consuming long-term runs was – at this stage – considered unnecessary.
Ferricyanide moistening
Results from a typical continuous run in which the open O2 cathodes were moistened with ferricyanide (0.5M, pH 7.5) are shown below in Fig. 8. This line of experiments was aiming at investigating the EcoBot-II system performance from the anode point of view, since the cathode would no longer be forming a limiting factor.
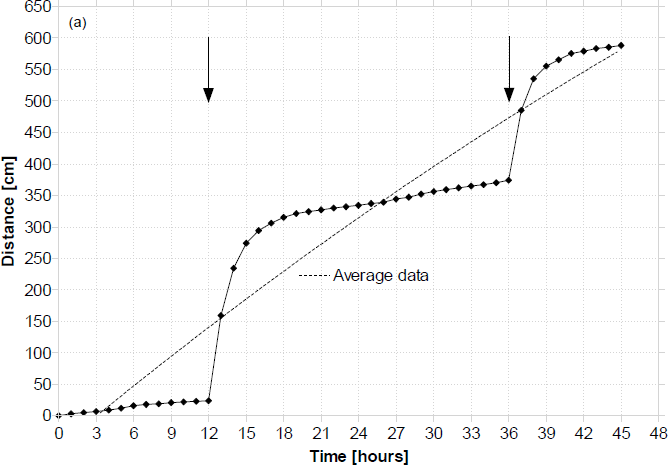
Typical distance vs. time profile for the EcoBot-II continuous runs. Arrows indicate the points of moistening the open O2 cathodes with 3mL of ferricyanide.
Typically the total distance covered in the 2-day period was 5.85m (max. 5.97m), and the time between movements (charge time) at the points of cathode moistening was reduced to 15–20 seconds. This would typically increase to an average of 7 minutes as the open cathodes were evaporating with operation time. The average distance covered per movement was 1.9cm. Fig. 9 illustrates the ambient temperature over the time of day for these 48-hour experiments. Towards the end of the experiment and as EcoBot-II was approaching the light source, it can be seen that the transmitted temperature was gradually beginning to increase. Data illustrated in Fig. 9 represent the typical indoor temperature profile of the laboratory. This was consistent with long-term data recorded from separate temperature sensors.
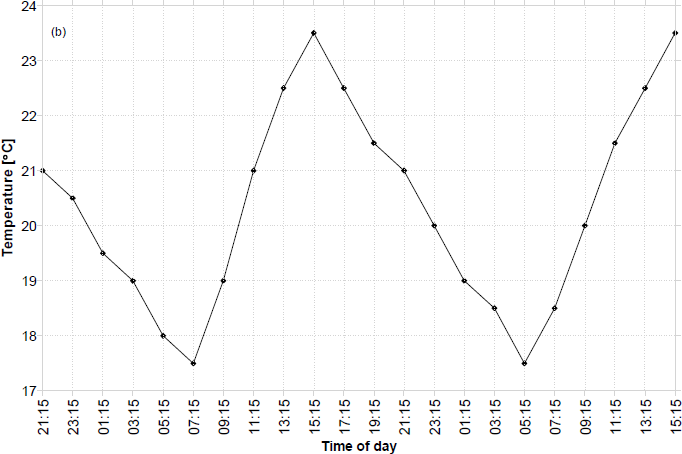
Typical ambient temperature profile transmitted from EcoBot-II as a function of the time of day.
In this paper, EcoBot-II is described which illustrates some of the key aspects of energetic autonomy, one of which is the extraction of energy from the environment. MFCs containing live bacteria were shown to utilise natural unrefined substrates, which could be found in the robot habitat. This widens the range of environments that autonomous robots could operate in. In order for a robot to be able to find and collect its own food, it may have to be built with a high level of software and/or hardware complexity and intelligence. For example, in the case of utilising fruits such as apples, it must incorporate location-recognition apparatus and in addition possess harvesting and breaking down mechanisms. In the case of insects, on the other hand, for example flies, fly traps based on pheromones could be integrated with the robot hardware. This type of trap, does not consume any electrical energy, and the fly attractant can last for long periods of time. One other way of achieving self-sustainability may be through the use of solar panels. Such a system would, however, need to have access (even infrequent) to solar radiation, which may limit its autonomy. Sewage sludge MFCs have recently been reported to exhibit accumulation properties in the form of sulphide and hence improved performance (Ieropoulos et al., 2005b). Activated sewage sludge samples have the ability of utilising common water pollutants such as sulphate, and excreting electro-active metabolites such as sulphide. It has been shown, that the longer the MFCs were left open circuit, the higher the burst of energy was upon load reconnection (Ieropoulos et al., 2005b). This implies that the performance of the robot may be improved simply by changing the time constant of the charge/discharge circuit. This was not exploited in the EcoBot-II experiments. The ultimate design, taking nature as an example, would be a continuous flow system, operating on the robot. This will ensure continuous or at least periodic inflow of fresh nutrients and periodic outflow of waste. The latter is not as critical as the former, since the bacterial cultures are robust enough to withstand prolonged exposure to suboptimal conditions. However, if there are not enough nutrients in their environment, then the lack of microbial power could prove detrimental for the robot. The waste from MFCs containing sludge will be the parts of the energy source that cannot be utilised/broken down (recalcitrant materials) plus dead microorganisms. Provided the original sewage sludge inoculum has been screened for pathogens and is pathogen free, the waste can be safely composted and will be of no danger to humans and/or nature in general. Data in Fig. 2 are part of an ongoing experiment and suggest that the MFC power output may be maintained at constant levels by periodic feeding. The degradation of power with operation time as a result of nutrient depletion and waste product accumulation is a common phenomenon especially after a period of 4 weeks. The fact that the power output increased back up to approximately the previous levels, after feeding and catholyte replenishment illustrates the potential of this system to produce energy over long periods of time (months or even possibly years).
The continuous or periodic inflow of nutrients/substrates and outflow of waste products will probably form the major aspects of the robot architecture and behavior. The energy to operate in this manner would come from the onboard bacterial cultures inside the MFCs. Both the live microbial ecosystem and the artificial robotic system will be mutually benefiting from this association. Bacteria will potentially generate the energy required by the robot to perform token tasks in return for the collection of food from the environment by the robot to feed and thus maintain the microbes. This is what we call artificial symbiosis.
Results illustrated in Fig. 4(b) suggest that the MFC and as a result the EcoBot-II performance could be improved by nearly 200%, when electrodes employed in the O2 cathodes had previously been modified with ferricyanide fixation. Furthermore, during the EcoBot-II endurance runs it was demonstrated that the robot efficiency could be improved by 3-fold, when the open O2-cathode electrodes were moistened with ferricyanide. In this case, the preset distance of 6m was covered in less than two days compared to one-third of the distance (2m) covered in 11 days following water moistening. These results reveal an additional potential for improvement.
Ferricyanide is a well defined electron acceptor useful in a laboratory environment and presently superior to the unmodified O2 cathode system. However, it is a toxic chemical that cannot be found in the natural environment. EcoBot-II was capable of operating without ferricyanide using distilled or rainwater (data not shown) or (artificial) seawater in combination with the unmodified O2 electrodes. These conditions resemble the pragmatically tough environment of the natural world. Autonomous robots can potentially be extremely useful in missions, which are dangerous, undesirable or inconvenient for human beings. An autonomous agent could be used in underwater explorations feeding on plankton and sending useful information on the ecology of the ocean. On the other hand, it could be sent to monitor the sewer system of a city, and by cleaning blockages, generate energy for its maintenance. Organic waste produced at the domestic or industrial level might even be used as the fuel for such a robot that may be patrolling the premises.
Conclusions
Our work has demonstrated the feasibility of extracting electrons from biological substrates (insects, fruits) using microbes in mediator-less MFCs to power small robots. Furthermore, it has been shown that it is possible to use oxygen the electron-acceptor of the MFC to work with air. EcoBot-II can successfully perform token tasks including phototaxis, temperature sensing and data transmission. Functional performance has been shown to continue over a period of ~twelve days with low system efficiency, for the closed system MFCs. Significant improvements could be achieved with periodic nutrient replenishment in a continuous flow MFC. The integration between the artificial agent and the live microbial ecosystem may be considered as a form of artificial symbiosis which could potentially be one method of developing autonomous robots.
