Abstract
Raspberry-like SiO2 particles with a nano-micro-binary structure were prepared by a simple sol-gel method using tetraethoxysilane (TEOS) and methyltriethoxysilane (MTES) as precursors. The chemical components and morphology of the SiO2 particles were characterized by Fourier transform infrared spectroscopy (FT-IR) and a Transmission electron microscope (TEM). The surface topography and wetting behaviour of the raspberry-like SiO2 surface were observed with a Scanning electron microscope (SEM) and studied by the water/oil contact angle (CA), respectively. The thermal stability of the prepared SiO2 particles was characterized by TGA analysis. The results show that the highly dispersed SiO2 particles initially prepared by the sol-gel method turn into raspberry-like particles with during the aging process. The raspberry-like SiO2 particles show superhydrophobicity and superoleophilicity across a wide range of pH values. The SiO2 particles were thermally stable up to 475°C, while above this temperature the hydrophobicity decreases and finally becomes superhydrophobic when the temperature reaches 600°C. The raspberry-like SiO2 particles which were prepared have potential applications in the fields of superhydrophobic surfaces, water-oil separation, anti-corrosion and fluid transportation.
1. Introduction
Superhydrophobic surfaces are currently a subject of great interest and enthusiastic study because of their various important applications. They show promising applications in many fields, such as self-cleaning, antifouling, fluid friction reduction in microfluidic devices, anti-stiction in MEMS devices, antibacterial coatings and building walls.[1–4] The self-cleaning mechanism learned from lotus leaves suggests that a combination of low surface energy materials and a dual-sized roughness is required to accomplish a superhydrophobic surface. Therefore, in order to obtain superhydrophobic films, the modification of the surface chemistry must always be in conjunction with the enhancement of the surface's roughness.[5] Many methods have been used to prepare superhydrophobic surfaces by mimicking lotus leaves' surface structure. However, most of the methods involve strict conditions, such as harsh chemical treatments,[6–7] expensive materials[8–9] or else complex processing procedures.[10–12] As a consequence of these limiting features, the practical application of superhydrophobic surfaces has been strongly limited in most cases. More recently, it was found that surfaces with superhydrophobic properties combined with other special properties have many other potential applications. For example, a superhydrophobic surface with a superoleophilic property can be used in the field of water-oil separation processes.[13–14] Superhydrophobic surfaces with a wide pH range may be widely used in the anti-corrosion industry in the near future.[15] Also, superhydrophobic particles are being used in the transportation of liquid marbles.[16–17]
In our previous study, we focused on the wetting behaviour of raspberry-like structured SiO2 particles prepared through a sol-gel method.[18] In this work, we make a further study on the formation, characterization and properties of the raspberry-like SiO2 particles with a nano-micro-binary structure. It is expected that the prepared SiO2 particles will have potential applications in fields of superhydrophobic surfaces, water-oil separation, anti-corrosion and the transportation of fluid, among many others.
2. Experimental
2.1 Materials
Tetraethoxysilane (TEOS, 98%, A.R.), methytriethoxysilane (MTES, 98%, A.R.), ammonia (NH3·H2O 25%, A.R.), ethanol (EtOH, 99.5%, A.R.) and deionized water have been used in this work. The chemicals were used as received.
2.2 Preparation of the Raspberry-like SiO2 particles
The nano SiO2 sol solutions were prepared by a modified Stöber method. Firstly, a mixture solution of EtOH (100.0g), H2O (10.0g) and ammonia (15.0g) was added to a 500mL flask equipped with a reflux condenser, a mechanical stirrer and dropping funnels and then heated in the water bath. When the temperature was raised to 60°C, the mixture of TEOS (20.0g) and EtOH (10.0g) was added into the flask drop-by-drop for about 30min at a stirring speed of about 250rpm. After feeding, the reaction was kept for 2h, and then the mixture of MTES (10.0g) and EtOH (10.0g) was added into the flask drop-by-drop for about 30min, with the reaction being allowed to keep for another 24h. The above prepared SiO2 sol was allowed to age for about 48h at room temperature so that the raspberry-like SiO2 particles could be obtained.
2.3 Characterization
FT-IR (Vector 33, Bruker, Germany) was used to study the chemical components of the SiO2 particles. The morphology of SiO2 particles in the sol solutions was observed by TEM (JEM-100CXII, Japan). The surface topography of the SiO2 film was observed by field-emission scanning electron microscopy (FESEM, LEO 1530 VP, Germany). Static contact angles (CA) were performed with an optical contact angle meter (OCA40 Micro, Dataphysics, Germany). The hydrophobicity and oleophilicity of the SiO2 films were measured with 5μL water and hexadecane, respectively. An average of five measurements was made to determine the hydrophobicity of the film. The thermal stability of the SiO2 particles was tested by thermo gravimetric analysis (Model STA449C, Netzsch, Germany) at a heating rate of 10°C/min from 100°C to 700°C.
3. Results and Discussion
3.1 Chemical components of SiO2 particles
As is shown in Fig 1, curve a is the FTIR spectrum of the SiO2 particles prepared by TEOS and MTES as precursors, while curve b is the FTIR spectrum of the SiO2 particles prepared by TEOS as a precursor.

FTIR spectra of the SiO2 particles (a) TEOS as a precursor; (b) TEOS and MTES as precursors
Comparing curves a and b, peaks of Si-O-Si (1094 cm−1, 798cm−1 and 470 cm−1) and Si-OH (960cm−1and 1630 cm−1) can be detected. While in curve b the peak of Si-C (1270 cm−1) and C-H (2950 cm−1 and 1402 cm−1) in the methyl group can be observed, it can also be noticed that the Si-OH group absorption band, at around 960 cm−1 and 1630 cm−1, is much weaker than that in curve a. The reason is that part of the Si-OH is replaced by Si-CH3.
3.2 Morphology and formation process of the raspberry-like SiO2 particles
Fig 2 shows the morphology of SiO2 particles first prepared (Fig 2 (a)) and aged for 48h (Fig 2(b)), we can see that the SiO2 particles that were prepared first are highly dispersed, but that after 48h of ageing time the SiO2 particles begin to assemble together and a raspberry-like particle is formed. Due to the -CH3 group from MTES, the raspberry-like SiO2 particles display excellent hydrophobicity. The formation process of the raspberry-like SiO2 particles is shown in Fig 3.

TEM image of the prepared SiO2 particles (a) before aging (b) aged for 48h

The formation process of raspberry-like SiO2 particles
3.3 Surface wetting behaviour of the raspberry-like SiO2 particles
Fig 4 shows the SEM image and water/oil contact angle of an SiO2 film formed by raspberry-like SiO2 particles. From Fig 4(a), we can see that there are many micrometer-scale concave and convex structures distributed on the surface of the SiO2 film; the micrometer-scale concave and convex structures consist of many nanometre particles and were due to the raspberry-like structure of the SiO2 particles. The combination of a nano-micro-binary structure and a hydrophobic/oleophilic -CH3 group leads to the superhydrophobic/superoleophilic properties of the SiO2 film. Besides this, the SiO2 film shows superhydrophobicity across a wide range of pH values. As can be seen from Fig 5, there is no obvious fluctuation of the CA values within the experimental errors over a pH range from 1 to 14. All of the CAs' values are within the range of about 156° to 162°, indicating that the pH values of the aqueous solution have little or no effect on the CAs for the SiO2 as-prepared surface. This phenomenon indicates that such a surface is superhydrophobic not only for pure water but also for corrosive liquids, such as acidic and basic aqueous solutions.

(a) SEM image and Water/Oil CAs of the raspberry-like SiO2 film; (b) Partial enlargement of Fig 4(a)

The relationship between pH value and CA on the SiO2 surface
3.4 Water marbles and the transportation of liquid droplets
Superhydrophobic SiO2 powder was obtained by the spray evaporation of the solution of raspberry-like SiO2 particles. By rolling a water drop on the raspberry-like SiO2 powder, liquid marbles were obtained. This SiO2-powder covered water droplet (i.e., liquid marble) can be placed on various substrates, such as glass, paper, wood and polymer coatings. Fig 6 shows a typical liquid marble placed on a mirror. It can be observed that, irrespective of the nature of the substrate, a small marble maintains its sphericity. The prepared liquid marbles are found to show a dramatically reduced adhesion to solid surfaces. Therefore, motion may be generated using gravity. Moreover, the quick displacement of the droplets without any leaks can be achieved because the viscous friction associated with motion is very small. All of these features have potential benefits in micro-fluidic applications.

Liquid marble (diameter: 3.5±0.2 mm) placed on a mirror
3.5 Thermal stability of the raspberry-like SiO2 particles
The thermo gravimetric analysis (TGA) curve for the SiO2 particles is depicted in Fig 7. The weight-loss phenomena with respect to the increase in the temperature within a range of 350-550°C corresponds to the oxidation of -CH3, and the SiO2 film finally develops a superhydrophilic nature. The thermal stability was confirmed by putting the SiO2 film sample in an oven at different temperatures for 2h in ascending order and then testing the water contact angle until the wetting behaviour of the SiO2 film changed. The test results are shown in Fig 8, showing that the SiO2 particles were thermally stable up to 475°C and that above this temperature the hydrophobicity decreases and finally turn into superhydrophilic when the temperature reaches 600°C.
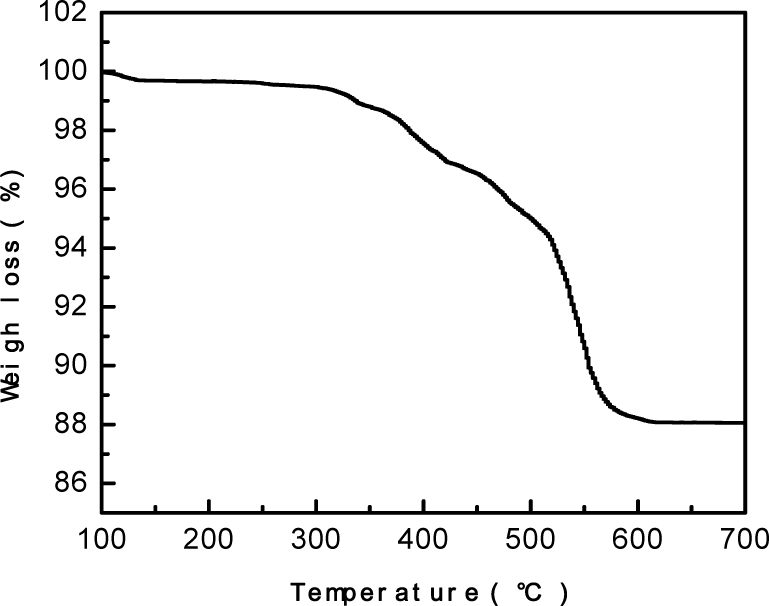
TGA curve of the SiO2 particles
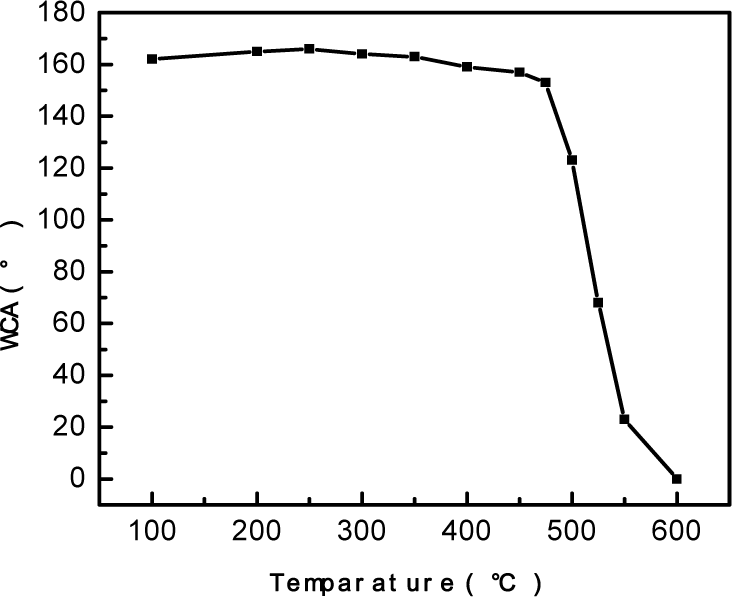
WCA of the SiO2 film with thermal treatment
4. Conclusion
Superhydrophobic SiO2 particles with a raspberry-like structure were fabricated through a simple sol-gel method using TEOS and METS as precursors. The raspberry-like structured SiO2 particles were initially formed from the highly dispersed SiO2 sol due to the gel's effects during the aging process. The raspberry-like SiO2 particles show superhydrophobic and superoleophilic properties due to the hierarchical structure of the SiO2 particles and the end -CH3 group. Besides, a superhydrophobic SiO2 surface is present not only for pure water but also for corrosive liquids and it has potential application in micro-fluidic transportation. The superhydrophobicity of the SiO2 particles can withstand up to 475°C and, above this temperature, the hydrophobicity decreases and finally becomes superhydrophilic when the temperature reaches 600°C.
