Abstract

In the past decade, there has been a significant growth in the literature with a focus on neuroimaging of psychogenic nonepileptic seizures (PNES). The expectation of this work is the identification of biomarkers for diagnosis, treatment response, and prognosis of PNES. With different studies identifying different potential regions of interest, one critique of the nascent literature is that some studies seemingly contradict other studies, showing divergent results in similar populations. Although the current findings of neuroimaging literature are not helpful to diagnose PNES in individuals, they provide a foundation for postulating neurobiological underpinnings of PNES. Examining the available PNES neuroimaging data and supplementing them with data from studies of other functional neurological disorders (FNDs)/conversion disorders (CDs) to derive commonalities allows for proposing and providing the initial structure of a network model involved in generation and maintenance of PNES (see Figure). This comparison may allow us to identify available clues and future goals to address existing deficiencies in the current science, thereby providing a well-supported and unified theory of FNDs/CDs.
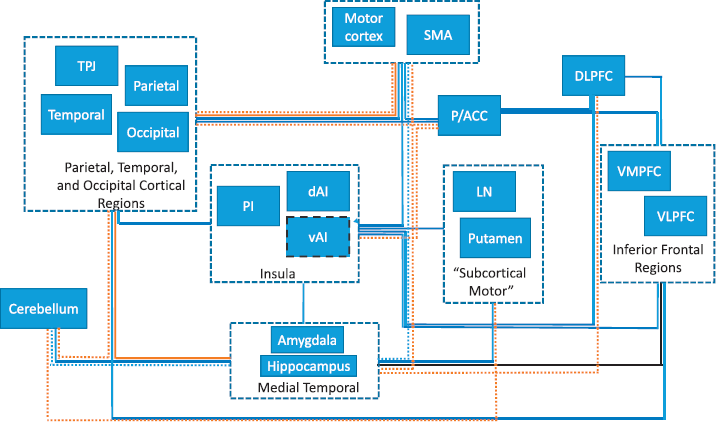
Schematics of possible nodes of the network underlying development and maintenance of psychogenic nonepileptic seizures (PNES) derived from structural neuroimaging studies of patients with PNES and other functional neurological disorders/conversion disorders (FND/CDs) and possible connections between those nodes derived from functional and structural connectivity studies. Colors indicate from which studies the connections were derived: BLUE = resting state connectivity; BLACK = structural findings/structural connectivity (dashed line around vAI is derived from the meta-analysis of some of the neuroimaging studies of PNES; 6); ORANGE = task-related functional connectivity; DASHED BLUE = connectivity noted in other FNDs/CDs, in addition to the connectivity in PNES; DASHED ORANGE = task-related functional connectivity (various tasks).
The aim of this review is to provide an increased understanding of the macro- and submacroscopic neuroimaging abnormalities that are present in patients with PNES. Thus, we will explore the idea that PNES/FNDs are a network, rather than a focal disorder, with a variety of known psychological profiles (1–3) and variable clinical semiologies (4, 5), both of which may depend on which part of the network is affected. Further, we will also examine evidence in support of speculations provided in some studies regarding their findings and the relationship between the findings and the disorder itself (6).
Structural Neuroimaging Data
Several studies have examined (either specifically or as part of the investigation) the presence/incidence of structural neuroimaging abnormalities in patients with PNES. While these studies have focused mostly on “visible” abnormalities (e.g., cortical malformations, cavernous angiomas, periventricular white matter disease, or posttraumatic lesions), other studies provide additional evidence for submacroscopic abnormalities (differences in cortical thickness, cortical atrophy, or changes in the integrity of the white matter tracts). Overall, it is widely accepted and expected that structural imaging in PNES should be—but does not need to be—normal. Moreover, it is well established that the presence of seemingly epileptogenic abnormalities in patients with new onset or ongoing seizures does not necessarily predict that the patient has or will develop epilepsy (7, 8).
Lesion Studies
While early epidemiologic studies of PNES did not report specifically on imaging findings (9, 10), other studies have found that up to 40% of patients with PNES have structural abnormalities on routine MRIs (11–14). Thus, while only a few studies have specifically described MRI abnormalities, it is clear that structural imaging abnormalities are prevalent in patients with PNES and may have negative implications for outcomes, despite the fact that the lesions are not the direct cause of PNES (12). One of the earlier studies found significantly higher prevalence of nondominant hemispheric lesions in patients with PNES (15). The finding of the nondominant hemispheric structural abnormalities, combined with the notion that regulation of certain emotions is predominantly right-hemispheric, has led Devinsky and colleagues to hypothesize that this combination may be facilitating the development of conversion symptomatology(15).
Volumetric Structural Analyses
MRI studies have also utilized volumetric analyses of regions of interest to identify structural abnormalities in patients with PNES. The presence of the aforementioned right-hemispheric asymmetry in cerebral pathology in PNES was recently supported by another study that showed rightwards asymmetry of the uncinate fasciculus (UF; structural connectivity) in PNES and postulated a relationship between this finding and the symptoms of depression and anxiety that are frequently present in this population (3, 16, 17). Further, this study showed an association between the age at PNES onset and the degree of UF asymmetry (16). However, this study did not examine the relationship between UF asymmetry and any other neuropsychiatric findings.
Another study documented decreased cortical thickness in the right motor and premotor regions. Here, also, while a relationship between cortical thickness and depressed mood was observed, a comparison to controls with similar mood disorders was not performed, questioning whether the findings are related to depression or PNES (18). Other studies did not confirm the rightwards structural findings (11, 19). In fact, studies have documented that at least some of the structural findings are present more so in the left than in the right hemisphere. For example, one study documented widespread bilateral cortical thickness, surface area, and sulcal depth abnormalities in areas involved in emotion processing, including the insula, orbitofrontal, and precentral regions (20). In this particular study, in addition to various positive and negative correlations between right medial orbitofrontal cortical thickness and neuropsychological tests, the authors observed increased thickness of the left inferior (ventral) anterior insula (20). A few studies that involved not only patients with PNES but also other FND semiologies (e.g., functional motor disorders [FMDs]) described structural differences between patients and controls that appear to be common across FNDs. For example, one study described reduced cortical thickness of the left anterior cingulate in patients versus controls, while within-group analyses showed positive correlations between personality trait testing and anterior cingulate and right lateral cortical thickness (21). Another study found inverse associations between left insular volume and functional neurological symptoms with childhood abuse burden in women (22). Also in this study, when men were included in the analyses, associations were noted between the symptoms of PTSD and anterior cingulate volume (22). Finally, one report linked, in univariate analyses, increasing hippocampal gray matter volume and 6-month mental health outcome in patients with FNDs (predominantly functional [psychogenic] movement disorders, PNES, and functional weakness); these correlations did not survive correction for anxiety and life events scores (23).
Diffusion Tensor Imaging
A few neuroimaging studies examined structural connectivity among brain regions. A diffusion tensor imaging (DTI) study showed predominantly left-hemispheric white matter tract changes connecting left UF, left superior temporal gyrus (STG), and left subcortical structures (24). The previously mentioned study (16) documented asymmetry in UF in patients with PNES compared to controls that was related to the age of onset. Another DTI study described changes in small-worldness in PNES when compared to controls with a shift towards more regular organization of the network specifically in the attention, sensorimotor, subcortical, and default-mode networks (25). Finally, a recent meta-analysis indicated that the area that may be semi-consistent between some studies is a specific area in the left anterior inferior insula—labeled in the study more broadly as left temporal lobe (6). The findings from the last study are in agreement with a study in patients with FNDs that found reduced left anterior insula volume in volumetric analysis in patients who reported the most severe impairment on SF-36; the authors also observed a relationship between reduced amygdala volumes and reduced mental health (26). These seemingly inconsistent and incongruent findings suggest that the structural abnormalities in PNES may be multifocal and, in conjunction with the variable semiological presentations of the PNES, indicate that abnormalities in various areas of the brain may result in similar clinical phenotypes. This notion is supported by the recent finding of higher incidence of multifocal neuroimaging findings in PNES versus epilepsy (11).
Neurometabolic Studies
Several metabolic studies in patients with PNES support the aforementioned multifocal structural findings. One single photon emission computed tomography (SPECT) study documented brain hypoperfusion in 3 of 11 participants, one of each: bilateral parietal, right temporal, or right hemispheric (27). Another SPECT study showed similarly multifocal abnormalities in 3 out of 10 patients with PNES (28). Finally, in a more recent SPECT study, 2 of 3 participants showed subtraction ictal SPECT co-registered to MRI (SISCOM) hypoperfusion in 1 participant in the left insula and in the right insula and lateral frontal lobe in the second participant (29). Overall, interictal and ictal SPECT findings in over 100 patients (some with comorbid epilepsy) included in the above and other studies indicate that 10 to 20 percent may have focal hypoperfusion. However, a single cerebral structure with ictal hypoperfusion that correlates with the PNES onset location has not been identified (29).
In contrast to SPECT studies where comparisons have been made to patients with epilepsy, a recent PET study compared patients with PNES to healthy controls to identify two areas of hypometabolism: right inferior parietal and central region, and bilateral anterior cingulate (30). The consistent finding from these studies is that the metabolic abnormalities are present in the minority of investigated patients in various brain areas and not necessarily in a single specific brain region. Nevertheless, many of the studies indicate involvement of brain regions that are part of, or are responsible for, emotion processing and motor control, both of which are postulated to be involved in initiation and maintenance of PNES. To the best of our knowledge, in none of these studies was correlation with neuropsychiatric variables performed.
Functional MRI Task Data
The available functional imaging data are even more complicated and difficult to interpret. However, clarity starts to emerge, as some of the studies correlate neuropsychiatric variables with neuroimaging results. The fMRI studies have been largely conducted using task (fMRI) and resting state fMRI (rs-fMRI), and their results are schematically depicted in the Figure, where the at present identified nodes of the network are derived from the structural studies and the connections between the nodes are derived from the structural and functional connectivity studies.
One study did not find any differences between healthy controls and patients with PNES for either one of two fMRI tasks: encoding of pictures with high sentimental value and Stroop color naming (testing for dissociation; 31). Of interest, these authors reported that their entire sample of 11 patients with PNES was free of any comorbid psychopathology, which occurs in less than 5% of PNES populations (31). In this study, the dissociation scale scores were correlated with rs-fMRI (see following). Other fMRI studies examined activation patterns in PNES using various versions of the emotion processing task (called “valence task” in some studies). One study compared the involvement of the emotion-processing circuits (emotional faces fMRI task) in PNES to matched healthy controls to show differences in responses to various facial emotions in frontal, parietal, and motor regions as well as in cerebellar, orbitofrontal/frontopolar, and insular regions (32)
Since there are currently very few fMRI studies in PNES, we supplemented the PNES data by reviewing activation patterns from other FND populations, with the understanding that FMDs (functional atasia-abasia, etc.) and FNDs/CDs (including PNES) are different semiologic expressions of the same psychopathological somatoform disturbance (33–36). In one study of patients with FMDs, the facial emotion processing fMRI task showed increased response in amygdala in FMDs compared to controls (37). In another study, the same authors showed that an action selection task activates the motor and emotion circuit differentially among patients with various FMDs and healthy controls (38). Finally, the same group analyzed data focusing on motor initiation in patients with FMD to document hypoactivation in the temporoparietal junction (TPJ), suggestive of deficits in multisensory integration in FMDs (39). In two other FMD fMRI activation studies, the authors compared patients with well-defined psychogenic dystonia or psychogenic tremor to controls, using emotional faces and intense emotion fMRI tasks (40, 41). In the first study, no differences were demonstrated among the groups in the activation patterns for standard motor tasks, indicating that the motor generators involved in the production of voluntary movement are the same in functional dystonia and healthy controls. They documented differential activations, however, in temporal gyrus, precuneus and inferior frontal gyrus with the faces task, while the intense emotion task also induced functional activation differences in insula, motor cortex, and fusiform gyrus (40). In the second FMDs study, patients with psychogenic tremor had differential activations in response to the emotional faces task in the para-cingulate and left Heschl's gyrus, while there were no differences in the intense emotion task between the groups (41). Finally, two other FMD studies need to be mentioned briefly (42, 43): In the first study, the authors showed that motor inhibition in patients with FMDs is mediated by the inferior frontal gyrus with extension to the anterior insula, with clear differences between patients with FMDs and feigning subjects, especially in the medial prefrontal regions (42). The second study, using a combination of passive movement and emotional stimulation, resulted in amygdala hyperactivity, while connectivity analysis showed increased interaction between the amygdala and subthalamic nucleus/SMA (43). These studies document both similarities and differences between specific brain regions’ involvement among various FNDs. Unfortunately, in most of the FNDs studies, correlations with neuropsychiatric measures or comparisons to participants matched for psychopathology are performed infrequently, leaving the psychopathological inferences of some of the studies speculative.
Functional Connectivity Data
Functional connectivity analyses provide some additional and necessary insights into alterations of the brain networks associated with PNES. Several studies have been conducted to date in patients with PNES, so studies in other FNDs are discussed only briefly. Four studies published on the same group of subjects addressed various aspects of resting state connectivity; although Structured Clinical Interview for DSM-IV (SCID) was conducted in the participants to exclude active psychopathology, correlations of comorbidities with neuroimaging were not performed. The first study assessed neural connectivity by the means of functional connectivity density mapping (FCDM) to show that, in comparison to 20 healthy controls, 18 patients with PNES had abnormal FCDM in frontal, sensorimotor, and occipital cortex in addition to insula and occipital cortex (44). In the second study, the authors combined DTI and rs-fMRI, to analyze connectivity differences between PNES and controls using graph theory (25, 45). As noted, the patients with PNES exhibited altered small-worldness in the structural and functional networks and altered nodal characteristics, which were thought to be related to the unstable cognitive–emotional and motor systems in PNES. Their third study duplicated the above results, performing a third type of analysis: fractional amplitude of low-frequency fluctuations (fALFF; 46). In the final manuscript, they examined the connectivity of the insula after dividing it into three subregions (ventral anterior insula [vAI], dorsal anterior insula [dAI], and posterior insula [PI]; 47). Interestingly, they found differential connectivity patterns of insular subregions: vAI was connected to the other insula, cingulate, SMA, and frontal and temporal gyri; dAI was connected with the other insular subregions as well as cingulate, precentral and supramarginal gyri, and parietal region; the PI was connected with the other insula, as well as cingulate and STG (47).
Overall, the findings of these four studies indicate altered functional connectivity within various parts of the networks that may be involved in PNES generation and maintenance. These network regions include executive control, frontoparietal, sensorimotor, and default mode networks that were shown to be significantly associated with dissociation symptomatology in one PNES study (31, 48). This study (31) supports the notion that while insula may be very involved in this generation and maintenance of PNES, the participation of its various parts may reflect their functional separation (49). Another study indicated differences in connectivity between specific brain regions between PNES and controls—inferior temporal gyrus and parahippocampal gyrus/uncus, between parahippocampal gyrus/uncus and middle temporal gyrus, and between STG and paracentral regions (32). Of importance is that the previously mentioned meta-analysis of all available neuroimaging studies of PNES points towards anterior inferior insula as the potential common structural and functional substrate of PNES (6)
The findings in PNES correspond well with findings from other FND fMRI studies. For example, one study identified increased connectivity and directionality of the information flow from the right amygdala to the supplementary motor cortex (SMC), indicating the possibility of greater interaction between these structures in FMDs and a mechanism for an upstream control and modulation of motor activity (37). Another study of life events in patients with CDs indicated differences in DLPFC and hippocampal connectivity during the performance of an escape task that was associated with increased fMRI activity in the TPJ and SMA (50). Other previously mentioned studies in patients with FMDs indicated changes in connectivity between left amygdala and left middle frontal gyrus (41)
Conclusion
In summary, the findings from a number of neuroimaging studies indicate that the submacroscopic abnormalities present in patients with PNES/FNDs are multifocal and, in some cases, associated with specific neuropsychiatric findings. The overlap among the networks involved in the generation and maintenance of PNES and other FNDs indicates that FNDs in general (and PNES in particular) are network disorders, and the symptoms associated with the disruption of various parts of the networks may result in the observed variation in the clinical symptoms (51). Unfortunately, the quality of the available studies specific to PNES is overall low, with very few studies examining the neuropsychiatric findings in conjunction with the neuroimaging findings or controlling for the neuropsychiatric abnormalities that are so prevalent in patients with PNES/FNDs.
Another major drawback of the currently available studies is the small sample size that does not allow comparison of patients with PNES/FNDs to controls matched for neuropsychiatric conditions. Finally, approaches to the investigations vary among centers and investigators and depend on availability of subjects and expertise in methods used to analyze the data.
To overcome these limitations, large studies are needed that specifically control for neuropsychiatric comorbidities. Further, the experiences of the seizures in patients with PNES are in many ways similar to experiences of patients with epilepsy (52, 53). Thus, comparisons to patients with epilepsy, such as the one recently performed (32), are needed to better delineate the network abnormalities that are specific to PNES and where an overlap with neuroanatomically based brain disorders—including multifocal, such as traumatic brain injury—is present. While treatments for seizures exist (54), understanding the functional neuroanatomy of PNES may, in turn, provide better targets for interventions for these patients and allow a more personalized approach.
