Abstract
The holy grail of epilepsy research is to understand the mechanisms underlying seizures so that patients with epilepsy can receive effective treatment or be cured, ideally with no significant side effects. Recent advances in neuroscience give such hope. Optogenetics is a modern neuroscience research tool that allows precise spatiotemporal control of defined cells and circuits and, thus, dissection of critical players and targeting them for responsive treatments. Here we review the state of the art of these approaches and their applications and implications in epilepsy research.
Opsinology
History: Where Do Opsins Come From?
The idea of using light to control neurons was first suggested a little more than a decade ago (1). These researchers developed an integrated system of the ingredients required to render neurons sensitive to light: rhodopsin, β-arrestin, and a G protein α subunit. When they expressed this combination of proteins in hippocampal cultured neurons, they found that exposing the culture to light reversibly activated neurons over several seconds, consistent with the slow time course of G protein-mediated activation of downstream ion channels. In a prescient commentary, they further pointed out how this general approach could be combined with genetic targeting to activate specific classes of neurons, based on specific promoter sequences. This goal would fulfill the dream set forth by Francis Crick (2), two decades earlier, of manipulating specific neurons while leaving others unaffected.
A year after Zemelman's report, Nagel et al. (3) demonstrated that an algal opsin from Chlamydomonas reinhardtii, channelrhodopsin2, would directly gate cationic (positive) current into cells, causing them to depolarize. This exciting advance meant that light could be used to rapidly modulate the activity of cells and, potentially, neurons. This potential was fulfilled in just 2 years when Nagel et al. (3) joined forces with Ed Boyden and Karl Deisseroth and colleagues (4) and showed that neurons indeed could be made to express this algal protein and that, when so expressed, they became sensitive to blue light: laser light pulses could directly, rapidly, and reversibly trigger neuronal action potentials! Two years later, members of this group (5) greatly expanded this concept with the description of halorhodopsin as a protein that could be expressed in neurons to rapidly and reversibly inhibit neural activity by activation of a membrane Cl− pump with exposure to yellow light. These pioneering studies established the proof of concept of real time, genetically-targeted, bidirectional optogenetic control of neurons.
Opto Big Bang: From One Opsin to Many
In the decade since the demonstration of rapid, real-time control of neurons by Boyden et al. (4), the field has exploded. A PubMed search for the term optogenetics in August 2014 yielded more than 800 citations, with more than two-thirds of these in the last 18 months. This is a rapidly evolving field. Many laboratories in the United States and overseas are working to develop new opsins, either from novel organisms or engineered from known opsins, noted above. New opsins extend the range of functionality of these light-sensitive proteins and enable an unprecedented level of control of neural circuits. Applications of these ever-evolving tools have been recently reviewed, and we refer readers interested in the implementation details to (6, 7).
For our purposes here, we will note that many different optogenetic proteins have been engineered, allowing for several interesting possibilities. 1) opsins with different excitation spectra allow independent control of two different circuit elements (8, 9), and thus, different circuit pathways and their intersections can be studied, 2) opsins are available with different activation times. Some, such as ChIEF, have extremely rapid kinetics that allow near millisecond level control of neural activity (10), 3) By contrast, step-function opsins (11) are more like bistable switches. Once activated by light, they continue to conduct current (and activate neurons) for extended periods, up to half an hour (5). These step-function opsins potentially allow for network mode-changing. For example, in thalamocortical networks, the recurrent oscillatory network activity underlying generalized absence seizures relies on periodic synaptic hyperpolarizations and the resultant rebound burst discharges, of thalamocortical relay neurons. A step function opsin that produced a persistent depolarization would switch the mode of thalamocortical cells by preventing hyperpolarization and subsequent rebound bursting, thus suppressing absence seizures. Similarly, small persistent membrane depolarizations in a population of neurons may render them responsive to inputs that would otherwise be insufficient to drive spiking. Thus, whole new populations of neurons could be recruited into networks with resultant enhancement of circuit responsiveness. Most recently, new versions of ChR2 have been cleverly engineered to conduct anions rather than cations (12, 13). These new opsins have the advantages of strong expression in neurons, and they do not activate Cl− or H+ pumps, which can have bystander effects due to altered ionic homeostasis as in the case of halorhodopsin (5) and Arch (14).
Doctor, Which Opsin Should I Take?
The rapid proliferation of opsins and development of strategies for implementation (Figures 1 and 2) have created new opportunities for causal dissection of circuits in epilepsy. Here, we outline several examples of application of optogenetic approaches to understand and control epileptic seizures.

A wide range of opsins for manipulating neural activity is already available. These come in a variety of colors (excitation spectra), types (inhibitory, excitatory, modulatory), and durations (from milliseconds to hours). Selection of the proper opsin and expression in the correct neuron subtype may lead to personalized optogenetic treatments in epilepsy. Drawing by Benedicte Rossi.
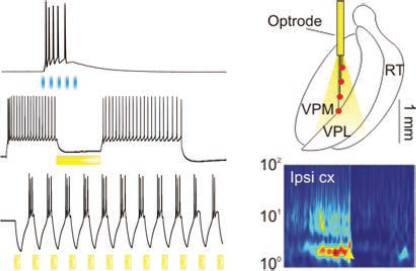
Control of neural activity requires careful consideration of the combination of the most appropriate opsin with the optimal illumination protocol. Left upper trace: Blue light depolarizes ChR2-expressing corticothalamic axons to evoke excitatory synaptic currents in a thalamic reticular neuron and trigger firing. Left middle trace: Yellow light hyperpolarizes an eNpHR-expressing thalamic relay neuron and silences ongoing firing. Left lower trace: Somewhat paradoxically, repeated yellow light pulses drive robust high frequency rebound bursts of action potentials at the offset of each light pulse. Right panel, upper: Diagram of an optrode device implanted in the ventroposteromedial thalamus (VPM) that allows delivery of yellow light to eNpHR-expressing thalamic relay neurons. This approach was effective in aborting post-stroke epileptic seizures (right, bottom, modified from 21).
Opsin Approach Leads to Discovery of a New Mechanism of Seizure Generation
The corticothalamic circuit underlying generalized absence epilepsy is a complex network comprising short and long loops of recurrent synaptic connectivity (15). Studies of this type of network with traditional methods, such as conventional electrical stimulation, have yielded ambiguous results as the stimuli nonspecifically activate heterogeneous groups of fiber projections, including cortical and thalamic axons. Optogenetic studies in mice lacking the gria4 gene, encoding GluA4 AMPA receptors, led to the discovery of a new mode of oscillation generation and seizures in corticothalamocortical circuits (16). This circuit dysfunction resulted from a specific defect in the excitatory synapses connecting corticothalamic and GABAergic reticular thalamic neurons. Loss of cortically-originating feed-forward inhibition led to overexcitation of thalamic relay cells, which in this mutant epileptic mouse had normal corticothalamic synaptic inputs. Because of the known specificity of the optogenetic approach, we could specifically activate and test the efficacy of cortical output in recruiting thalamic feedforward inhibition, thereby identifying unambiguously which synaptic components in this complex circuit were affected. Identification of a specific circuit defect offers the potential of modifying that particular circuit as a therapeutic approach. This possibility is discussed in the following sections.
Discovery of Key Targets for Preventing Ictogenesis
In addition to dissecting seizure initiation points in the brain, optogenetic approaches have also been used to determine if severe status epilepticus could be delayed or modified. In a recent study, Sukhotinsky and colleagues (17) used the inhibitory opsin enhanced halorhodopsin (eNpHR) to test the role of hippocampal excitatory neurons in the lithiumpilocarpine model of acute evoked seizures in awake behaving rats. Optical inhibition of eNpHR-expressing hippocampal pyramidal neurons before seizure onset almost doubled the time between pilocarpine injection and onset of seizures, and seizure evolution and intensity were diminished. These findings reveal an essential role of hippocampal excitatory neurons in this model of ictogenesis.
Discovery of Key Circuits (and Cells) for Seizure Control
Three recent studies with optogenetics in different models of epilepsy allowed research groups to pinpoint key cells to target for controlling seizures.
Post-Stroke Epilepsy
Our recent findings build on an extensive existing literature of global networks regulating seizure susceptibility (e.g., 18, 19). This work indicates that one might target brain regions for seizure therapy different than the initial focus of seizure initiation. In a rat model of cortical photothrombotic stroke with resultant post-stroke epilepsy (20), we showed that thalamocortical neurons that project to the peri-infarct cortex undergo changes that are secondary to the cortical injury and lead to thalamic circuit hyperexcitability. This finding raised the possibility that thalamocortical neurons in this structure remote from cortex contributes to the seizures. Notably, targeting these hyperexcitable neurons with inhibitory opsin eNpHR3.0 and reducing their activity during automatically-detected seizures were sufficient to abort electrographic and behavioral seizures. More interestingly, this seizure reduction occurred without apparent adverse effects (21). This anti-epileptic efficiency of thalamic targeting is intriguing, as it shows that structures remote from the initial injury can be targeted to regulate seizures. This strategy has several advantages: It does not directly affect eloquent cortex, and the periods of thalamic inhibition required for seizure control can be quite brief (i.e., less than 1 second), suggesting minimal disruption of normal cognitive function. Thus, the thalamus or other relatively small and easily targeted subcortical structures might be optogenetically activated or inactivated, potentially in real time, to treat epilepsies resulting from cortical injuries and malformations—epilepsies that are often intractable to medical therapy.
Temporal Lobe Epilepsy
Proof of concept of seizure control in the temporal lobe epileptic circuit was first demonstrated in hippocampal slice cultures, in which eNpHR expression was driven in excitatory neurons with a viral construct containing a promoter of Ca2+/calmodulin-dependent protein kinase IIα (22). Similar constructs have been exploited in a variety of brain regions to specifically target excitatory neurons. Tonneson and coworkers (22) found that inhibiting halorhodopsin expressing excitatory cells with yellow light would disrupt epileptiform bursting induced by electrical stimulation in cultured slices, demonstrating the potential of this approach to modify aberrant electrical activity of seizures.
This potential was realized recently. Ivan Soltesz's laboratory demonstrated that targeting multiple specific neuronal cell types can effectively reduce seizures in a model of severe epilepsy induced by intrahippocampal injection of the excitotoxin kainic acid (23, 24). The resulting post-status epilepticus temporal lobe seizures range from mild to severe and are quite variable, creating challenges for seizure detection and control. Nevertheless, real-time detection, using software now available in the public domain, was coupled with optogenetic control to reduce seizure duration and severity. Seizure control could be obtained by targeting and activating parvalbumin-containing hippocampal interneurons with ChR2 or by inhibiting eNpHR-expressing hippocampal excitatory neurons.
Neocortical Epilepsy
Recent studies also show the efficacy of optogenetic and related approaches in models of focal cortical epilepsy. Focal injections of tetanus toxin in the brain produce chronic seizures (25), with injections into cortex producing focal cortical epilepsy (26). The latter approach was coupled with simultaneous injection of viruses containing one of two activity-modifying therapeutic agents, eNpHR2.0 or a voltage-gated potassium channel Kv1.1 (27). Each of the viral approaches was designed to infect local excitatory neurons in the region of the epileptogenic insult with an exogenous protein that would suppress excitability. Resulting spontaneous seizures and epileptiform responses were suppressed by each of these approaches, either through constitutive expression (with Kv1.1), or in a controlled fashion, with yellow light (with eNpHR) (27). A recent paper reported on a complementary strategy to treat focal cortical epilepsy (28), based on the so-called Designer Receptor Exclusively Activated by a Designer Drug (DREADD) approach (29). Local excitatory cortical neurons were driven to express a modified cholinergic receptor that was engineered to be unresponsive to the native agonist, acetylcholine (ACh). Then acute focal cortical seizures were induced in the same cortical region by injection of one of two chemoconvulstants, picrotoxin or pilocarpine. Systemic administration of clozapine-N-oxide (CNO), which can activate the exogenous, engineered ACh receptor expressed in excitatory neurons in the cortical focus, but not native ACh receptors expressed throughout the brain, suppressed seizures induced by either acute chemoconvulsant (28). The effectiveness of these studies suggests that, in general, gene therapy might be especially effective in well-localized focal epilepsies in which the region of dysfunction can be directly targeted. The Wykes study (27), in particular, shows that optogenetics can be effective when targeted at inhibition of excitatory neurons.
These early successes in controlling seizures suggest future therapeutic avenues in which the activity of specific types of neurons in key epileptic network nodes can be targeted for novel interventions. The key steps are identification of the nodes, next delineation of the critically involved neurons, then gene therapy to introduce activity modifiers specifically into those neurons, and finally delivery of activating agents (light or designer drugs) to those neurons at the appropriate time.
Mind the Gap: Experimental Considerations While Using Opsins
Optogentic tools are very powerful. They allow researchers to determine causality between changes in activity of specific neurons and neural network and behavioral states. At the same time, appropriate controls are critical, and specificity, efficacy, and selectivity all need to be demonstrated. Otherwise, what appear to be promising results might be incorrect or misleading.
Specificity
It is critical to ensure that the observed effect is specifically mediated by the opsin activation. This novel approach requires delivery of high-intensity light into the brain, and little is known about potential off-target effects, such as local heating, which can alter neuronal firing or induce tissue damage. To exclude this and other potential nonspecific effects, appropriate controls must be used, including those in which control viral constructs with either no opsin or an inactive opsin are expressed in the cells. This experiment allows determination of optimal parameters of optical stimulation (power, wavelength, duration, and pattern) that can be used to obtain opsin-specific effects.
Efficacy
It is also important to quantify the effect of the light on neuronal firing. For example, ChR2 and stabilized step-function opsin (SSFO) are used to activate cells (i.e., increase their firing rates). However, if the light intensity is too strong, the effect might be opposite of that desired, that is, inducing a depolarization block and resultant cessation in firing activity. Without recording the effect of the light on the firing of the cells, one might assume that the target cell is activated when, in reality, its firing is inhibited. Similarly, when using the inhibitory opsins, it is important to ensure that the light protocol is effective at reducing the firing rate of the opsin-expressing cells. These firing characterization studies can also lead to better quantification and understanding of the degree of change in firing required to produce the desired network/behavioral effect. In some cases, a change in the firing pattern might be desired and should be tested. For example, a change from bursting to tonic firing or vice versa might produce a robust effect on seizures even in the absence of a change in the mean firing rate. Therefore, fine quantification of the spiking is required to develop the “minimal” intervention methods to stop seizures without unwanted effects; this might be obtained with a relatively weak light intensity that would switch firing patterns (by mildly altering membrane potential) without dramatically reducing or activating neuronal firing.
Selectivity
A final control is to ensure that the targeting and effects are cell specific such that only the intended neuronal population is directly affected in the intended manner. Since opsins exert their neural actions through activation of ion channels or membrane ion pumps, there is the potential for bystander effects in which ion accumulation or depletion occurs in the intracellular or extracellular space. This issue is especially important for Cl pumps, such as halorhodopsin, as effects on intracellular [Cl] could have dramatic effects on synaptic inhibition, which relies on a stable Cl gradient.
Conclusion and Future Directions: Opsinophilia versus Opsinophobia
Optogenetic tools offer tremendous advantages for understanding complex networks and potential critical choke points. Therefore, these tools are especially applicable to the study of epilepsies, which represent network dysfunction of focal or generalized origin.
Newly engineered opsins are continually being developed to optimize their expression in different types of membranes (some to target axons, others to target somata), avoid cell toxicity, and enhance cell specificity, as well as enhance target-specific expression in subsets of neurons. Recently developed approaches include combinatorial opsins that allow for targeting different cell types that can be controlled simultaneously and independently. These abilities should improve optical approaches that specifically and effectively control seizures (e.g., by enhancing the activity of inhibitory while reducing the activity of excitatory cells). The feasibility of such an approach was recently demonstrated in cortex and thalamus by co-expression of distinct opsins in different neuronal classes that can be independently and simultaneously controlled with different wavelengths (8).
Beyond the advantages of optogenetics in understanding and treating dysfunctional circuit dynamics in animal models, can this approach be applied to human patients with epilepsy? So far, optogenetic methods have used viral constructs to express foreign protein in neurons. Such approaches based on gene therapy might lead to lifelong changes in the brain; therefore, long-term safety and efficacy must be determined to ensure the stability of opsin-expression for efficacy and to avoid toxicity that might result from foreign protein expression.
