Abstract
Optical coherence tomography (OCT) imaging plays an important role in the management of retinal diseases. However, limitations and pitfalls should be taken into consideration when applying this noninvasive imaging technology. The aim of this review is to discuss several possible sources of error in the conduct and interpretation of OCT imaging.
Introduction
Retinal imaging is key for diagnosis, therapy, and management of patients with macular and retinal diseases. Most importantly, during the last decade, optical coherence tomography (OCT) imaging has revolutionized diagnostics and treatment of patients with diseases of the posterior pole, including intravitreal application of pharmaceuticals as well as surgical interventions.
Acquisition of OCT images is a fast, noninvasive procedure that is relatively comfortable for patients as well as for the operating examiner. However, there are several limitations and pitfalls in the conduct and interpretation of OCT imaging. This article aims to add to a meaningful and focused use of this imaging modality in clinical practice.
Pitfalls in OCT Image Acquisition
Several potential sources of error during acquisition of OCT images exist. Usually, OCT images are not recorded by the evaluating ophthalmologist. Accordingly, accurate training, guidance, monitoring, and quality control of the medical-technical staff is important.
Acquisition Protocol
The fast acquisition time of OCT images with modern devices has resulted in broad application of this imaging technology in clinical practice (1). Increasing acquisition of dense OCT volume scans, however, has also raised new questions: How can one evaluate all these scans carefully? Do denser scans provide more important diagnostic information? How can one handle the increasing data volumes in terms of processing and storage with existing computer and network systems?
In part, these challenges can be addressed by reasonable application of distinct scan protocols for specific diseases and symptoms. In exudative macular diseases (i.e., various forms of choroidal neovascularization [CNV]), volume scans have the advantage of dividing the central retina into equal proportions in order to identify fluid also outside the foveal center. On the other hand, in diseases of the vitreoretinal interface, the fovea and the optic nerve head are most important, while the representation of eccentrically located retinal spaces within the macula is less important. These requirements for the density of the scanning field are achievable with the star scan. In contrast to the volume scan, the star scan gives a more precise representation of a circumscribed retinal area where the scans intersect (i.e., the fovea), while less dense information is acquired from eccentric areas (2) (Fig. 1). In unclear situations, the option of re-recording with a more detailed and suitable scan protocol in individual cases should be considered.
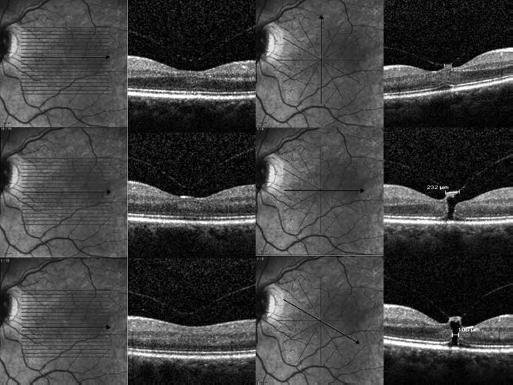
Application of the 19-line volume scan (left panels) versus a star scan protocol (right panels) in an eye with vitreomacular traction and full-thickness macular hole. Note that the 3 central B-scans in the volume scan fail to detect the relevant pathologic findings.
For diseases of the optic nerve, an adequate scanning protocol must also be applied.
In glaucomatous diseases, peripapillary circular scans are used to determine the thickness of the retinal nerve fiber layer, or papillary centric star scans for the analysis of Bruch membrane opening at the margin of the papilla.
To assess papillary swelling, however, a volume scan that is centered on the optic disc represents a suitable recording protocol (3).
Acquisition Technique in General
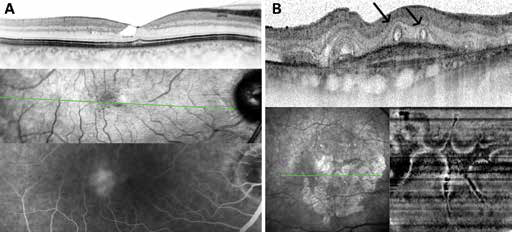
The optimal alignment of the camera with even illumination of the entire en face image and the setting of further parameters such as focus and length of the scan arm are important prerequisites for high-quality OCT imaging. Furthermore, a sufficient signal quality (signal strength) is required. Incorrect settings should be identified and if necessary a reacquisition should be initiated (Fig. 2A).

A basic evaluation of the scan quality should be performed in a first step when beginning the interpretation. In addition, the technical staff should not only be adequately trained, but also instructed to check the quality and completeness of the data directly after the recordings; at this time point, the patient is still in front of the device and there is the possibility of immediate repeat recording of images with the aim of better quality (Fig. 2B). For example, incorrect application of the recording technique can lead to a finding outside normal limits due to a lack of knowledge/training if the definition of the center of the fovea is incorrectly set when acquiring the papillary Bruch membrane opening scan (Fig. 2A).
Using Different OCT Devices
No common industry standard has been established for OCT imaging. There is no standardization regarding limits for the automatic determination of retinal thickness or algorithms for the determination of the fovea-papilla axis for the alignment of scans in the context of glaucoma diagnostics. In addition, device-dependent differences in the appearance of retinal thickness may also occur, since OCT B scans are usually displayed as stretched images in the vertical direction (see also discussion on quantitative evaluation).
Accordingly, for better comparability, the same patient should be examined with the same device platform. It should also be noted that, by software updates, the algorithms and definitions of the automatic segmentation lines may change and therefore the comparability of subsequent recordings and their evaluation can be limited.
Using the follow-up Mode
Automatic registration and matching of OCT images of the same retinal location is an essential tool for assessment in the monitoring of subtle changes over time. In order to avoid artefacts or inaccuracies, it is advisable to keep the same focus between different imaging sessions and to avoid any tilting of the head in the chin rest during image acquisition. In the context of a systematic analysis, it was found that tilting of the head in the chin rest can lead to an incorrect determination of the thickness of the peripapillary nerve fiber layer or retina (4).
Pitfalls in OCT Interpretation
Viewing OCT Images
In general, printouts allow only a rough overview of OCT findings. These provide an orientation, but are not suitable for an adequate evaluation of modern OCT imaging. This should also be taken into account in quality control by a third party. In contrast, viewing the collected data on the monitor offers the possibility to evaluate all collected B-scans individually. Evaluation of the foveal or other single scans, for example, would not be sufficient to determine disease activity under anti-vascular endothelial growth factor (VEGF) treatment for exudative age-related macular degeneration (AMD) (Fig. 3).

Choroidal neovascularization (CNV) under anti-vascular endothelial growth factor therapy. Evaluation of only the central B-scan for activity of the CNV lesion
Focus on Relevant Findings
With an increasing number of B scans within a raster, focusing on the relevant findings may be challenging. Herein, precise knowledge of retinal and macular diseases appears to be mandatory. In addition, the implicit learning in the sense of pattern recognition plays an important role in image evaluation. A multitude of typical OCT findings has been described in individual retinal and macular diseases; e.g., differentiation of primary diseases that are complicated by CNV can be supported by recognition of characteristic OCT findings: dome-shaped elevations of the retinal pigment epithelium (RPE) in the presence of soft drusen indicate AMD as primary disease; marked thickening of the choroid in OCT images without soft drusen would point to central serous retinopathy as primary disease.
Artifacts
With spectral-domain OCT (SD-OCT), in contrast to the older time-domain OCT technology, motion artifacts are much less frequent. However, even cooperative patients cannot suppress microsaccades. Especially in complex scanning protocols, these subtle inevitable eye movements may lead to visible deviation of single scans within a raster scan. Application of the so-called fundus tracking (registration of eye movements) accounts for these movements and may overcome these artifacts. Compared to structural SD-OCT imaging, motion artifacts are playing an increasingly important role in the recently introduced OCT angiography (5, 6).
However, applying fundus tracking does not ensure exact pixel-to-pixel accuracy of the en face image with the OCT scan, but is subject to some tolerance. This tolerance is usually even greater when registering previous OCT images for follow-up scans (7–9). Systematic analyses, also on different devices from the same manufacturer, showed deviations of up to 35 μm (8).
Although these changes are not relevant to most clinical questions, they should be taken into account in the assessment of very subtle changes, e.g., reticular pseudodrusen (7, 9).
Hyperreflective changes in the vitreous (e.g., floaters) or on the surface of the retina (e.g., epiretinal membranes) may lead to projection artifacts to the point of complete suppression of structures in deeper retinal layers. Herein, comparison with en face modalities or ophthalmoscopy is helpful.
Particularly when applying automatic analysis algorithms (e.g., automated calculation of the thickness of the peripapillary nerve fiber layer), projection artifacts have to be considered.
Therefore, the investigator should evaluate the segmentation in each B scan and, if necessary, correct manually. Possible reasons for false segmentation are larger blood vessels at the rim of the optic disc or changes of the nerve fiber layer due to a posterior vitreous detachment.
Because of the lack of reflectivity in the perpendicular laser beam, the parafoveal Henle fiber layer is usually not distinguishable from the outer nuclear layer. By tilting the head in the chin rest and thus changing the direction of the laser beam in relation to the retina, the Henle fiber layer becomes hyperreflective and thus can be delimited from the still hyporeflective nuclear layer (10).
Quantitative Analysis
The quantitative evaluation of OCT findings, such as measurement of retinal thickness, individual retinal layers, or pathologic lesions, requires a precise definition of individual parameters. To date, there is no industry standard or consensus, and different terms are used in parallel such as central retinal thickness, subfoveal retinal thickness, foveal center point thickness, or foveal central subfield. The method of scaling must also be taken into account when measured values are specified in the metric system.
By applying the Early Treatment Diabetic Retinopathy Study grid to confine retinal regions, the exact centration of the overlaid grid has to be verified and—if necessary—corrected. Particularly in the presence of pathologic changes of the central retina—e.g., pigment epithelium detachments (PEDs)—the automated software algorithms may fail to find the exact position of the fovea. Therefore, positioning and segmentation of the relevant landmarks is crucial for meaningful and correct quantitative analysis by automated algorithms.
The OCT interpretation is usually based on the 1:1 pixel presentation mode, in which the image information in the lateral compared to the anteroposterior dimension is compressed approximately by 3 to 4 times depending on the device. This is because the anterior-posterior resolution of OCT imaging is much better than the lateral resolution. Therefore, for better visibility of microstructural changes, OCT images in the common viewing mode are artificially compressed in the anterior-posterior dimension. However, this compression is disadvantageous if additional manual measurements of surfaces or distances on individual B scans are made.
It was shown that the quantification of areas or distances in the 1:1 pixel presentation mode was prone for an overesti-mation of values in the anteroposterior dimension. Therefore, measurements should be performed in the 1:1 μn presentation mode (11, 12).
Inaccuracies of measurements within B scans may further occur if tilted scans are present, i.e., if the retinal layers are not orthogonal to the laser beam. This is usually due to an oblique head position or nonoptimized alignment of the camera during recording. This may be misleading when performing measurements, e.g., of the retinal thickness orthogonally to the retinal layers; however, to determine correct values, measurements should be performed parallel to the beam path.
Misinterpretation of OCT Findings
Correct interpretation of OCT findings is prerequisite for treatment decisions. Knowledge of the anatomy of the retina and its visualization on OCT images is mandatory. The exact assignment of hyperreflective and hyporeflective layers on OCT images to anatomical structures is under debate. It is recommended to apply the proposed lexicon for anatomic landmarks in normal posterior segment SD-OCT based on the consensus of the International Nomenclature for Optical Coherence Tomography Panel (13).
Subretinal Fluid vs PED
Differentiation between subretinal versus sub-RPE fluid (i.e., PED) is important for differential diagnosis and for monitoring and treatment of diseases, e.g., CNV.
Subretinal fluid is bordered by the photoreceptor outer segments and the RPE/Bruch membrane complex, while sub-RPE fluid is located between the RPE and Bruch membrane, typically characterized by a dome-shaped elevation of the RPE, termed PED.
Differential Diagnosis of Intraretinal Fluid
Hyporeflective cavities within the retina are usually interpreted as intraretinal fluid. The causes may be diverse and differential treatment decisions must be made. Disruption of the blood-retina barrier with leakage on angiography and focal retinal thickening as seen in diabetic macular edema or in neovascular AMD would be addressed by administration of antiexudative substances. However, intraretinal hyporeflective cavities in X-chromosomal retinoschisis or retinoschisis in high myopia are not caused by disruption of the blood-retina barrier but are due to a degenerative cleavage of the retinal tissue. There is no indication for treatment by antiexudative substances, i.e., anti-VEGF or steroids.
In macular telangiectasia type 2, intraretinal cystic cavities typically occur without retinal thickening, but with a thinning of the retina in the sense of degenerative changes and tissue loss (Fig. 4A). In patients with AMD, intraretinal cystic cavities may also be observed as signs of a chronic degenerative process without clinically manifest active neovascularization (14). In this situation, there is no indication for VEGF inhibitor therapy; however, there is need for continuous monitoring to recognize possible reactivation of new formation of CNV.
Furthermore, in presence of macular holes, some B scans may show cystoid cavities. If there is no B scan visualizing the full thickness defect, misinterpretation may occur. In this situation, star scans are advantageous (see also above).
Outer Retinal Tubulations
Outer retinal tubulations (ORT) have to be distinguished from intraretinal cystoid spaces. Outer retinal tubulations are tube-shaped, interconnecting formations usually located in the outer nuclear layer that may occur in a multitude of degenerative retinal diseases. The characteristic features are rounded, hyporeflective cavities, which are surrounded by a strongly hyperreflective line (15, 16) (Fig. 4B). Histologically, ORT are regarded as degenerating photoreceptors that become arranged in a circular or ovoid fashion (16). Differentiation of ORT is relevant for patient management since its presence without exudative changes is not an indication for anti-VEGF therapy.
Epiretinal Membranes vs Choroidal Folds
Epiretinal membranes in different manifestations are frequently observed in OCT imaging as hyperreflective lines internal to the inner limiting membrane and typically evoke a wave-like appearance. In this situation, the retinal surface is corrugated, while the hyperreflective RPE/Bruch membrane complex appears smooth.
If the RPE/Bruch membrane complex is likewise wave-like, this can be a sign of choroidal folds (Fig. 5).
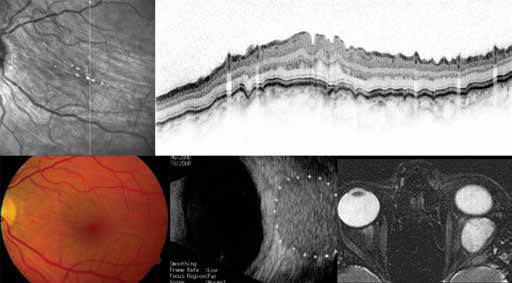
Retrobulbar hemangioma (note the retrobulbar mass on ultrasound and signal enhancement in magnetic resonance imaging) evoking choroidal folds. The wave-like pattern includes all retinal layers as visualized by optical coherence tomography imaging. Note corresponding near-infrared reflectance image and fundus photography.
Focusing only on OCT
Optical coherence tomography imaging alone should not be the only basis for treatment recommendation. Optical coherence tomography imaging should be regarded as an additional diagnostic procedure and not as a replacement of funduscopy, angiography, or other imaging modalities. Furthermore, the patients’ complaints need to be considered in decisions for surgical interventions (Fig. 6).
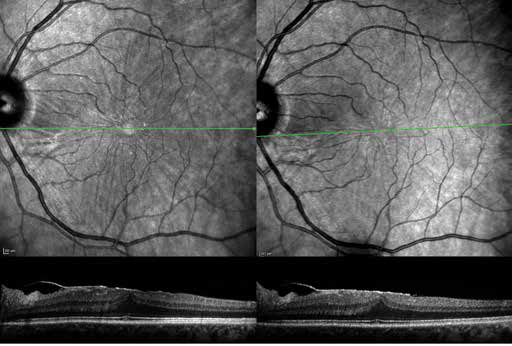
Marked epiretinal membrane on optical coherence tomography imaging. There is no progression over 1 year follow-up. Furthermore, the patient does not complain of severe visual symptoms. Therefore, surgical intervention was not performed.
Typical situations where additional diagnostic evaluation is mandatory are retinal hemorrhages since OCT devices use long-wavelength light in the near-infrared range with little or no interference in the area of bleeding (Fig. 7).
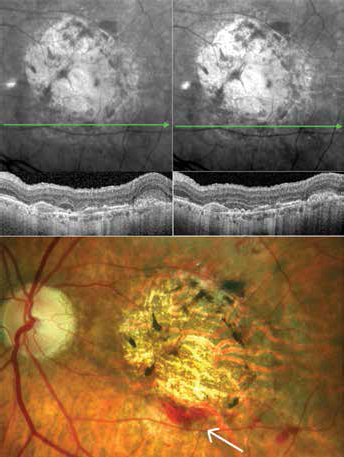
Hemorrhages (arrow on fundus photograph) are frequently not visualized by near-infrared reflection and optical coherence tomography imaging due to the wavelength used.
Furthermore, funduscopy is particularly important for monitoring patients with diabetic retinopathy or retinal vein occlusion, where evaluation of the peripheral retina is crucial.
Angiography needs to be applied to disclose disruption of the blood-retina barrier (e.g., in presence of CNV or due to inflammatory retinal diseases).
Particularly in disease of the vitreoretinal surface, the patients’ complaints are crucial for therapeutic decisions. Epiretinal membranes or vitreomacular traction may be prominent on OCT imaging although the patient does not have any symptoms. On the other hand, subtle appearing OCT findings may correlate with serious symptoms.
Of note, if the findings on OCT imaging cannot be clearly assigned or if there is an obvious discrepancy with the patients’ complaints, it is reasonable to perform further imaging and diagnostic procedures.
Previously published in German in Der Ophthalmologe (Schmitz-Valckenberg S, Brinkmann CK, Fleckenstein M, et al. [Pitfalls in retinal optical coherence tomography imaging.] Ophthalmologe 2017;114:275-290).
Footnotes
Financial support: No financial support was received for this submission.
Conflict of interest: S. Schmitz-Valckenberg: financial and nonfi-nancial support for the conduct of clinical trials by Alcon/Novartis, Allergan, Bayer, Bioeq/Formycon, Heidelberg Engineering, Optos, Roche/Genentech, Topcon, and Zeiss Meditec; consultancy of, honoraria, and travel cost support by Alcon/Novartis, Bayer, and Heidelberg Engineering. C.K. Brinkmann: financial and non-financial support for the conduct of clinical trials by Heidelberg Engineering, Newtricious, Novartis, and Zeiss Meditec; consultancy of, honoraria, and travel cost support by Heidelberg Engineering, Newtricious, and Novartis. B. Heimes: financial and nonfinancial support for the conduct of clinical trials by Novartis, Alcon, Allergan, Acucela, Roche, and Ophthotech; consultancy of, honoraria, and travel cost support by Novartis and Bayer. S. Liakopoulos: financial and nonfinancial support for the conduct of clinical trials by Heidelberg Engineering and Zeiss; consultancy of, honoraria, and travel cost support by Novartis, Bayer, Heidelberg Engineering, Allergan, and Zeiss. G. Spital: financial and nonfinancial support for the conduct of clinical trials by Novartis, Ophthotech, Pfizer, Acucela, Genentech, Roche, and Bayer; consultancy of, honoraria, and travel cost support by Allergan, Bayer, Novartis, and Alimera Science. F.G. Holz: financial and nonfinancial support for the conduct of clinical trials by Acucela, Alcon, Allergan, Bayer, Genentech, Heidelberg Engineering, Novartis, Ophthotech, and Roche; consultancy of, honoraria, and travel cost support by Allergan, Avalanche, Bayer, Genentech, Heidelberg Engineering, Johnson & Johnson, Novartis, Ophthotech, and Roche. M. Fleckenstein: financial and nonfinancial support for the conduct of clinical trials by Alcon/Novartis, Allergan, Bayer, Bioeq/Formycon, Heidelberg Engineering, Novartis, Optos, Roche/Genentech, and Zeiss Meditec; consultancy of, honoraria, and travel cost support by Alcon/Novartis, Bayer, Heidelberg Engineering, and Roche/ Genentech.
