Abstract
Purpose
To evaluate the performance of various tests with automated and subjective evaluation for primary diagnostics of glaucoma in a normal clinical setting.
Methods
Subjects referred because of suspicion of glaucoma were recruited. All subjects had full ophthalmologic evaluation with stereophotography of the optic nerve head (ONH), red-free retinal nerve fiber layer (RNFL) photography, scanning laser polarimetry (SLP), optical coherence tomography (OCT), and standard automated perimetry (SAP). Analysis of all results was made jointly by 5 glaucoma specialists, with a consensus for each eye as healthy, glaucomatous, or glaucoma suspect. Results from each method were evaluated against this consensus, followed by a subjective evaluation of each method by a glaucoma specialist, comparing this with automated classification by the devices.
Results
Of the 101 subjects and 202 eyes examined, 23 eyes were by consensus glaucomatous, and 23 were glaucoma suspect. Sensitivity was best with ONH photos and worst with SAP, while SLP had a better sensitivity but poorer specificity than did RNFL photographs and OCT. Subjective evaluation of SLP, OCT, and SAP data gave better sensitivity and specificity than did classification by numeric values from the devices only, with OCT performing better in automatic classification than did SLP or SAP.
Conclusions
None of the current methods is superior to others in diagnosing glaucoma, and the accuracy of automated tests was better when subjectively evaluated by an experienced ophthalmologist. Diagnosis of glaucoma should be based on a combination of test results interpreted by a clinician.
Keywords
Introduction
Glaucoma is a progressive, degenerative disease of the optic nerve (1). Since treatment can only stop its progression or slow it, early diagnosis is important to prevent serious effects on vision and possible blindness. Glaucoma has conventionally been diagnosed by detection of typical structural changes in the optic nerve head (ONH) and corresponding visual field (VF) defects in automatic perimetry testing (2). The corresponding retinal nerve fiber layer (RNFL) defects can be visualized with red-free photographs of the RNFL (3). However, diagnosing glaucoma in the early or moderate stage can be challenging. Appearance of the healthy ONH varies, and discrimination between healthy and glaucomatous ONH may be difficult in early glaucoma (4). Visual field testing is the only way to measure the functional damages glaucoma causes, but the success of VF testing depends on the patient's subjective performance, and is vulnerable to many possible artefacts (5). Sometimes VF defects appear only after structural damage is already detectable (6). Interpretation of RNFL red-free photographs is also difficult. Slit-like defects may be easily detectable, but diffuse defects of the RNFL may go undetected. Retinal nerve fiber layer red-free photography is also dependent on the clarity of the optical media and on sufficient pigmentation of the retinal pigment epithelium (3).
In glaucoma diagnostics, new diagnostic methods offer benefits. Scanning laser polarimetry (SLP) and optical coherence tomography (OCT) allow quantitative measurements of the RNFL thickness around the ONH (7). The advantages of SLP and OCT measurements are that they are noninvasive, are quick to perform, and allow comparison of their measurements against normative databases. Thus, they have become widely popular. Because of large individual variation in the normal RNFL configuration, both methods can produce aberrant results (7). Moreover, in both methods, eyes with large refraction errors have been excluded from the normative databases, making these methods prone to diagnostic inaccuracies in these eyes.
In glaucoma diagnostics, no method seems convincingly better than others (7). Scanning laser polarimetry and OCT have been equally good at recognizing glaucoma (8-9-10-11-12-13-14-15), OCT has been more accurate than SLP (16-17-18-19), ONH photographs have performed better than SLP or OCT (20, 21), and in one study, VF examination performed better than did OCT, SLP, or RNFL (22).
The purpose of this study was to examine patients suspected of having previously undiagnosed glaucoma for comparison of the diagnostic accuracy of currently popular methods of glaucoma diagnosis. We also compared the ability of the numeric values that the devices provide to diagnose glaucoma, as compared with assessment of the data by glaucoma specialists. Additionally, we evaluated interobserver variability in diagnosing glaucoma or glaucoma suspicion based on differing tests.
Methods
Our study had the approval of the Helsinki University Hospital Ethics Committee and followed the tenets of the Declaration of Helsinki (study registration number: 378/13/03/02/2008). It took place at the Helsinki Private Eye Hospital between February 2010 and January 2012, when we prospectively collected data from consecutive subjects referred by private practitioners for examinations because of suspected glaucoma. The aim was to recruit 100 patients. The subjects were invited for a thorough ophthalmologic examination and evaluated for suitability for the study. The examination included best-corrected visual acuity, measurement of intraocular pressure (IOP), a thorough biomicroscopic examination including gonioscopy, and funduscopic examination in mydriasis. Participants signed an informed consent approved by the Ethics Committee.
Inclusion criteria were at least one of the following clinical findings:
Optic nerve head with a cup-to-disc (C/D) ratio of 0.6 or more
Difference of 0.2 or more in C/D ratios between eyes
Local notch in the ONH neuroretinal rim
Divergence from the inferior-superior-nasal-temporal (ISNT) rule (ISNT being the order of neuroretinal rim width from widest to narrowest in healthy eyes)
Mean IOP higher than 21 mm Hg on at least 3 consecutive measurements
Exclusion criteria were previously diagnosed glaucoma in either eye, other major ocular morbidity (including age-related macular degeneration, diabetic retinopathy, and active or previous uveitis), previous major ocular trauma, and any ocular surgery (including laser refractive surgery). Eyes with cataract surgery performed at least 6 months earlier were included. No exclusion criteria concerned age, refraction, or race. For all consecutive subjects meeting our study criteria, we included both eyes. All underwent stereophotography of the ONH, red-free photography of the RNFL, SLP, OCT, and standard automated perimetry (SAP).
Optic nerve head stereophotography
The pupils of all eyes were dilated with tropicamide eyedrops (Oftan Tropicamid 0.5%; Santen Pharmaceuticals). With a Canon F-E film camera at a 40° field of view, 2 images of the ONH were taken at different angles. A slide including the 2 images was viewed with a stereoscopic viewer. Diffuse and local defects of the ONH rim were evaluated.
Retinal nerve fiber layer photography
Pupils were dilated with tropicamide eyedrops. A Canon EOS20D digital camera at a 60° field of view took red-free photographs of the RNFL. From several images, the technician selected the best-quality ones, adjusted the contrast and brightness of the images, and printed them on photographic paper. Diffuse and local defects underwent evaluation (23).
Scanning laser polarimetry
GDxPRO ECC (Carl Zeiss Meditec, Inc.; software version 1.0) served for taking SLP images through undilated pupils, a technique described in detail elsewhere (24-25-26). Measurement of retardation of light passing through the birefringent RNFL provides an estimate of RNFL thickness. The mean of 3 consecutive scans served for analysis. A fixed concentric band centered on the ONH with a 2.4-mm inner and 3.2-mm outer diameter allowed generation of peripapillary retardation measurements. If any peripapillary atrophy area fell within the measurement band, the band was made larger. The software provides a color-coded thickness map of the RNFL and also compares the estimated RNFL thickness to a normative database. The printout highlights deviations from the normative database. Summary parameters with the average RNFL thicknesses measured within the band for the temporal, superior, nasal, inferior, temporal (TSNIT), superior, and inferior sectors are displayed and compared with the normative database. Deviations are color-coded. The nerve fiber indicator (NFI) generated is calculated with a neural network algorithm and is based on the entire RNFL thickness map. This summary value is intended to represent the likelihood of glaucomatous RNFL loss (24).
Optical coherence tomography
With Cirrus OCT (Carl Zeiss Meditec; version 5.1.1.6), we took OCT measurements from the ONH and peripapillary RNFL through undilated pupils, a technique described elsewhere (27). Optic disc scans (Optic Disc Cube 200 200 protocol) allowed measurement of peripapillary RNFL thickness and optic disc topography. The best-quality disc scans (good signal strength, avoidance of RNFL misalignment and of blinking or saccades) were selected. The software provides a color-coded thickness map of the RNFL and compares it with a normative database. An RNFL deviation map with deviations highlighted is produced. Average thickness for quadrants (superior, inferior, temporal, nasal) and clock-hour sectors as well as summary parameters of average RNFL thickness, RNFL symmetry, ONH rim area, average C/D ratio, average vertical C/D ratio, and cup volume are given with deviations from the normative database highlighted.
Standard automated perimetry
All subjects underwent a Central 24-2 SITA Standard Threshold Test with the Humphrey Automated Perimeter (Carl Zeiss Meditec; HFA II 750-6202-14.0/14.0). The manifest refraction had an age-specific correction, and in the test pupils were undilated.
No quality requirements for the images or VFs were predefined. The examiners had the responsibility to evaluate whether the examinations could be analyzed or excluded. Evaluations were performed by 5 experienced glaucomatologists: 2 from the private sector and 3 from a university hospital. They all were experienced and familiar with all examination methods. Each category of tests (ONH photographs, RNFL photographs, SLP, OCT, and VF) underwent separate evaluation by 3 glaucomatologists. After that, all 5 glaucomatologists met and reviewed together all tests of each eye and agreed whether the eye was healthy, had glaucoma, or produced a suspicion of glaucoma. This procedure resulted in 3 classifications:
Consensus: The entire examiner group of 5 glaucoma specialists working together made a categorization of each eye as normal, glaucoma suspect, or with glaucoma. Testing of subsequent sensitivities, specificities, and diagnostic accuracies of all separate examination methods utilized this consensus as the reference classification.
Subjective classification: Three glaucoma specialists analyzed each examination method separately, each making the assessment alone, without any further patient information. They were also blinded to the results of all the other tests of that eye. First they categorized each eye as normal, glaucoma suspect, or with glaucoma and, secondly, as a forced choice between normal and glaucoma. If at least 2 specialists agreed (classification of normal, glaucoma suspect, or glaucoma), that category was the one chosen for the eye. Those tests rated as noninterpretable were classified as negative findings.
Automated classification: The interpretation of SLP, OCT, and SAP results can be done subjectively by the examiner or strictly according to software-provided parameters. In the consensus classification (1) and the subjective classification of separate methods (2), the examiners used subjective evaluation of the printouts, parameters, and their comparison to normal databases provided by the software. In the automated classification, each method used only software-based parameters. Cutoffs were as follows:
For SLP, an NFI of >35 was classified as glaucoma suspect and >55 as glaucomatous. Retinal nerve fiber layer integrity parameters of <5% classified the eye as glaucoma suspect (color-coded as any color on the printout) and of <1% as glaucomatous (color-coded as yellow or red on the printout) if a deviant value in any location (TSNIT, superior, or inferior average) gave a positive finding.
With OCT, we evaluated RNFL thickness in the quadrants and clock hours. The cutoff was set at <5% for glaucoma suspect (color-coded as yellow or red on the printout) and at <1% for glaucoma (color-coded as red on the printout), with a deviant value in any sector giving a positive finding.
For SAP, we classified the glaucoma hemifield test by labeling borderline as glaucoma suspect and outside normal limits as glaucoma, and, secondly, classifying either mean deviation or pattern standard deviation at a value of p<5% for glaucoma suspect and p<1% for glaucoma.
These 3 classifications allowed us to compare diagnostic accuracies of single examination methods with the consensus classification as the reference. Interobserver variabilities of all 5 methods were also tested, and subjective and automated classifications of SLP, OCT, and SAP compared.
Results show data first with the cutoff set between normal and suspect (with normal being the first group and suspect and glaucomas together in the second group), and, secondly, grouped as normal and glaucoma according to the forced-choice classification. Examinations classified as noninterpretable were classified as negative findings. These could include VFs with low reliability, images of poor quality, and RNFL photographs with a nonpigmented fundus where the RNFL is not visible.
The IBM SPSS Statistics v22 software package served for statistical analyses. Sensitivities, specificities, and diagnostic accuracies were calculated for each examination method against the consensus. For SLP, OCT, and SAP, we used Cohen kappa statistical method to test agreement of the classifications according to the indices of the devices and the subjective classification by glaucoma specialists against the consensus. The same method served to calculate interobserver variability of classification among 3 specialists for each examination method.
Results
Of the 109 subjects who were referred because of suspicion of glaucoma, 101 fulfilled the inclusion criteria and were included. Of these, 52 were included because of high IOP, 27 because of a high C/D ratio, and 15 met the criteria for both high IOP and high C/D ratio. Four patients were included because of divergence from the ISNT rule and 2 because of difference in C/D ratios between eyes. Referrals came from 44 ophthalmologists, each referring a mean 2.3 patients (SD 1.9, range 1-9). All subjects were white and of Finnish ethnic origin. Their mean (SD) age was 58.5 (9.6) years, with a range from 33 to 77 years. Of the 101, 83 patients (82%) were female. Of 202 eyes, 4 had undergone cataract surgery with intraocular lens implantation. All eyes had open angles on gonioscopy (Schaffer grade at least 3). Twelve eyes had exfoliation syndrome and none had findings suggesting pigment dispersion syndrome. Visual acuity was at least 1.0 (Snellen equivalent) in 183 eyes (91%) and between 0.5 and 0.8 in the remaining 19, in which the decreased visual acuity was attributable to cataract with no other ocular morbidity.
The mean (SD) spherical equivalent of the eyes was −0.67 D (1.9 D) with a range from −7.75 to +3.0 D. No eye was treated for glaucoma or ocular hypertension. The mean (SD) IOP of the eyes was 21.3 mm Hg (3.2 mm Hg) with a range from 13 to 30 mm Hg.
Consensus
After all 5 experts met to discuss and used all available data from all examination methods, of 202 eyes, 23 were classified as glaucomatous and 23 as glaucoma suspect, leaving 156 eyes classified as normal. Of the 101 patients, findings referring to glaucoma or glaucoma suspicion were detectable bilaterally in 17 (34 eyes) and unilaterally in 12 (12 eyes). Of all patients, 16 were classified as having glaucoma in either eye, 13 as having possible glaucoma in either eye, and 72 as normal in both eyes.
Subjective classification of each separate method versus consensus
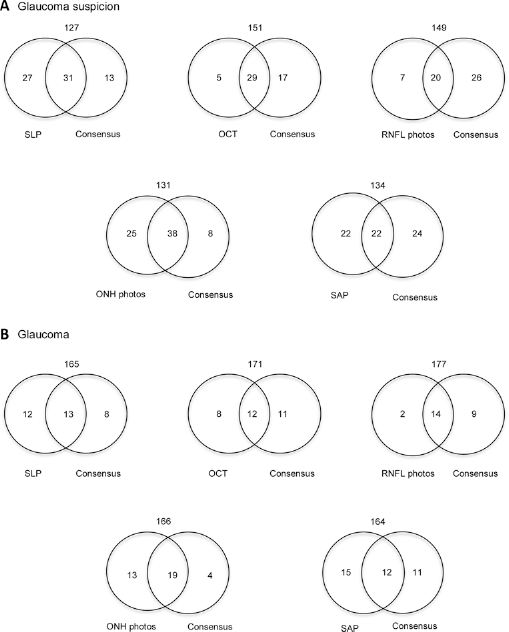
Results of the subjective classifications by glaucoma specialists of single examinations versus the consensus appear as Venn diagrams in Figure 1A for glaucoma suspicion and Figure 1B for glaucoma. Sensitivities, specificities, and diagnostic accuracies appear in Table I. Numbers of examinations classified as not interpretable (examiner 1/examiner 2/examiner 3) were SLP 5/1/0, RNFL photographs 21/19/18, and SAP 9/4/2. For OCT and ONH photographs, all examinations were classified as interpretable by all.
Diagnostic performance in detecting glaucoma or glaucoma suspect eyes
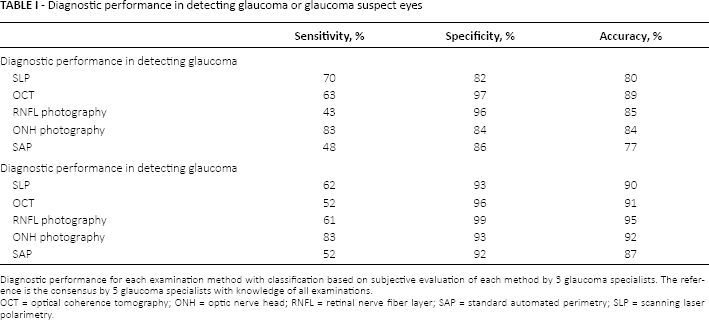
Diagnostic performance for each examination method with classification based on subjective evaluation of each method by 3 glaucoma specialists. The reference is the consensus by 5 glaucoma specialists with knowledge of all examinations.
OCT = optical coherence tomography; ONH = optic nerve head; RNFL = retinal nerve fiber layer; SAP = standard automated perimetry; SLP = scanning laser polarimetry.
Venn diagram representing numbers of eyes classified (
According to the analyses, SLP had better sensitivity but lower specificity (higher false-positive rate) than did OCT or RNFL photographs. The best sensitivity was achieved with ONH photographs, which, however, had a somewhat lower specificity than did OCT or RNFL photographs. Standard automated perimetry had the poorest sensitivity, with moderate specificity, resulting in the poorest accuracy. All methods performed better in classifying normal and definite glaucoma (Tab. I) than between normal and glaucoma/glaucoma suspect (Tab. I).
Subjective versus automated classification
Performance of the software-based classification of SLP, OCT, and SAP was analyzed against the consensus (Tab. II). In comparison of software results with those of the glaucoma specialists (Tab. II), specificities and accuracies in the former were poorer. Software specificity was especially weak in differentiating normal from glaucoma/glaucoma suspect.
Diagnostic performance in detecting glaucoma or glaucoma suspect eyes
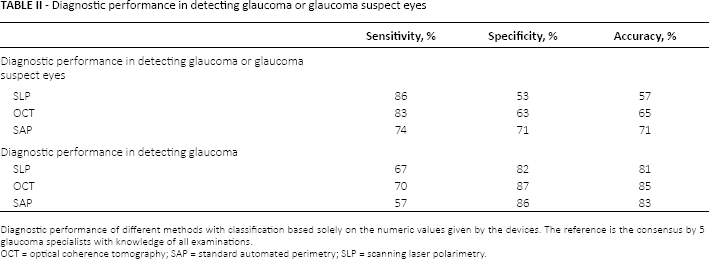
Diagnostic performance of different methods with classification based solely on the numeric values given by the devices. The reference is the consensus by 5 glaucoma specialists with knowledge of all examinations.
OCT = optical coherence tomography; SAP = standard automated perimetry; SLP = scanning laser polarimetry.
Table III shows agreement with the consensus of classifications based on subjective assessment and those based on software-based indices. In recognizing normal versus glaucoma/glaucoma suspects, subjective evaluation achieved better agreement than did software parameters. The best overall agreement was for the subjective evaluation of OCT (Cohen kappa 0.659). Of the automated classifications, the best agreement was achieved by OCT quadrants (0.532). In SLP, NFI performed better than did RNFL integrity. Standard automated perimetry was poorest of the 3 devices when compared with subjective assessment and software-based parameters.
Agreement of the consensus with subjective evaluation and with automated classifications with software-provided parameters, Cohen kappa coefficients
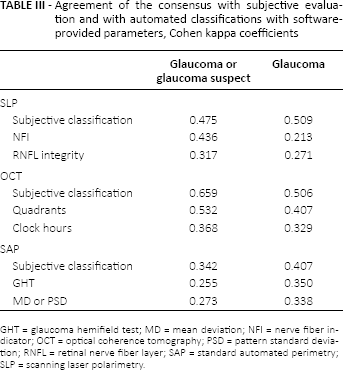
GHT = glaucoma hemifield test; MD = mean deviation; NFI = nerve fiber indicator; OCT = optical coherence tomography; PSD = pattern standard deviation; RNFL = retinal nerve fiber layer; SAP = standard automated perimetry; SLP = scanning laser polarimetry.
As to agreement in recognizing normal versus glaucoma eyes, subjective evaluation achieved better agreement than did the automated classification for all 3 devices. The best overall agreements were in subjective evaluation of SLP (0.509) and OCT (0.506). Of the automated classifications, the best agreement with the consensus was again for OCT quadrants (0.407).
Figure 2, A-C shows Venn diagrams for classification by numeric indices given by the device, subjective evaluation, and consensus. For SLP and SAP, the number of false-positives was markedly higher for classification by numeric indices than by subjective evaluation. For OCT, the numeric classification and subjective evaluation were in good agreement, but the number of false-positives fell in the same range as with SLP and SAP.
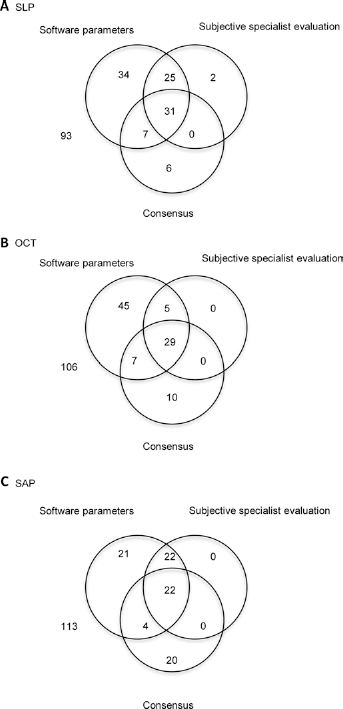
Venn diagrams of (
Interobserver variability
Table IV presents Cohen kappa values for interobserver variability. In analysis of classification of normal versus glaucoma/glaucoma suspect, OCT (0.688) and SAP (0.665) had the smallest interobserver variabilities. With classification between normal versus glaucoma analysis, SAP (0.648) and RNFL photography (0.614) showed the smallest interobserver variabilities. In both analyses, SLP showed the greatest variability.
Cohen kappa values for interobserver variability among 3 glaucoma specialists for all 5 examination methods
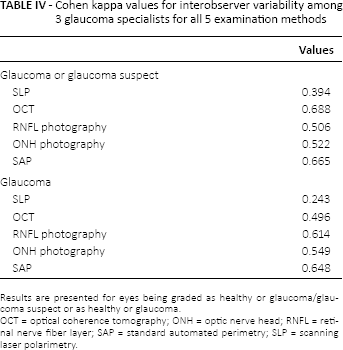
Results are presented for eyes being graded as healthy or glaucoma/glaucoma suspect or as healthy or glaucoma.
OCT = optical coherence tomography; ONH = optic nerve head; RNFL = retinal nerve fiber layer; SAP = standard automated perimetry; SLP = scanning laser polarimetry.
Discussion
Of 202 tested eyes, 23 were classified as glaucomatous, 23 as glaucoma suspects, and 156 as healthy. Optic nerve head photographs had the best sensitivity and SAP the worst, while SLP had a better sensitivity but poorer specificity than RNFL photographs and OCT. Subjective evaluation of SLP, OCT, and SAP data resulted in better accuracy than did classification by numeric values from the devices only.
In all studies testing diagnostic accuracy, results depend on the reference. In many of those studies, nearly half the patients had glaucoma, and half have been healthy (14, 16, 22, 28-29-30). This is uncommon in real life. Any diagnostic test will perform better in a setting in which half the subjects have the disease. Our study design is a real-life situation, in which private ophthalmologists referred patients for tests because of clinical suspicion of glaucoma. We thus had no predefined number of healthy and glaucomatous eyes. Moreover, we used no gold standard examination as the reference. We used inclusion criteria for the study because the reasons for referral for glaucoma tests can be variable and we wanted to have at least some criterion for glaucoma suspicion.
We did not exclude narrow angles, but none were found; therefore, our results are valid only for open-angle glaucoma. A total of 12 eyes (6%) had pseudoexfoliation, but otherwise no condition that could cause secondary glaucoma was present in the eyes included. Since optic nerve damage is similar in primary open-angle glaucoma and pseudoexfoliation syndrome (31), we did not exclude eyes with this condition. Our study examined the results of only certain devices and their performance in glaucoma diagnosis, but in real life it is important to consider the whole picture, including a thorough examination of the eye, IOP, corneal thickness, the age of the patient, and family history.
For VF examination, we chose to use 24-2 SITA Standard VF test, which is not the gold standard, but is widely used in glaucoma diagnostics. Optical coherence tomography devices are available from many manufacturers, and they are evolving. Ganglion cell analysis was not available in our OCT at the time of the examinations, but it can give more information especially in early stages of glaucoma. Heidelberg retinal tomography is another useful device in glaucoma diagnostics, which we did not have access to in this study.
Because of our small number of glaucomatous eyes, diagnostic accuracies of the examinations were lower than in many studies where the proportion of glaucomatous eyes has been higher and classification has been predefined by VF and ONH findings (14, 16, 22, 28-29-30). In our study, as in others (32), the tests disagreed in classifying eyes as glaucomatous and healthy. Adding more tests may result in a greater number of eyes with glaucoma suspicion, or may result in better certainty as to whether an eye has glaucoma.
Differences were detectable among our methods in sensitivity, specificity, and accuracy. The best sensitivity came in evaluation of the ONH stereophotographs. Because it had rather low specificity, its diagnostic accuracy was in the same range as for the other tests. The high sensitivity may in part result from the appearance of the ONH as being one of the reasons for referrals. Comparable diagnostic accuracies, sensitivities, and specificities of ONH photography have emerged (28). The best specificity was in the evaluation of RNFL photographs; this method seldom gives false-positive results. This sensitivity is, however, compromised, because a significant number (10%) of photographs received the rating of noninterpretable. In addition, among those whose RNFL photographs were classified as noninterpretable, the proportion of glaucomatous eyes according to the consensus was greater than among the rest of the eyes. The poorest diagnostic accuracy was for SAP; its low sensitivity may be at least in part due to inclusion of early cases of preperimetric glaucoma. When comparing SLP and OCT, we found better sensitivity for SLP, and better specificity for OCT, in contrast to reports of the opposite (18, 33, 34).
Printouts of SLP and OCT include comparisons of their results to the normative database. The Humphrey SAP also uses a normative database and provides the Glaucoma Hemifield Test. Comparison of the diagnostic accuracy of these automated test results with the accuracy of the same tests reviewed by experienced glaucomatologists allowed the examiner to take into account the quality of the test, the magnitude of possible deviation from the normative database, and anything else that possibly was only an artefact and not diagnostic for glaucoma. The main reason for poor diagnostic accuracies of the automated classification for glaucomatous/glaucoma suspect/healthy was low specificity, but for glaucomatous/healthy, it was low sensitivity. A recent study (35) using only automated classification for OCT reported excellent specificity but poor sensitivity for subjects who developed VF damage. A large study by Banister et al (34) including subjects with no previous glaucoma diagnosis evaluated classification based only on automated test results and performed no subjective evaluation of these. They reported performance for OCT parameters similar to ours, but for SLP, their sensitivity was much lower and specificity higher than in our automated results section. For all our 3 examinations, agreement with the consensus improved when an experienced examiner interpreted the results.
What would be ideal is to have only one test to detect glaucoma with reliable automated results. In our study, agreement with a consensus of all automated tests was fair or moderate. Cohen kappa values ranged between 0.27 and 0.53, which means that the automated classification methods misclassified half to two-thirds of patients. Even after subjective interpretation of the results, agreement with the consensus was still fair to moderate; if the options were healthy or glaucoma, 50% to 60% of eyes were misclassified.
If a test is very difficult to interpret, its interobserver variability may increase. Evaluation of the ONH is known to be difficult (36) and that was the case also in our study, because of large variation in normal ONH appearance. In our analysis of the RNFL photographs, a diffuse defect was difficult to detect. As for SAP, the result is dependent on patient performance. In analysis of OCT and SLP, comparison of the measurements to the normative database may make interpretation easier. Our results with every method were separately evaluated by 3 glaucoma specialists, and we subsequently evaluated interobserver variability between them. When the choices for evaluation were only healthy and glaucoma, the best agreement between examiners was for SAP and RNFL photographs, with, surprisingly, the worst agreement for SLP and OCT. The reason may be the large proportion of false-positive findings with the numeric values interpreted by the 3 specialists differently. However, when the choices were healthy/glaucomatous/glaucoma suspect, agreement between examiners was better. A recent study compared healthy, preperimetric glaucomatous, and perimetric glaucomatous eyes and reported good interobserver agreement in detecting glaucoma; the more examination methods were combined in the assessment, the better (37). Another study used ONH stereophotographs and SLP (28). Both methods achieved better agreement than we did. This may be due to the higher proportion of glaucomatous eyes in their setting (55%), and because all their glaucomatous eyes had VF defects.
Of 101 subjects examined, glaucoma diagnosis was made in 16. If 16% of patients tested for glaucoma have a positive result, it can be discussed whether the threshold for referring patients for glaucoma tests is appropriate. A low percentage indicates that the threshold may be too low. A too high percentage and threshold for referral would indicate that many glaucoma patients would go undetected.
Our results suggest that none of the diagnostic methods is superior to the others in regard to diagnostic accuracy. Even though some methods provide parameters comparable to those of normative databases, a glaucoma diagnosis cannot be based on these numbers without any assessment by an ophthalmologist. Test accuracy improves if printouts are subjectively evaluated by an experienced ophthalmologist. Furthermore, for making diagnoses one should not rely on only one single test result. Different tests will find different patients to have glaucoma. Thus, combining results of several differing methods with clinical status, IOP, age, and family history allows the clinician to make the best guess. One must, however, keep in mind that an increased number of examinations raises costs, and also the possibility of a false-positive result in any one test.
Footnotes
Financial support: Supported by Glaukooma Tukisäätiö Lux and Helsinki Private Eye Hospital.
Conflict of interest: None of the authors has conflict of interest with this submission.
