Abstract
A case of symptomatic epiretinal membrane (ERM) is presented. The discussion revolves around key questions:
1. What do we know about this disease entity?
2. What is the traditional approach to symptomatic ERM?
3. What are the major complications of pars plana vitrectomy (PPV)?
4. Is there any technical solution available for increased safety during vitrectomy?
5. What are the advantages of wide-angle light probes?
6. How can dye-related chemotoxicity be reduced?
7. How non-contact wide-angle viewing systems could help during vitrectomy?
8. What were the results of a safe standardized approach to symptomatic ERM?
Keywords
Case presentation
A 67-year old male patient presented to the Ophthalmology Unit, Public Hospital of Desio, complaining of decreased vision and metamorphopsia. The patient underwent best-corrected visual acuity (BCVA), biomicroscopy, applanation tonometry, infrared (IR) reflectance, and spectral-domain optical coherence tomography (SD-OCT) (Spectralis, HRA, Heidelberg, Heidelberg, Germany).
The clinical examination disclosed a BCVA of 20/63, with a positive Amsler test. Indirect ophthalmoscopy showed macular wrinkling with increased vascular tortuosity. The SD-OCT confirmed the presence of epiretinal membrane (ERM) causing an increase in central macular thickness (CMT) up to 470 μmand distortion of normal macular anatomy (Fig. 1).
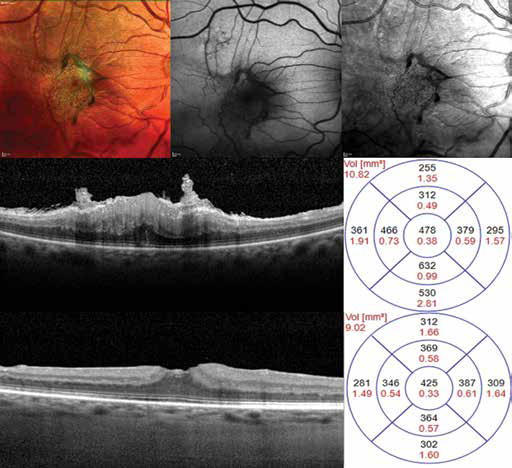
Multimodal imaging of a 67-year-old male patient presenting with epiretinal membrane (ERM) in his right eye. Upper row: multicolor, fundus auto-fluorescence, infrared imaging of the patient. Central row: spectral domain optical coherence tomography (OCT) showing increased central macular thickness (CMT) due to the ERM. Lower row: OCT of the same patient 3 months after undergoing pars plana vitrectomy and ERM peeling. CMT significantly lower with respect to baseline.
What do we know about this disease entity?
ERM, also known as macular pucker or cellophane maculopathy, is a common disorder affecting the vitreoretinal interface, resulting from the proliferation of cells along the internal limiting membrane (ILM) of the macula (1, 2). The main risk factor for this condition is age. Other risk factors include trauma, ocular inflammation, and previous ocular surgery. The overall incidence in a single eye is ∼1.1% and increases to 6% in individuals aged ≥65 years (3-4-5-6). The most common symptoms associated with ERM formation are distorted vision (metamorphopsia), blurred vision, macropsia, and micropsia (7, 8).
What is the traditional approach to symptomatic ERM?
Traditional 20- and 23-gauge pars plana vitrectomy (PPV) with membrane peeling has been used successfully for many years to treat ERM with a postoperative morphologic and functional improvement (9-10-11). Nowadays, the introduction of 25- (25G) and 27-gauge (27G) PPV systems have shown superiority over traditional greater-gauge techniques, in terms of shorter surgical times, faster wound healing with less conjunctival scarring, decreased postoperative inflammation, and reduced postoperative astigmatism (12-13-14-15-16).
Cataract extraction through phacoemulsification and subsequent intraocular lens (IOL) implantation can be performed simultaneously to PPV in presence of visually significant cataract.
What are the major complications of PPV?
The complication rate of PPV has been extensively reported in the current literature (17-18-19). Although surgical peeling represents a mainstay of ERM treatment, the frequency of peri- and postoperative complications has been globally estimated at 0.4%-0.8%. The major events are bleeding (1.5%-2%), macular edema (5.9%-9.4%), iatrogenic retinal breaks and retinal detachment (5.5%-10%) (20-21-22-23). Endophthalmitis represents a less frequent event in vitrectomized eyes, because of the absence of the vitreous (24).
Is there any technical solution available for increase safety during vitrectomy?
First, 25G PPV, coupling a high cutter rate with low infusion pressures and adjustable raising aspiration speed, is preferable to traditional greater-gauge techniques: the small size, the stiffness and the positioning of the probe mouth close to the probe's tip allows precise surgical maneuvers and may help operators’ confidence, even for less experienced surgeons.
Moreover, wide-angle light probes, less toxic intraocular dyes, and non-contact wide-angle viewing systems could provide an even safer and repeatable approach to surgical ERM removal for many reasons: encouraging a micro-incisional approach, reducing illumination-related chemotoxicity, and increasing fundus visualization.
Nevertheless, these new devices are often proposed on the market without solid scientific literature to support their use, and surgeons tend to build their own personal procedures based on experience, habit, financial resources of medical structure or even personal interests. Lack of procedural standardization could lead to a dramatic increase in the rate of surgery-related complications.
What are the advantages of wide-angle light probes?
The use of wide-angle light probes enables the visualization of different retinal areas at the same time, while focusing on the operating field. In delicate steps, such as induction of posterior vitreous detachment, it can dramatically reduce the risk of retinal damage (25, 26). Moreover, the possibility to keep the probe at a greater distance from the retina reduces the endoillumination-related toxicity. As there are some concerns regarding retinal phototoxicity, even with the standard xenon lower-wavelength filter (420 nm), we encourage the use of stronger filters, such as amber filter (515 nm), that reduces the overall risk up to 30 times maintaining a good visibility for the surgeon (27-28-29). In addition, it is advisable to set the light source at 30% of the power output and to dim the room lights.
How can dye-related chemotoxicity be reduced?
Many dyes are available for selective intraoperative staining of the intraocular structures (chromo-vitrectomy), including trypan blue, brilliant blue (BB), indocyanine green (ICG), and infracyanine green (30, 31). Trypan blue is thought to be the ideal dye for ERM identification, while ICG and BB are ideal for ILM peeling, although there are concerns about their potential retinal toxicity (32). Lutein is a lipophilic carotenoid traditionally found in fruits and vegetables that is physiologically present in the macula lutea (33, 34). Recently, lutein has been proposed as a less toxic non-vital dye for retinal tissues compared to other pigments (35, 36).
Moreover, steady and controlled dye injection, achieved by means of instruments such as the Drip Dropper (Katalyst Surgical LLC, Chesterfield, USA), a rubber bulb reservoir with a stainless-steel needle ending into a silicon-coated diamond tip, help in directing the dye jet straight into the macula (37).
How non-contact wide-angle viewing systems could help during vitrectomy?
Non-contact wide-angle vision systems provide a high-quality fundus image, that is brighter, more stable and with a wider angle if compared with traditional contact systems (38, 39).
What were the results of a safe standardized approach to symptomatic ERM?
The results of such a standardized intraoperative schedule for the treatment of idiopathic ERMs were encouraging.
We enrolled a total of 87 eyes of 87 patients (50 males, 57.5%), with mean age 75.9 ± 6.7 years (55-90 years) and mean refractive error 0 ± 2.0 D spherical equivalent. Seventy-one eyes were phakic (81.6%); significant difference was found in age between phakic (group 1) and pseudophakic eyes (groups 2 and 3), the former being younger, while baseline visual acuity did not meaningfully differ among the groups (Tab. I). All the eyes were scheduled for surgical removal of ERM. Both the functional and the anatomical results at the end of the follow-up period were favorable, showing a positive trend towards the post-operative recovery.
Demographics and main clinical characteristics of study population
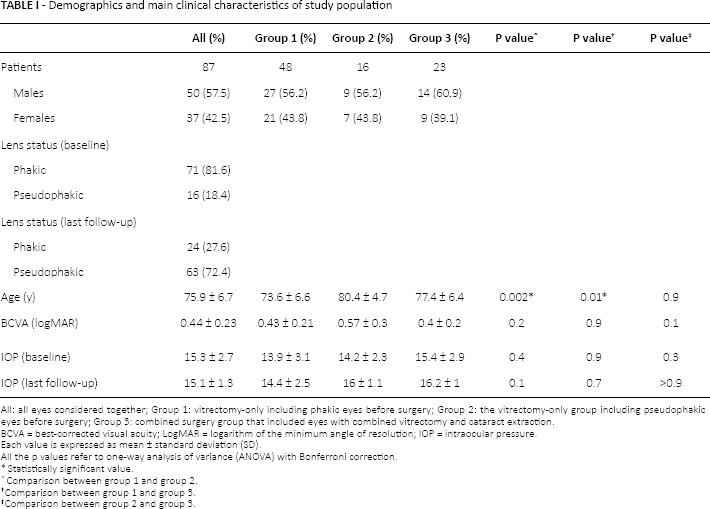
All: all eyes considered together; Group 1: vitrectomy-only including phakic eyes before surgery; Group 2: the vitrectomy-only group including pseudophakic eyes before surgery; Group 3: combined surgery group that included eyes with combined vitrectomy and cataract extraction.
BCVA = best-corrected visual acuity; LogMAR = logarithm of the minimum angle of resolution; IOP = intraocular pressure.
Each value is expressed as mean ± standard deviation (SD).
All the p values refer to one-way analysis of variance (ANOVA) with Bonferroni correction.
Statistically significant value.
Comparison between group 1 and group 2.
Comparison between group 1 and group 3.
Comparison between group 2 and group 3.
In fact, all the eyes showed successful complete membrane removal and visual and anatomical improvement: macular thickness decreased by 73.7 μm (−16.4%) and visual acuity increased by 0.14 logMAR (+32%) from baseline (p<0.001). Considering the groups separately, the greater and earlier relative increase in visual acuity was recorded in the combined-surgery group, with an evident difference from baseline as early as month 4 instead of month 6 (p = 0.01).
In conclusion, a checked instrumental approach is strongly encouraged to achieve safe removal of idiopathic ERMs, a feasible surgery, and predictable outcome. This approach can be applied to diverse posterior-segment vitreoretinal surgeries, including retinal detachment and macular hole closure.
Footnotes
Financial support: Giuseppe Querques consultant for: Alimera Sciences (Alpharetta, Georgia, USA), Allergan Inc (Irvine, California, USA), Heidelberg (Germany), Novartis (Basel, Switzerland), Bayer Shering-Pharma (Berlin, Germany), Zeiss (Dublin, USA). Francesco Bandello consultant for: Alcon (Fort Worth, Texas, USA), Alimera Sciences (Alpharetta, Georgia, USA), Allergan Inc (Irvine, California, USA), Farmila-Thea (Clermont-Ferrand, France), Bausch And Lomb (Rochester, New York, USA), Genentech (San Francisco, California, USA), Hoffmann-La-Roche (Basel, Switzerland), Novagali Pharma (Évry, France), Novartis (Basel, Switzerland), Bayer Shering-Pharma (Berlin, Germany), Sanofi-Aventis (Paris, France), Thrombogenics (Heverlee, Belgium), Zeiss (Dublin, USA), Pfizer (New York, USA), Santen (Osaka, Japan), Sifi (Aci Sant'Antonio, Italy).
Conflict of interest: None of the authors has financial interest related to this study to disclose.
