Abstract
Purpose
Effective postinsertion management of central venous access devices (CVADs) is important to prevent CVAD-associated complications, including catheter-associated bloodstream infections. Although there is a wealth of evidence-based guidelines available to guide the care of CVADs, applying their recommendations to the clinical setting across variable patient groups, CVAD types and international healthcare settings is challenging. This may result in patients receiving suboptimal care.
Methods
A cross-sectional descriptive study using an online survey was performed with an aim to determine current CVAD site care practices internationally. The CVAD site care domains included skin antisepsis, dressing selection, frequency of dressing change and device securement practices across impaired and unimpaired CVAD sites.
Results
Clinicians (n = 1044) residing in 34 countries reported diversity in their practice, with the majority of respondents practicing as nurses (89%) from North America (81%) as vascular access specialists (52%). The respondents’ reported practice was variant, with differing inconsistency to guidelines throughout each of the domains. There was wide variance in the management of CVAD sites with impaired skin integrity, such as rash, skin stripping/adhesive-related injuries and drainage. Vascular access clinicians reported high levels of confidence in managing CVAD sites, including those with impaired skin.
Conclusions
These inconsistencies are reflective of the complex and heterogeneous populations requiring CVADs, the evidence available to support practice in this area, the skills and knowledge of the clinicians caring for them and the resources of the healthcare setting. Further research and education is necessary to ensure that CVAD site care is undertaken effectively to minimise preventable complications.
Introduction
Central venous access devices (CVADs) provide necessary access for the administration of infusion therapy in acute care and home care settings. Although great strides have been made to reduce bloodstream infections during placement of these devices with the introduction of the central line insertion bundles (1), the risk of complications associated with care and use of these devices has remained apparent. Infections related to vascular access devices may be caused by ineffective or improper care and maintenance of these devices. Microbial contamination and colonisation of the CVAD entrance site is considered to be a major source of CVAD-associated bloodstream infections in short and long-term devices (2). CVAD skin antisepsis and dressings can reduce microbial colonisation of the CVAD insertion site, thereby reducing CVAD-associated bloodstream infections (3, 4). Adherence to best practice recommendations is crucial to minimise CVAD maintenance-related complications such as catheter-related infections, catheter dislodgment and thrombosis (5).
A wealth of international evidence-based guidelines (68–11) is available to direct the care of CVADs in an effort to prevent complications. The majority of guidelines recommend the use of clear transparent CVAD dressings for wound coverage, skin antisepsis with more than 0.5% chlorhexidine gluconate in alcohol and CVAD securement devices (68, 11). However, research indicates inconsistencies in the use of these best practice recommendations within the provision of patient care (5, 121416). Specific deficits previously highlighted within cross-sectional surveys involved the variable use of proper hand hygiene (15), antimicrobial solutions (5, 16) and dressing and securement products (5, 16). Clinicians frequently have a poor knowledge of the practices required to prevent CVAD-associated infections and complications (17), and an underlying lack of policy for CVAD management (12) was evident. Although these studies have reported fundamental irregularity within the CVAD management of specific populations and countries, the international variation in CVAD management across patient groups has not been previously described.
Reducing non-evidence based variability in CVAD site management practices plays an important role in reducing CVAD complications, as illustrated in the success of the Central Line Insertion Bundle project (1). However, variability in CVAD site care may be to some patients’ benefit. Impaired skin integrity is prevalent in the patient populations requiring CVADs due to conditions such as cellulitis, dermatitis, chemotherapy-induced injuries, medical adhesive related skin injury (MARSI), moisture-associated skin damage and exudate at the site (4, 18). International evidence-based guidelines provide little strategy in the provision of CVAD site management for patients who have impaired skin integrity. Clinicians are left little support and evidence when providing CVAD site care to this complicated, yet widespread, population. This lack of evidence to support clinician decision-making and practice may impact on the rates of failure and complication evident in patients with CVAD.
The goals of this international study were to:
identify current practices for the site management of CVADs;
recognise current practices for the site management of CVADs for patients with impaired skin integrity;
rate current clinician confidence and decision-making regarding the site management of CVAD; and
describe current training and institutional policies to support CVAD site management practice.
Methods
Design
A cross-sectional, descriptive, international study using a questionnaire was undertaken between April and August 2014.
Participants
Interdisciplinary clinicians currently caring for CVADs and able to read and write English were invited to participate in the online survey. In order to maximise response rates, clinicians were invited to participate via vascular access collaborations, including Canadian Vascular Access Association (CVAA; 585 members); Infusion Nurses Society (INS; 7000 members); World Congress of Vascular Access (WoCoVA; approximately 1500 delegates); Association of Vascular Access (AVA; 2757 members) and the Alliance for Vascular Access Teaching and Research Group (120 members). Attempts were made to distribute the questionnaire to the European Society for Parental Nutrition (ESPEN); however, they were unable to participate during the study period. Participation of clinicians was voluntary and responses were collected anonymously. These organisations emailed all members, providing information regarding the questionnaire and a link to the online questionnaire server (Survey Monkey®). For purposes of this survey, the term CVAD refers to peripherally inserted central catheters (PICCs), non-tunnelled catheters, tunnelled catheters and implanted ports only. Haemodialysis and umbilical catheters were not included in the survey. Because the survey was only available in English, exclusion criteria included clinicians unable to understand written English and those who provide care only for haemodialysis catheters.
Development of the survey
The available literature surrounding CVAD site management suggested the survey of six key domains: site antisepsis, skin preparation, dressings, securement, clinician training and institutional policies (68–11). From these domains, 34 closed questions were generated. Each item was formatted as multiple-choice questions with multiple response alternatives or options; ranking of response alternatives (first, second or third preference); or five-scale Likert-type response alternatives to measure the level of confidence or frequency (17). Open text responses were provided within each of the key domains, to facilitate the respondents providing additional details regarding their practice, outside of the survey response alternatives. Demographic questions were also included to describe the characteristics of the survey respondents (years of experience, age, gender, profession).
Content validity and feasibility
Content validity and feasibility was established prior to the survey distribution to ascertain whether the items within the questionnaire were adequate to describe the research questions and to ascertain the practical utility of the questionnaire (17). Content validity was achieved by a review of the questionnaires’ items by seven experts in the topic area. The experts were identified as leading interdisciplinary researchers and current clinicians in the field of vascular access with representatives from Canada, the United States and Australia.
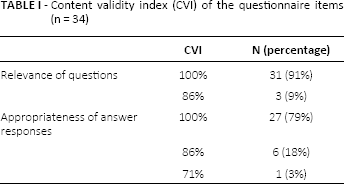
Using a four-point level of agreement (1, not; 2, somewhat; 3, quite; 4, highly), the experts were requested to provide feedback on the individual questions regarding the relevance of the question and appropriateness of the answer responses. The Content Validity Index (CVI) (19) was then calculated to describe the proportion of experts who gave the item a rating of three or four. The experts were then required to recommend whether to 1, delete the item; 2, major revision; 3, minor revision or 4, keep the item as it is.
Table I describes the CVI of the individual items. Overall, the experts recommended minor improvements to two items (6%) and no improvements to 32 items (94%). No items were recommended for major improvements or deletion.
Content validity index (cvi) of the questionnaire items (n = 34)
Feasibility testing of the questionnaire was performed by distributing the survey to eight current clinicians from Australia, the United States and Canada. The survey ascertained the length of time to complete the questionnaire, how easy it was to complete and whether the language and phrases used in the scale were clear and understandable (20). Overall, feasibility respondents reported that it took between 6 and 15 min to complete the questionnaire, the questions were easy to understand and the questions followed a logical sequence. The respondents reported small technical difficulties in one of the survey questions. Following the feedback from the content validity analysis and feasibility respondents, the questionnaire was revised.
Ethical considerations
Ethical approval was gained through the Veritas Institutional Review Board (Montreal, Canada) and Griffith University's Human Research Ethics Committee (Brisbane, Australia) prior to study commencement. Participants were given a detailed information statement once they accessed the survey link; completion of the questionnaire implied consent to participate. No recruitment incentives were provided. Anonymity and confidentiality of the survey respondents was assured.
Data analysis
Data were entered and analysed using PASW Statistics Version 22.0 (SPSS Inc., Chicago, Illinois, USA). Basic frequencies were calculated for all variables and any extreme or obviously incorrect data were re-checked for accuracy. Descriptive statistics were used to describe the survey results including frequencies and percentages to describe categorical data. Relationships between respondent characteristics (e.g. certification in vascular access, population) and clinician confidence were assessed using Chi-square (21). Variables with p value less than 0.05 were considered statistically significant. All missing data are explained in the tables and the Results section.
Results
Sample characteristics
Overall, there were 1113 respondents, of whom 69 were removed during data cleaning, as the respondent reported that they did not provide care for CVADs and/or did not read English, thereby not meeting the study inclusion criteria. The final number of respondents was 1044, with 34 participating countries represented (Fig. 1). Table II provides the demographic characteristics of the participants. The majority of the respondents were nurses from North America with greater than 10 years of CVAD experience. A large percentage of the respondents self-identified as vascular access specialists.
Demographic characteristics of respondents to cvad management survey (n = 1044)
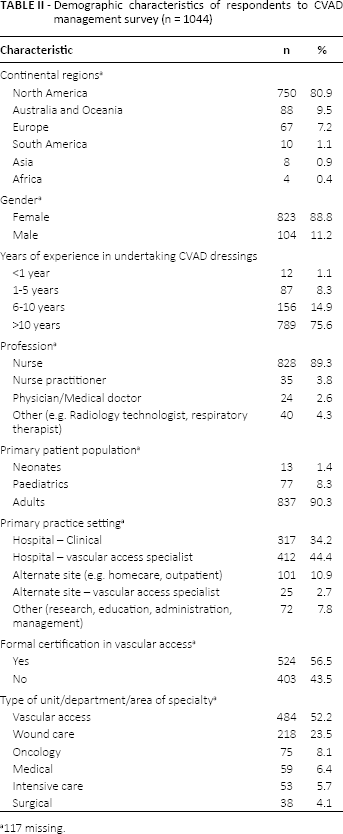
117 missing.
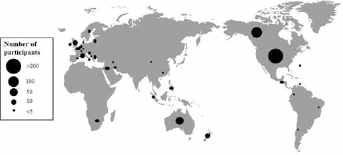
National distribution of survey respondents (n = 1044).
Survey results
CVAD site management: All CVAD
Table III describes the reported practice by vascular access clinicians throughout the domains of CVAD site management. This includes the use of varied strategies for skin antisepsis, securement, dressing, anti-microbial dressings and techniques to maintain sterility during dressing changes. Of those who reported 80% stated that they performed dressing changes every 7 days, 94% were using application of transparent dressings to the CVAD site and 83% used stabilisation devices.
CVAD dressing and securement practices (n = 1044)
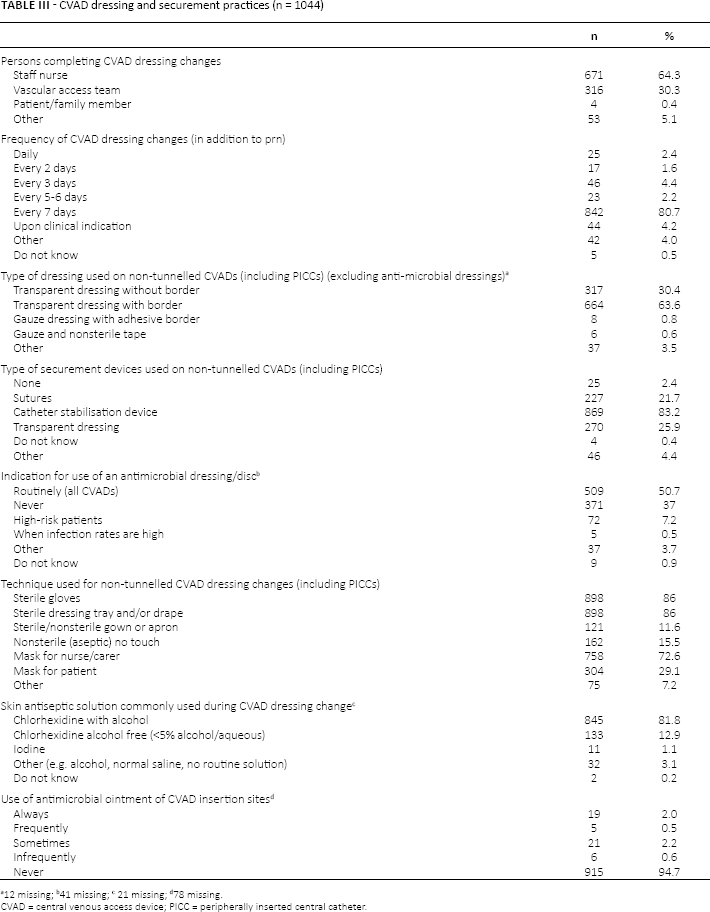
12 missing
41 missing
21 missing
78 missing.
CVAD = central venous access device; PICC = peripherally inserted central catheter.
CVAD site management: patients with impaired skin integrity
Table IV describes the vascular access clinicians’ reported practice surrounding CVAD site management for patients with impaired skin integrity across a variety of clinical conditions. Only 15% of respondents (n = 151) reported using topical agents (such as ointments) on CVAD sites for patients with impaired skin integrity. When questioned regarding skin sensitivity to antiseptic solutions, the majority of survey respondents reported that, in that clinical situation, they would change from chlorhexidine with alcohol-based solution to an iodine-based solution (n = 356; 34.1%) or an alcohol-free/aqueous-based chlorhexidine solution (n = 201; 19.3%). However, respondents also varied between using chlorhexidine at a lower percentage than previously used, pure alcohol, normal saline and sterile water.
Clinicians preference of cvad dressing product across impaired site conditions a
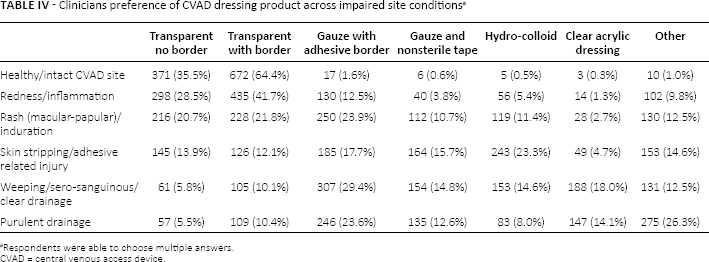
Respondents were able to choose multiple answers.
CVAD = central venous access device.
Clinician confidence and decision-making
Although the majority of vascular access clinicians reported feeling confident at managing CVAD sites (Tab. V) with each of the impaired skin integrity conditions surveyed, those clinicians with formal vascular access training were statistically significantly more confident at caring for patients with healthy/intact skin (p = 0.003); inflamed/reddened skin (p = 0.029); rash/induration (p<0.0001); adhesive-related skin stripping (p<0.0001); weeping (p = 0.001) and purulent drainage (p<0.0001). Patient population was not significantly associated with the level of clinician confidence.
Clinician confidence in providing cvad site care across impaired site conditions (n = 1044)
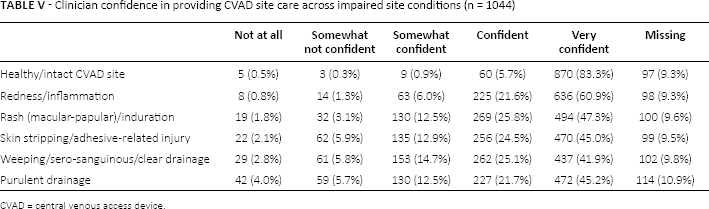
CVAD = central venous access device.
Institutional policies
There was large variance in the reported training and institutional policies surrounding the securement and dressing of CVAD. This included the frequency of training, which varied between never (n = 77; 7.4%) to more than once per year (n = 115; 11.0%), and the format in which this training and assessment of dressing change skills was provided. A large number of the respondents receive supervised demonstrations of CVAD dressing changes (n = 504; 48.3%) with validation of skills once per year (n = 348; 33.3%). The majority of organisations had a written dressing change policy or clinical guideline (n = 895; 85.7%), but only a minority of the organisations represented had a procedure or algorithm for the management of CVAD sites with impaired skin integrity (n = 119; 11.4%).
Discussion
This online survey is the first to identify current CVAD site care practices internationally in various healthcare settings (hospital, homecare, clinics, skilled nursing facilities). As seen in previous surveys, there were many reported variations and inconsistencies between healthcare settings, countries and patient populations. These practices were also occasionally inconsistent with international best practice guidelines. This is reflective of the complex and heterogeneous populations requiring CVADs, and the resources, skills and knowledge of the clinicians caring for them.
Skin preparation
Chlorhexidine is the antiseptic solution consistently recommended for site care by international clinical practice guidelines (68–11), which is reflected in respondents’ practice (n = 978; 94.7%). The use of normal saline as a skin cleansing agent was reported by a very small number of respondents; this is of concern, as saline is not an antiseptic solution with antimicrobial properties and is not one of the cleansing agents recommended by any of the aforementioned authoritative guidelines.
Although antiseptic solutions have traditionally been applied in a circular fashion, manufacturers of antiseptic swabsticks [i.e. Soluprep™ (3M St Pauls); Chloraprep™ (Carefusion San Diego)] now recommend application in a back-and-forth grid pattern with friction to cleanse the surface layers of skin, which is the most common pattern of application reported in this study. Clinicians are encouraged to confirm the appropriate application method with the best available evidence, including the product manufacturer. It is imperative that the antisepsis be allowed to dry prior to application of the dressing. Inadequate drying time may potentiate contact dermatitis and inactivate the adhesive contained in the CVAD dressing or securement product.
Dressing change practice
Most respondents reported use of transparent dressings (n = 981; 94%) (primarily with borders; n = 664; 63.6%), with the majority reporting dressing change frequency of every 7 days (n = 842; 81%), which is consistent with international guideline recommendations (68–11). The reporting of high routine usage of antimicrobial dressings differs from previous research (n = 509; 50.7%). The majority of respondents reporting the routine use of antimicrobial dressings were from the United States (US) (n = 411; 81%), Australia (n = 25; 5%) and the United Kingdom (UK) (n = 8; 1.6%), but respondents from Canada, Europe, South America and Asia also indicated that they were routinely used. The CDC and EPIC3 recommend to only use these dressings under specific conditions, that is with catheter-related bloodstream infection rates not decreasing despite infection prevention measures (6, 7). Also, the currently available evidence has only demonstrated the efficacy of at decreasing CVAD-associated bloodstream infection rates in short-term, non-tunnelled CVADs in the intensive care setting (3, 2224).
Catheter stabilisation, via sutures or sutureless securement device, is an important element of CVAD dressing and securement practice, as highlighted in international guidelines (7, 8). Respondent reports of no use of a stabilisation device (2.4%) are of concern, as poor catheter securement may result in catheter dislodgment and movement is associated with complications such as phlebitis, infection, thrombosis, infiltration/extravasation, interruption to treatment and new device insertion (2527). Traditional transparent dressings no longer meet the revised definition of catheter stabilisation (8) due to the poor tensile strength. Manufacturers are now providing transparent dressings with integrated securement properties (e.g. SorbaView SHIELD®; Centurion Medical Products, Michigan, USA), with clinical evidence (2729).
Traditionally, CVAD dressing changes have been performed using sterile technique in North America following the US Infusion Nurses Society (INS) recommendation (11); however, 16% of respondents (n = 162) practice use of non-sterile aseptic technique. This is consistent with the use of the evidence-based aseptic-nontouch technique (ANTT), supported by the US Centres for Disease Control and Prevention (CDC) (7) and the UK Royal College of Nursing (RCN) (9). The rationale for clinicians and institutional decision-making between ANTT and sterile technique may be due to a variety of factors, including cost (30).
Our survey indicates that although the majority of respondents reported that the staff nurse is responsible for performing dressing changes, notably 30% are performed by a specialised vascular access team. The role of specialist vascular access teams in performing CVAD dressing changes has been debated, with early studies reporting decreased bloodstream infections with CVAD dressing changes performed by vascular access teams, in comparison to general staff; however, these were multi-interventional studies and thus results cannot be assumed to be directly related to the use of vascular access teams (3133). Since then, a study found equivalence in the risk of CVAD-related septicaemia between expert and general staff undertaking CVAD dressing changes [risk ratio 1.14 (RR); 95% confidence intervals (CI) 0.26-6.42; p = 0.70] (34). The use of vascular access specialists as a means to ensure use of best practice during dressing change is of dispute, considering the resources associated with their training, employment and availability.
Management of skin impairment at CVAD sites
Although there is strong evidence guiding the routine care of CVAD sites, there is a paucity of literature addressing the incidence, aetiology or management of sites with impaired skin integrity (18). Skin damage may be due to infection, moisture, medical adhesives and irritants, leading to conditions such as exit-site infection, skin tears and stripping, tension blisters and contact dermatitis (4, 35, 36).
Given the lack of evidence to guide practice, the reported high level of confidence in caring for these compromised sights was unexpected (Tab. V). As vascular access specialists are often consulted for management of these complicated dressings, it is reassuring to see their confidence in their ability to manage these sites. However, as clinicians who are not vascular access specialists are less confident, there is a need for recommendations to educate clinicians in managing these conditions.
Although transparent film dressings are the reported preferred dressing for the management of sites with intact skin or redness/inflammation, there is an extensive variety of dressing preferences for management of non-intact skin, providing another clear indication of the need for practice guidance in care of compromised sites (Tab. IV). Transparent dressings remain the preferred dressing for the management of sites with macular-papular lesions (rashes), followed by gauze and hydrocolloid dressings. Skin stripping or other adhesive-related injuries such as skin tears are most commonly managed by gauze, then hydrocolloid dressings, followed by transparent dressings. However, it should be noted that consensus recommendations for skin tears caution against the use of hydrocolloid and transparent dressings, as they may cause skin stripping and further damage to the site if not removed properly (37).
Respondents reported treating CVAD sites with drainage similarly to skin injury, preferring gauze first, followed by a preference for clear acrylic absorbent dressings (which are also recommended for management of skin tears), although they are considered off-label use with intravascular devices (37). Clinicians also report using topical haemostatic agents in the presence of bleeding at a site; these agents have been shown to be effective for controlling site bleeding and may be related to a reduction in catheter-associated bloodstream infections (38). In open-text responses regarding management of purulent sites, the common sequence of events involved culture of the site, consultation of a physician and removal of CVADs, with several recommending the use of an antimicrobial dressing. The INS Standards (8) recommend notifying the licensed prescriber of signs of infection, including drainage at the site and culturing those sites with purulent drainage. Clinicians also reported positioning the dressing over the exit site to avoid the skin damage if feasible, using a second dressing to manage the skin injury. Respondents’ comments about allowing the damaged skin to be exposed to air highlights the need for clinicians in vascular access to better understand the principles of wound healing. As Leblanc and colleagues (37) indicated in their best practice recommendations for skin tears, moist wound healing is an integral component of wound healing, proven to accelerate wound healing. Several clinicians also reported consulting a wound care specialist for suggestions, which is a key recommendation in the consensus statement recommendations for management of medical adhesive related skin injuries (MARSI) (35).
Although avoidance of topical agents, other than skin antiseptics and protectants, is a basic principle of vascular access (8), respondents have reported the use of topical agents, antibiotic, steroid ointment/cream, corticosteroid spray, barrier creams particularly in a site unresponsive to traditional management. This wide variety in respondent response may be due to the lack of research to support practice and decision-making in this area.
The availability of a variety of products confounds the clinician's management of skin conditions. As several respondents indicated, they are limited to only gauze and/or transparent dressings. Another respondent indicated that dressing availability is based on the purchasing practices of the hospital. This serves as a reminder to clinicians to advocate for product solution to optimise clinical outcomes.
Education
Competency of the clinician providing site care is the foundation of safe delivery of vascular access and infusion therapy. Each of the internationally recognised guidelines for the management of CVAD (68–11) strongly recommends annual education and competency validation, as this is a recognised measure to improve patient safety. As written policies and procedures are the basis for clinical decision making and professional accountability, it is concerning that 4% of vascular access clinicians responded ‘don't know’ as to whether their organisation has a written policy, procedure or practice guideline. Approximately 90% of clinicians reported not having a procedure or algorithm for management of compromised sites, clearly adding to the extensive variety in management of these sites. These findings serve as a reminder for organisations to have current written CVAD site care policies and procedures and a competency validation process to confirm adherence to these policies in an effort to promote optimal outcomes and protect both patients and clinicians.
Professional certification of clinicians in infusion and vascular access nursing further supports the infrastructure of an organisation's infusion therapy programme. Although over half of the respondents indicate that they have formal certification (e.g. VA-BC, CRNI, CVAA, CPUI), this likely is not representative of the body of infusion and vascular access clinicians. Furthermore, it is concerning to see respondents reporting a lack of awareness of professional certification programmes, again an indicator of the need for professional associations and healthcare organisations to market the availability and value of certification.
Limitations
There are several limitations evident within this study. The response rate is lower than desired, which may be related to the availability of the survey in English only and limited availability of computer/Internet access in some countries. However, representation from 34 countries was achieved, which is high considering the language and internet limitations. With a self-response survey, there is always potential for selection bias and this is reflected in the large number of vascular access specialists responding. The main distribution sources for the survey were professional associations specialising in infusion therapy and vascular access in North America and the percentage of survey respondents with vascular access credentials and practicing in North America reflect this. The results may not be generalisable to the nonspecialised clinician population outside of North America. Response bias is also a concern in that participants may be inclined to respond to the perceived correct answer; however, anonymity of participants has been used to maximise accuracy (39). Although our survey tool has not been externally tested for reliability and validity, the rigorous internal validation process of the survey development strengthens the quality of the survey.
Conclusion
This international, descriptive, cross-sectional survey revealed a varying consistency in routine CVAD site care practices, in terms of skin antisepsis, dressing choice, technique and frequency of change. However, there is a wide variance in the management of CVAD sites with impaired skin integrity, such as rash, skin stripping/adhesive-related injuries and drainage; this is likely a function of the lack of related written policies and procedures and published literature. These results identify implications for practice and research to address the CVAD site care and the diversity in management of CVAD sites with impaired skin. Future research and educational guidelines when considering CVAD management needs to focus on:
Research and practice guidelines addressing the appropriate choice of antiseptic agent, topical agent and dressing type to manage sites with impaired skin integrity;
Research into the efficacy of dressing change upon clinical indication on CVAD sites;
Guidance to enhance capacity of clinicians as advocates for product selection in their organisations;
Stronger guidance on clinical competency validation, including frequency and method of validation; and
Increased patient education materials (verbal, print and other media) and validation of patient education comprehension and performance.
This description of current management practices of CVAD sites with skin impairment will be used to guide the creation of an algorithm by the study authors for the management of CVAD sites with impaired skin integrity. This is the collaborative work of an international advisory panel that convened in May 2014 to address the gap in literature and identify strategies to optimise care of these compromised sites. This will then lay the foundation for an international multidisciplinary panel to develop guidelines for management of skin impairment at CVAD sites.
Footnotes
Acknowledgement
We gratefully acknowledge the contributions of the 1044 nurses, nurse practitioners, physicians, radiology technologists, respiratory therapists and engineers who participated in this survey.
Financial support: This project was funded through a World Congress of Vascular Access (WoCoVA) taskforce and an unrestricted educational grant from 3M as part of a broader CVAD site care project, with technical advice for survey development from E. Tardif, 3M Professional Services Specialist.
Conflict of interest: Daphne Broadhurst has served as a speaker for 3M, Carefusion and BD. Nancy Moureau has served as a speaker and educational consultant with 3M, Access Scientific, Analogic, Angiodynamics, Arrow/Teleflex, Baxter, Carefusion, Cook, Excelsior, Genentech, Hospira, Nexus, Vascular Pathways and Vygon. Amanda Ullman has received funding from CVAD dressing manufacturers (Centurion Medical Products; Carefusion; 3M).
