Abstract
Purpose
As a step to large-scale clinical trials, we conducted a small-scale exploratory study to clarify whether the secondary lumen of the double-lumen (DL) peripherally inserted central catheters (PICCs) could perform as well as the secondary lumen of the DL centrally inserted central catheters (CICCs), with an acceptable complication rate in the perioperative surgery period.
Methods
Forty thoracic esophageal cancer patients requiring central venous catheterization during the perioperative period were assigned to the DL-PICC (4.5-French, 60-cm) group or the DL-CICC (16-gauge, 30-cm) group, with 1:1 randomization. The primary endpoint was the completion rate of continuous catecholamine infusion via the secondary lumen during the observation period.
Results
Thirty-two cases (14 cases in the PICC group and 18 cases in the CICC group) were analyzed. Continuous catecholamine infusion via the secondary lumen was completed in all 32 cases. No major complications related to PICC/CICC placement/maintenance were noted in the groups during the median observation period of 6 days.
Conclusions
The secondary lumen of the DL-PICCs performed as well as the secondary lumen of the DL-CICCs with acceptable safety during the relatively short perioperative period of these thoracic esophageal cancer patients (UMIN Clinical Trial Registry UMIN000008131).
Keywords
Introduction
Because of the extensiveness and invasiveness of radical esophagectomy, the cardiopulmonary status of patients is frequently unstable during the procedure (1, 2). In esophageal cancer patients, catecholamine is used almost routinely for perioperative management to stabilize circulatory dynamics. Intravenous solutions of total parenteral nutrition, extracellular fluid replacement, maintenance fluids, as well as simultaneous administration of catecholamine are usually delivered through a multi-lumen central vascular line, in which each lumen has an independent administrative route (3). Catecholamine is basically administered via the secondary lumen, and not via the side tube of the main lumen of a central vascular line, primarily because the catecholamine line should be independent of the fluid line to maintain a precisely adjusted flow rate during its administration. Multi-lumen centrally inserted central catheters (CICCs) have been used almost routinely in esophageal cancer patients.
The successful use of a peripherally inserted central catheter (PICC) was first reported in the 1970s (4, 5). Subsequently, the use of PICCs has rapidly increased because of their ease of placement, safety and cost-effectiveness compared with conventional CICCs. With the advent of ultrasound guidance, complications, such as an arterial puncture, hemothorax, and pneumothorax, related to subclavian CICC placement have become extremely rare (6); however, complications that occur could be serious (7). The incidence of these complications is virtually 0.0% in PICC placement (8, 9).
Compared with the use of multi-lumen CICCs, the use of multi-lumen PICCs has been limited, primarily because studies showing comparable performance and safety of the secondary lumen of multi-lumen PICCs to those of multi-lumen CICCs are lacking; for example, in esophageal cancer patients, subclavian placement of multi-lumen CICCs is considered to be a mainstream technique in perioperative management. To our knowledge, the performance of the secondary lumen of PICCs/CICCs has not been fully compared. Expanded application of multi-lumen PICCs enables safer perioperative management of esophageal cancer patients.
The aim of this study is to clarify whether the secondary lumen of PICCs could perform as well as the secondary lumen of CICCs, with an acceptable complication rate in patients in the perioperative surgery period. As a step to large-scale clinical trials, we aimed to conduct a small-scale exploratory study.
Materials and Methods
Study design
The study was designed as a first-step exploratory study for large-scale clinical trials; therefore, we set the expected number of cases to 40 at a maximum. Forty thoracic esophageal cancer patients requiring central venous catheterization during the perioperative period were enrolled in this study between November 2012 and October 2013 in our hospital. The inclusion criteria were as follows: a patient requiring central venous catheterization during the perioperative period, and age between 20 and 79 years. The exclusion criteria were as follows: patients with a huge apical bulla or severe pulmonary emphysema, patients who had taken warfarin or an antiplatelet agent within 2 weeks of the placement of the catheter, patients with a past history of pulmonary embolism, deep vein thrombosis or endocarditis, patients in whom a catheter was placed during or after surgery, and patients judged to be inappropriate as a study target. The observation period was from the placement of the catheter to discharge of the subject from the intensive care unit (ICU) to the surgical ward.
After an oral explanation to each patient using description documents, we obtained full written consent from the patients. The patients were randomly assigned to the PICC or the CICC group using a central allocation system. The study was designed with 1:1 randomization and was scheduled to enroll 20 patients in each arm. Radical esophagectomy with gastric reconstruction was performed on all the patients as curative surgery for esophageal cancer. During surgery, a mediastinal lymph node dissection was performed in the left lateral position. Subsequently, abdominal and cervical lymph node dissection and gastric reconstruction were performed in the supine position. The patient was transferred to the ICU with intubation after surgery and was extubated on the following day (POD 1). In the ICU, the patient received critical care and returned to the surgical ward after confirmation of stability of the hemodynamic and pulmonary conditions.
Catheters
As shown in Fig. 1a, b, we used 4.5-French (Fr), 60-cm double-lumen PICC (Covidien Japan, Tokyo, Japan; catalog no. 1945-23-WG) and 16-gauge (G), 30-cm double-lumen CICC (Covidien Japan, Tokyo, Japan; catalog no. 1716-12WBG). The outer diameter of these catheters is comparable (1.5 mm). The PICCs used in this study are non-power injectable. The PICCs and CICCs are made of polyurethane. The secondary lumen of PICCs/CICCs has a smaller cross-sectional area than the main lumen, and is associated with lower flow rates (Fig. 1c, d). In both types of catheters, the cap color and the tube color of the main lumen are different from that of the secondary lumen, and furthermore, the visible length of the secondary lumen is marginally longer than that of the main lumen, so that each lumen could be easily identified before use.
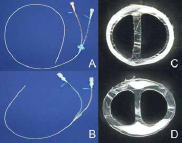
Placement procedures
The PICCs and CICCs were placed by boarded-certified surgeons. During the placement procedure, the surgeons complied with the maximal barrier precautions. CICCs were placed under ultrasound guidance, whereas whether to use ultrasound guidance in PICC placement was at the discretion of the surgeon. Whether to place the catheter in the left side or the right side was also at the discretion of the surgeon.
The catheters were placed using the Seldinger technique. The PICC and CICC placement technique was described elsewhere (10). The surgeon placed the catheter over the guide-wire and put the catheter tip position appropriately in the superior vena cava under fluoroscopic guidance. Exceeded catheter from the catheter exit site was rolled and fixed onto the arm. We preferred the basilic vein in the midarm for PICC placement. When accessible basilic vein was not found, the cephalic vein in the midarm or median cubital vein was selected. Chorhexidine in alcohol was the most appropriate skin antiseptic for the prevention of catheter-related bloodstream infection (11). However, the manufacturer's recommendations for the PICCs/CICCs used in this study prohibited the use of alcohol to avoid damage to catheters. Therefore, the placement site was disinfected with iodine-based disinfectant and covered by a transparent film. The catheter tip position was re-confirmed by a chest X-ray according to “carina” criteria (12).
Maintenance
For the preoperative bowel preparation, the diet was stopped 1 day prior to surgery, and extracellular fluid replacement solution was administered via the main lumen for rehydration. During and after surgery, the extracellular fluid replacement solution or maintenance solution was primarily administered via the main lumen. During and after surgery, dopamine was generally administered via the secondary lumen when indicated. Flushing with heparin solution before and after administration of drugs and before locking the catheter was performed using a 10 ml syringe (11).
Prior to surgery and during surgery, the flow rate of the main lumen was adjusted manually. In the ICU following surgery, the flow rate of the main lumen was adjusted automatically using an infusion pump (Terumo, Tokyo, Japan; catalog no. TE-261). The flow rate of the secondary lumen was adjusted automatically using a syringe driver (Terumo, Tokyo, Japan; catalog no. TE-331S) during surgery and in the ICU after surgery.
Endpoints
The primary endpoint was the completion rate of continuous catecholamine infusion via the secondary lumen during the observation period. The secondary endpoints were complications related to the placement, the procedure time spent during catheter placement, hemorrhage at the placement site, catheter malfunctions, catheter obstructions (the main lumen and the secondary lumen), catheter-related bloodstream infections, phlebitis and venous thrombosis.
Definition of events
Catheter malfunction was defined as follows: inadequate dripping without the use of the infusion pump and the syringe driver, and an event that sounded an alarm on the infusion pump and the syringe driver, except for obstructions of the catheter. Alarms sound under conditions of inadequate dripping, increased line pressure and air mixing in the catheter. The reasons for the alarms could be determined by different types of warning signals. As we previously reported (13), hemorrhage at the placement site was recorded in cases of bleeding after the placement on the basis of a three-grade scale of “fair”, “little” and “none”. The grade “fair” indicates that the hemorrhage required pressure hemostasis for control; the grade “little” indicates limited bleeding noted as clot formation in a dressing; the grade “none” indicates no observable bleeding.
Data collection
Data were recorded on a patient registration form during the observation period. After randomization, the patient's demographic data, including age, gender, height and body weight, were registered. Additional data, including the placement site, the procedure time spent for the catheter placement and complications related to the placement procedure, were prospectively recorded. Any complications and catheter malfunctions were recorded by registered medical practitioners, attending doctors and nurses.
When handling the data, we removed all or part of the information that identifies the individual and conducted connectable anonymization using the assignment identification code instead of the information. The correspondence table was stored securely.
Statistical analysis
The data were expressed as median (range) values. The statistical analysis was performed using JMP® software (SAS Institute, Cary, USA; JMP version 11.0.0). The comparison of endpoints was performed using the t-test and the χ2 test when indicated. A p-value of less than 0.05 was considered to indicate statistical significance.
Results
Of 40 cases, one case (allotted to the PICC group) had unsuccessful catheter placement due to technical reasons, and seven cases (five cases in the PICC group and two cases in the CICC group) did not use the secondary lumen during the observation period; these cases were excluded from the study. In these seven cases, the operations were completed without any problems, and circulatory dynamics were stable during the perioperative periods; therefore, catecholamine administration via the secondary lumen was not used. As a result, 32 cases (14 cases in the PICC group and 18 cases in the CICC group) were subjected to the analysis. In all of the cases of both groups, a peripheral intravenous catheter was placed preoperatively.
The characteristics of patients and procedural data were summarized in Table I, and there was no significant difference in the characteristics of patients between the two groups. In the PICC group, PICCs were placed in the following veins: right basilic vein, eight cases; left basilic vein, three cases; right cephalic vein, one case; right median cubital vein, one case; and left median cubital vein, one case. PICCs were placed under ultrasound guidance in the right basilic vein in two cases. The median observation period was 6 (24–7) days. The median use periods of the secondary lumen were 2 (13) days in the PICC group and 2 (1–4) days in the CICC group (p = 0.2716).
Characteristics of patients and procedural data
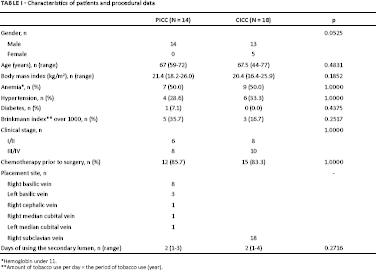
Hemoglobin under 11.
Amount of tobacco use per day × the period of tobacco use (year).
The results are summarized in Table II. The secondary lumen of the double-lumen PICCs performed comparably about the continuous catecholamine infusion via the secondary lumen as well as the secondary lumen of the double-lumen CICCs. In fact, the continuous catecholamine infusion via the secondary lumen was completed in all 32 cases.
Results of the primary and secondary endpoints
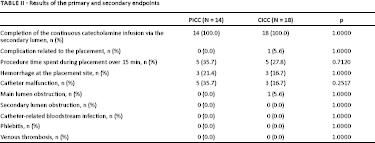
In the CICC group, an accidental arterial puncture was observed in one case. The bleeding was easily controlled with finger compression. No other complications related to catheter placement were noted in the series. Hemorrhage at the placement site was noted in three cases in the PICC group (21.4%) and in three cases in the CICC group (16.7%); all of the cases of hemorrhage at the placement site were graded as “little” and were observed within 24 h after placement. In these three cases in the PICC group, the catheter was placed in the left basilic vein in one case, in the right basilic vein in another case and in the right median cubital vein in the other case. Catheter malfunctions were noted in five cases (inadequate dripping: three cases; pump alarm: two cases) in the PICC group (35.7%) and in three cases (inadequate dripping: two cases; pump alarm: one case) in the CICC group (16.7%). Inadequate dripping prior to surgery was not noted in either group. However, inadequate dripping during surgery when the patient was placed in the left lateral position was observed in three cases in the PICC group and in two cases in the CICC group. In these three cases in the PICC group, the catheters were placed in the right basilic vein. These inadequate drippings were resolved immediately by changing the arm position. In the ICU, alarms of the infusion pumps were triggered in two cases in the PICC group because of temporary inadequate dripping caused by elbow flexure and in one case in the CICC group because of air mixing in the lumen. In these two cases in the PICC group, the catheter was placed in the left basilic vein relatively near the elbow in one case and in the right median cubital vein in the other case. A main lumen obstruction was encountered in one case in the CICC group in the ICU and was not observed in the PICC group. An obstruction of the CICC was noted 7 days after placement. In this case, the secondary lumen was used for the administration of the catecholamine infusion at days 5 and 6 after placement and was used as the fluid/nutritional route 7 days after the placement. A secondary lumen obstruction was not encountered in any of the cases.
As shown in Table III, there was no significant difference in the maximum dosing rate or in the intraoperatively administered doses of catecholamine solution via the secondary lumen between the two groups.
Maximum dosing rate and intraoperatively administered doses of catecholamine solution via the secondary lumen of PICCs and CICCs

Discussion
The use of PICCs in clinical practice has expanded widely. However, in esophageal cancer patients, the use of multi-lumen PICCs during the perioperative period remains limited mainly because there is a lack of literature demonstrating comparable performance and safety of the secondary lumen of multi-lumen PICCs to those of multi-lumen CICCs. The secondary lumen of the double-lumen central vascular line has a lower flow rate due to smaller cross-sectional area than the main lumen. Almost all of the previous reports about PICCs focused on the function of the main lumens, and evaluations of the secondary lumen have not been fully conducted. Our result about the secondary lumen relieves the anxiety regarding whether the long length of the catheter could negatively affect the flow rate. Temporary inadequate dripping was observed in several cases during and after surgery; however, all of these situations could be resolved immediately and there was no influence on the primary outcome. On the basis of these results, we conclude that there was no difference in the administration ability of the secondary lumen between the PICCs and CICCs, at least during the relatively short perioperative period. Furthermore, in the two groups, the complication rate was acceptable.
Previous literatures stated about the use of double-lumen PICCs in critical care (3, 14). However, “critical care” does not usually include the intraoperative period, whereas the “perioperative period” includes the preoperative, intraoperative and postoperative periods. We focused on the intraoperative period of esophagectomy and demonstrated the usefulness of double-lumen PICCs during the perioperative period, and furthermore, we evaluated the performance of catheters in the decubitus position.
Power-injectable PICCs have many advantages in critical care because of their very high flow rates and easy monitoring of central venous pressure (3, 14); however, we chose to use non-power injectable PICCs in this study. This is because power-injectable PICCs have not been approved by Japanese regulatory authority yet, therefore not available in the Japanese market. This may partially reflect the short history of the use of PICCs in Japan. The use of PICCs has increased in Japanese clinical practice primarily in the second half of the 2000, and PICCs remain used primarily for total parenteral nutrition and not for critical care. Thus, there is a wide gap in the use of power-injectable PICCs between Japanese and Western countries. In the near future, power-injectable PICCs will be introduced in Japanese clinical practice and a further study using power-injectable PICC will be necessary.
Although some PICCs were placed in the cephalic vein or median cubital vein, these veins have a higher risk of catheter-related complications than does the basilic vein (15). In one case in the PICC group, the catheter was placed in the basilic vein relatively near the elbow. Furthermore, one case in the PICC group had unsuccessful catheter placement. These were obviously related to the lack of ultrasound guidance. In recent reports, the use of ultrasound guidance is strongly recommended for any central venous access (11, 16). However, in Japanese clinical practice, CICC placement under ultrasound guidance has been routinely performed, whereas PICC placement under ultrasound guidance has not been 100% routinely performed. Thus, many PICC placements are still being performed without ultrasound guidance. Considering the current situation, we did not prescribe the routine use of ultrasound guidance in the protocol of this study and, as mentioned above, the use of ultrasound guidance in PICC placement was at the discretion of the surgeon. However, the placement of catheters without ultrasound guidance has already become obsolete. Therefore, we must introduce the routine use of ultrasound guidance in PICC placement and educational instruction of this technique must be implemented. PICC placement under ultrasound guidance in a deep vein of the upper midarm (basilic or branchial vein) may be necessary to avoid catheter-related complications (14, 17).
Temporary inadequate dripping during surgery with the patient in left lateral position was noted in three cases in the PICC group. This temporary inadequate dripping was resolved immediately by changing the arm position; however, surgeons and anesthesiologists in the operating room should take care regarding the status of dripping during surgery, particularly when the patient is placed in the decubitus position.
Our study has the following major shortcomings: a small numbers of cases and a very short usage time, which markedly limits generalizing our results to clinical practice. The study was designed as a first-step exploratory study for large-scale clinical trials; therefore, the enrollment number was designed to be only 40 patients. In previous reports (18–21), the catheters were used mainly for total parenteral nutrition and chemotherapy; thus, the observation periods were relatively long in those studies. For example, the observation period of Pikwer's meta-analysis was 5-174 days (18). In our perioperative series, the median observation period was 6 days, which was much shorter than the above reports. The median use periods of the secondary lumen were also very short (2 days in the PICC/CICC group). We focused on short-term observations in the perioperative period of thoracic esophageal surgery because that type of usage is most typically seen in daily practice. However, another study is necessary to evaluate the performance of double-lumen PICCs over longer periods. Our study is a pilot study; therefore, to generalize these results in clinical practice, a continuous accumulation of clinical cases would be necessary.
The secondary lumen of the double-lumen PICCs performed as well as the secondary lumen of the double-lumen CICCs with acceptable safety during the perioperative period of thoracic esophageal cancer patients. Increased use of multi-lumen PICCs might lead to safer perioperative management of esophageal cancer patients. Especially, esophageal cancer patients with poor lung function may be good candidates for the use of double-lumen PICCs.
Footnotes
Acknowledgement
The authors thank the attending doctors and nurses who participated in this study and the colleagues who advised us in conducting this study.
This study was approved by the Clinical Research Ethics Review Board of our institution (approval number: #12076) and performed in accordance with the Declaration of Helsinki. Written informed consent was obtained from all patients.
Financial support: None.
Conflict of interest: This study was partially supported by Covidien Japan (Tokyo, Japan). The authors declare no further conflict of interest.
