Abstract
Purpose
Conventional safety intravenous (IV) catheters prevent blood exposure during catheter placement but blood leakage from the unconnected catheter still frequently occurs. In the current study a new safety IV catheter equipped with a blood leakage control septum was assessed under routine clinical conditions.
Methods
This prospective observational trial was conducted at the KKRN (Katholisches Klinikum Ruhrgebiet Nord), Germany, September/October 2012. Peripheral IV access was established in presurgical patients using either the investigational (“IS3”) or a conventional safety IV catheter (“IS”). Incidence of blood leakage during placement and subsequent (dis-)connection procedures, duration of placement as well as handling conditions were compared.
Results
A total of 200 IV accesses were established (n(IS3) = 102 and n(IS) = 98). Blood leakage during catheter placement (4.9% (IS3) vs. 61.2% (IS); p<0.001) and blood contaminations (3.9% (IS3) vs. 14.3% (IS); p = 0.01) were significantly reduced for IS3. All blood leakages observed with IS3 were due to improper technique. No blood leakage occurred during repeated (dis-)connections of IS3 (blood leakage IS: 74%). Using IS3, vein compression was not required (no compression: 98%) and duration of catheter placement was significantly shorter (t(IS3) = 69.6 ± 22.4 s vs. t(IS) = 85.2 ± 28.2 s; p<0.001).
Conclusions
The investigational IV catheter effectively prevented blood leakage, thereby reducing contamination risk and workload associated with cleaning. Omission of vein compression facilitated and shortened IV catheter placement.
Keywords
Introduction
Exposure to blood and body fluid bears the risk of contamination and infection with blood-borne pathogens such as hepatitis or human immunodeficiency virus (1). Healthcare professionals working at the patients' bedside or the operating theatre are especially at risk of percutaneous or mucocutaneous blood exposure (2). During placement of intravenous (IV) catheters blood exposure can occur via needlestick injury (i.e., percutaneous exposure) or blood splashes and blood spillage from the unconnected catheter (i.e., potential contamination of mucous membranes, conjunctiva or nonintact skin). Percutaneous exposure during IV catheter placement can effectively be prevented using safety-engineered devices, especially those equipped with a passive safety mechanism (3, 4). However, these devices do not prevent blood splashes and blood spillage from the unconnected catheter that occur frequently due to insufficient compression of the punctured vein during insertion or subsequent (dis-)connection procedures (5, 6). Although the infection risk via mucocutaneous exposure during IV catheter placement is considered to be very low (7), it is desirable to protect healthcare professionals from any kind of blood exposure and to avoid blood contamination of the surroundings in order to reduce workload associated with cleaning and disinfection. Therefore, peripheral IV catheters have been developed to reduce blood leakage, but only few studies are published that investigate the efficacy of these devices (8).
The current study was designed to assess the performance of a recently marketed passive safety IV catheter under routine clinical conditions. This IV catheter is equipped with a bidirectional septum located in the catheter hub that was developed to control flow of blood and to prevent blood leakage from the unconnected catheter. The septum closes immediately after removal of the introducer needle or subsequent disconnections and opens upon connection to a luer device (e.g., infusion line or three-way stopcock) allowing in- and outward fluid flow. In order to assess efficacy and clinical acceptability of the investigational IV catheter, occurrence of blood leakage during IV catheter placement and subsequent connection and disconnection procedures as well as handling properties and ease of use were investigated in comparison with a conventional passive safety IV catheter.
Materials and Methods
This study was a prospective observational (noninterventional) clinical trial conducted over a 2-month period (September–October 2012) at the KKRN (Katholisches Klinikum Ruhrgebiet Nord), Germany. It was designed in accordance with the ISO standard for clinical investigations of medical devices for human subjects (ISO 14155:2011) and aimed to compare two CE-certified peripheral IV catheters with respect to blood leakage and handling properties. The hospital's Ethics Committee was informed about all aspects of this study.
The study was conducted with consecutive presurgical patients in need of peripheral IV access. Exclusion criteria were not defined. Peripheral IV access was established using over-the-needle peripheral IV catheters with a passive needle-shielding mechanism to prevent needlestick injuries. Both devices came with the same kind of introducer needle. In contrast to the reference product (B. Braun, Introcan Safety®, “IS”), the investigational product (B. Braun, Introcan Safety® 3, “IS3”) was equipped with a bidirectional septum located in the catheter hub that prevents blood flow from the unconnected catheter. IV catheters were placed by anesthetists or nursing staff in the operation theater according to the hospital's routine and the respective “Instruction for Use.” Participating personnel was trained in handling of both IV catheters. During training it was pointed out that correct functioning of IS3 requires removal of the introducer needle and prevention of high intravascular pressure.
The primary objective was to investigate the incidence of blood leakage during IV catheter placement and subsequent catheter connection and disconnection procedures such as blood sampling or change of infusion line (hereinafter referred to as catheter manipulations). Blood leakage and onset of leakage (immediately, within 5-15 s, after more than 15 s) were recorded as well as blood contaminations of the needle and the surrounding area. The secondary objective of this study was ease of IV catheter placement assessed via a user survey about IV catheter design and handling, and time needed for IV catheter placement.
All data were documented in the case report form that was monitored for completeness and accuracy. All data given are presented as mean ± SD. Sample size was estimated according to the “rule of three” in order to detect an incident with an occurrence of 1.5% and an alpha error of 0.05. Statistical analysis was performed with the statistical software package SPSS. Frequencies were analyzed using the nonparametric Chi-Square test and continuous data were tested using the Mann-Whitney U-test. All statistical tests were two-tailed; p values <0.05 were considered statistically significant.
Results
General conditions of IV catheter placement
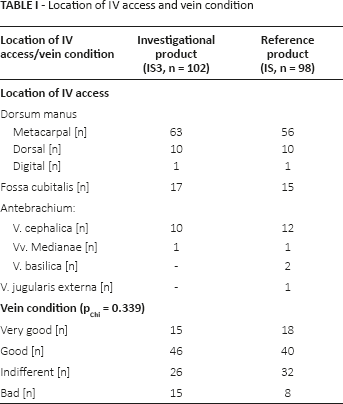
A total of 200 peripheral IV accesses were established using either the investigational (n(IS3) = 102) or the reference IV catheter (n(IS) = 98). Participating personnel was well experienced as reflected by the number of IV catheters usually placed per week (n(IV catheters per week) = 22 ± 12). Location of peripheral access and vein condition was comparable for both products (Tab. I).
Location of IV access and vein condition
Blood leakage during IV catheter placement
The incidence of blood leakage was significantly lower for IS3: Blood leakage was observed during 5 of 102 catheter placements (4.9%) using IS3 and during 60 of 98 catheter placements (61.2%) performed with IS (p<0.001, see Tab. II for onset of blood leakage). All blood leakages observed with IS3 were due to improper technique. In one case blood leakage occurred alongside the stylet through the indwelling catheter because the introducer needle was not removed immediately after venipuncture and closure of the septum was not possible. In three cases the tourniquet was not released before removal of the introducer needle so that blood pressure exceeded the capacity of the integrated septum and blood leakage occurred immediately after removal of the needle through the unconnected catheter hub. In another case catheter connection to a luer device was delayed and blood leaked through the unconnected catheter hub (onset of blood leakage > 15 s after removal of the introducer needle). Blood contaminations of needle, safety shield and the surroundings were significantly less frequently reported for IS3 (Tab. II).
Incidence and onset of blood leakage during IV catheter placement
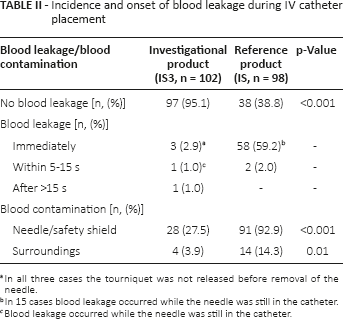
In all three cases the tourniquet was not released before removal of the needle.
In 15 cases blood leakage occurred while the needle was still in the catheter.
Blood leakage occurred while the needle was still in the catheter.
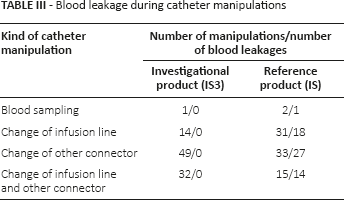
Blood leakage during catheter manipulations
A total of 177 catheter manipulations have been reported (Tab. III). In 160 cases the catheter was manipulated once (n(IS3) = 92 and n(IS) = 68), in seven cases twice (n(IS3) = 2 and n(IS) = 5) and in one case more than twice (IS). No blood leakage was detected during manipulations of IS3, while blood leakage occurred immediately after disconnection in 74% of the manipulations of IS.
Blood leakage during catheter manipulations
Ease of IV catheter placement and fixation
Successful IV catheter insertion without any problems at the first go was reported in 91.2% and 84.7% of the placements with IS3 and IS, respectively (p = 0.158). Several attempts of puncturing were required in 10 cases (n(IS3) = 3 and n(IS) = 7). Difficulties to introduce the IV catheter (n(IS3) = 9 and n(IS) = 15) were reported to be predominantly due to bad vein condition. The only adverse device effect reported was a hematoma at the puncture site (IS). Vein compression during removal of the needle proximal to its tip was required in 2 of 102 IV catheter placements performed with IS3 and in 94 of 98 cases with IS (p<0.001); 80% of the investigators that did not compress the vein stated that omission of vein compression improved the procedure of IV catheter placement since both hands could be used during removal of the needle and catheter connection. Time needed for IV catheter placement was significantly shortened by ~15 s when IS3 was used (í(IS3) = 69.6 ± 22.4 s vs. t(IS) = 85.2 ± 28.2 s, p<0.001).
Discussion
The current study shows that the integrated septum of the investigational IV catheter effectively prevents blood leakage from the unconnected catheter hub under routine clinical conditions. Using the investigational IV catheter, blood leakage during insertion was reported in 4.9% of the placements compared to 61.2% of the placements performed with the conventional IV catheter. All blood leakages observed during insertion of the investigational IV catheter were caused by improper technique. Thus, the integrated septum worked reliably and completely prevented blood leakage from the unconnected catheter hub in case the following safety recommendations were respected: release of the tourniquet before removal of the introducer needle, removal of the introducer needle immediately after venipuncture and prompt connection of the catheter to a luer device after removal of the needle. According to the manufacturer, the septum has meanwhile been further improved so that blood leakage is now also prevented in case of delayed catheter connection.
No blood leakage was observed during subsequent manipulations of the investigational IV catheter in contrast to an incidence of blood leakage of 74% during manipulations of the conventional IV catheter. The integrated septum even prevented blood leakage after repeated catheter manipulations. To our knowledge, no other IV catheter currently available offers this safety feature.
Blood contaminations of the surrounding area were significantly reduced when the investigational IV catheter was used. This minimizes the risk of blood exposure for healthcare professionals and helps to markedly reduce the workload associated with cleaning and disinfection.
The study further shows that the integrated septum obviates the need for vein compression, thereby improving the process of IV catheter placement as stated by the participating clinicians. The time needed for IV catheter placement was significantly shorter for the investigational IV catheter, presumably due to omission of vein compression. Taken together the investigational IV catheter considerably reduced the workload associated with IV catheter placement by shortening the procedure itself and reducing the extent of cleaning procedures.
No needlestick injuries were reported confirming that the passive needle shielding mechanism of both IV catheters effectively protects healthcare professionals from percutaneous blood exposure.
In the current study blinding was not possible due to obvious differences in appearance of both devices. Further, the sequence of the IV catheters tested was not randomized but occurred in a chronological order (reference IV catheters first followed by investigational IV catheters). These limitations are not considered to largely influence the findings of the study. Occurrence of blood leakage is a clearly defined parameter and a potential training effect during the use of the reference product seems negligible since all investigators were well experienced in the technique of IV catheter placement and trained on both IV catheters.
In conclusion, the IV catheter tested in the current study provides a significant technical improvement as the integrated septum completely prevents blood leakage from the unconnected catheter hub in case of proper use. It thus contributes to protect healthcare professionals from mucocutaneous blood exposure and helps minimizing workload associated with blood contaminations of the surroundings. To ensure correct functioning of the device, results from this study lead to the recommendation to release the tourniquet before removal of the needle, to remove the needle immediately after venipuncture and to connect the catheter promptly after removal of the needle. This study further showed that the integrated septum not only prevents blood leakage but also obviates the need to compress the punctured vein, thereby improving and shortening the procedure of IV catheter placement.
Footnotes
Acknowledgments
The authors would like to thank Heike Fries, PhD, B. Braun Melsungen AG, for planning and coordination of all study activities and Katharina Achilles, PhD, for providing medical writing services.
Conflict of interest: Prof. Dr. Haeseler has received honorarium from B. Braun Melsungen AG for consultation and presentation of results.
