Abstract
Background and aim
Early diagnosis of premalignant or malignant pancreatic cysts is essential to improve prognosis. Sampling of pancreatic cyst fluid by fine-needle aspiration during endoscopic ultrasonography (EUS) enables cytopathological examination combined with biochemical analysis. This study aimed to provide an aid based on biological markers for the preoperative management of patients with pancreatic cysts.
Methods
Pancreatic fluids obtained by EUS-guided fine-needle aspiration from 115 patients with cystic lesions were assayed for amylase, lipase, carcinoembryonic antigen (CEA), CA 19-9 and CA 72-4. In addition, chromogranin A (CgA) and neuron-specific enolase (NSE) were measured in 28 fluid samples.
Results
ROC curve analysis of the different markers for diagnosis of mucinous cysts showed that CEA had the highest area under the curve (0.93, 95% CI 0.87-0.97), with a sensitivity and specificity of 89% and 93%, respectively, at the cutoff value of 317 μg/L. The CgA and NSE concentrations in 5 NET cysts (median values of 210 [63-492] and 68.5 [9-496] μg/L, respectively) were higher than in 23 other cysts (median values of 8 [7-828] and 2.7 [0.5-35.8] μg/L, respectively) (p = 0.0015 and p = 0.0045, respectively).
Conclusions
CEA is the best marker for identifying a cyst as mucinous. In case of low levels of CEA, our results suggest that CgA and NSE measurements may be helpful in the diagnosis of a neuroendocrine tumor and therefore deserve further investigation.
Keywords
Introduction
Detection of pancreatic cysts has become much more frequent over the last few years, mainly as a result of improved abdominal imaging techniques. Several studies have reported a prevalence of 2.4%-2.6% with magnetic resonance imaging (MRI), sometimes reaching 13.5% (1, 2). The frequency in the general population has been estimated at 1% (3, 4). This broad range of prevalence values may be partly due to the criteria of inclusion of the patient population, in particular whether or not patients with potential pancreatic disease were excluded from the analysis. Pancreatic cysts are discovered incidentally in many cases and have different clinical signs and malignant potential. Some are totally asymptomatic, others are detected in the context of pancreatitis, a mass syndrome, or an alteration of the general state of the patient. Although the majority are benign, early diagnosis and formal identification of the histological type is critical because of the potential malignant nature of some cyst types that can be precursors of pancreatic cancer and would therefore require surgical treatment or closer follow-up.
The histological classification distinguishes benign cysts such as pancreatic pseudocysts (PCs) and serous cystadenomas (32%-39%) from malignant or premalignant cysts that include mucinous cystic neoplasms (10%-45%), intraductal papillary mucinous neoplasms (IPMNs) (21%-33%) (5) and solid pseudopapillary neoplasms (SPNs). Occasionally, solid tumors such as pancreatic neuroendocrine tumors (NETs) may undergo cystic degeneration and present as a solid cystic mass (6). Although they were once thought rare, these cystic lesions have been found to account for 13%-17% of pancreatic NETs (7, 8). Although the final diagnosis is based on histopathological examination of surgical specimens, cytopathological examination of pancreatic cyst fluid obtained by puncture during endoscopic ultrasonography (EUS) is widely used, with a reported sensitivity of approximately 50% for the distinction between mucinous and nonmucinous pancreatic neoplasms (9, 10), and 71% for the diagnosis of cystic pancreatic NETs (8). In addition, analysis of biological markers such as amylase, lipase, carcinoembryonic antigen (CEA), and the tumor-associated mucins CA 19-9 and CA 72-4 in cyst fluid is valuable in patients whose initial diagnosis is uncertain and also for the follow-up of pancreatic cysts (11-13). When combined with histological analysis, they allow better identification of the malignant nature of a cyst (14, 15). The existing biological algorithms show that CEA is the most accurate tumor marker for diagnosing a mucinous pancreatic cystic neoplasm, although the accuracy and cutoff levels vary between laboratories (16-18). However, if a cyst is not mucinous, the risk of malignancy is still not zero and cystic NETs pose a problem in clinical diagnosis.
The objective of this study was to establish retrospectively the cutoff values for the usual biological parameters in our laboratory for diagnosis of mucinous pancreatic neoplasms. Since chromogranin A (CgA) and neuron-specific enolase (NSE) are well-characterized biological markers that allow the diagnosis of NETs, we studied the utility of measuring them in cyst fluids.
Methods
Collection of pancreatic cyst fluids
One hundred and fifteen patients with cystic lesions of the pancreas detected by transabdominal ultrasonography or computed tomography (CT) were referred to the Department of Digestive Endoscopy at the Cochin Hospital, Paris, France, between January 2012 and May 2016 and underwent EUS. The procedure was in accordance with the recommendations of the local ethics committee and all patients gave their informed consent.
Pancreatic cyst fluids were collected by EUS-guided fine-needle aspiration (FNA). The majority of the fluid was examined by the same cytopathologist for every patient and the rest was analyzed for biochemical markers.
Based on imaging and cytopathology, the pancreatic cysts were classified into 5 categories: PC, mucinous cystadenoma/cystadenocarcinoma (MCA), IPMN, NET, and SPN. Twenty-six tumors are designated as “others” in Table I, including 17 tumors that could not be classified, 6 serous cystadenomas, and 3 pancreatic cystic lymphangiomas.
Characteristics of the patients
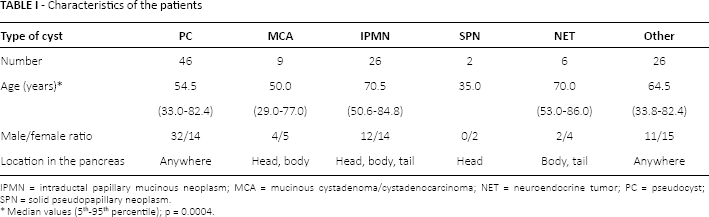
IPMN = intraductal papillary mucinous neoplasm; MCA = mucinous cystadenoma/cystadenocarcinoma; NET = neuroendocrine tumor; PC = pseudocyst; SPN = solid pseudopapillary neoplasm.
Median values (5th-95th percentile); p = 0.0004.
Quantification of tumor markers and enzymes
CEA, CA 19-9 and CA 72-4 were measured using immunoassays on a Cobas E analyzer (Roche Diagnostics). Serial dilutions in an appropriate reagent were carried out for linearity studies and to rule out the Hook effect. A pool of cyst fluids was used as a diluent of a serum with high levels of markers for recovery studies. Amylase and lipase activity were detected by enzymatic colorimetric methods on a Cobas 8000 analyzer (Roche Diagnostics).
In some cases, when the volume of the fluid sample was sufficient, additional assays were performed for NSE and CgA with a Cobas E analyzer and the time-resolved amplified cryptate emission method on the Kryptor system (B.R.A.H.M.S GmbH, Thermo Scientific) (19), respectively, on samples stored at −30°C for up to 1 year before analysis. The limit of detection of the CgA test was 6.24 μg/L and the 20% CV value was 9.04 μg/L. The corresponding values for NSE were 0.05 and 0.25 μg/L.
Statistical analysis
The results are given as medians (5th-95th percentile) and were analyzed using the Kruskal-Wallis or Mann-Whitney test as appropriate to determine differences between groups. Areas under the curve (AUCs) estimated from receiver operating characteristic (ROC) curves were established to define optimal cutoffs. We used a 5% significance level in all comparisons. Statistical analyses were performed with MedCalc version 12.7.7.0.
Results
The median age of the study patients was 61.0 [34.3-83.5] years; there were 61 men (median age 62.0 years [34.1-82.9] and 54 women (median age 61.0 years [34.2-83.6]). As presented in Table I, the majority of cysts were PCs. Of these 46 patients, 14 had a history of pancreatitis and one with chronic alcoholism was managed in anticipation of pancreatitis. Malignant or potentially malignant cysts, including MCA, IPMN, NET and SPN, were diagnosed in 43 (37%) patients. The 6 groups of patients differed significantly in terms of age (p = 0.0004): patients with IPMNs were older than those with MCAs, SPNs and PCs and patients with NETs were older than those with SPNs and MCAs.
Figure 1 shows the concentrations of the 5 usual markers in fluids obtained from the different types of cysts. They all differed significantly among the 6 types. CEA levels were higher in MCA and IPMN fluids than in the other 4 cyst types. CA 72-4 and CA 19-9 levels were higher in IPMN fluids than in other cyst types except MCA. Amylase and lipase levels were higher in PC fluids than in all other fluids.
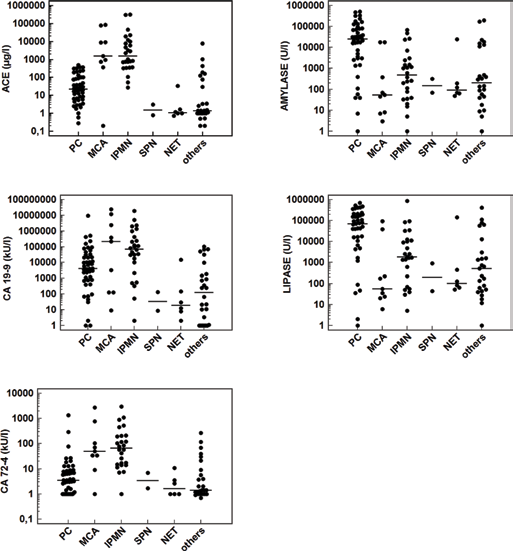
Concentrations of CEAa, CA 19-9b, CA 72-4a, amylasec and lipased in cyst fluid from patients with pancreatic cysts. Individual concentrations are presented as dots and are plotted logarithmically to accommodate extreme values. The horizontal line represents the median for each marker in each group. The Kruskal-Wallis test was used for comparison between groups.
All markers differed significantly between mucinous and nonmucinous cysts (Tab. IIA). ROC curve analysis for diagnosis of mucinous cysts showed that the AUCs obtained with CEA (0.93) and CA 72-4 (0.87) were higher than that obtained with CA 19-9 (0.76) (Tab. MB). The sensitivity and specificity for CEA were 89% and 93%, respectively, at the cutoff value of 317 μg/L.
Marker analysis in cyst fluids
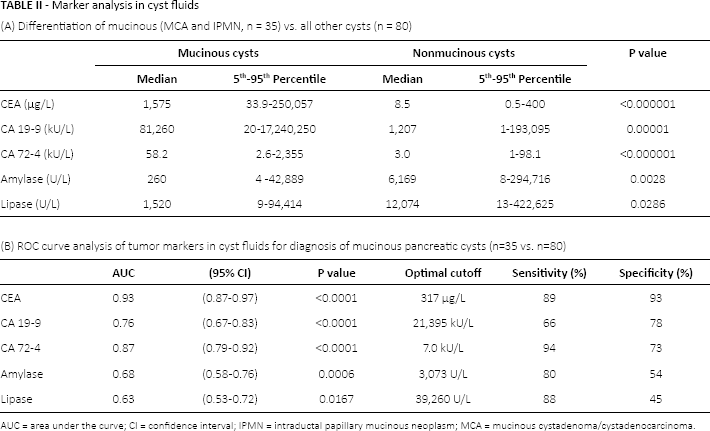
AUC = area under the curve; CI = confidence interval; IPMN = intraductal papillary mucinous neoplasm; MCA = mucinous cystadenoma/cystadenocarcinoma.
In addition, recovery studies performed for tumor marker analysis in pancreatic cyst fluids are presented in Table III. A pool of cyst fluids was used as a diluent of 2 sera with high levels of CEA and CA 72-4 for the former and high levels of CgA and NSE for the latter. Conversely, the pool of cyst fluids was diluted in zero standard for CA 19-9 measurements. The percentage recovery ranged between 87% and 106%. Then CgA and NSE were measured in 5 of 6 NETs (median values of 210 [63-492] and 68.5 [9-496] μg/L, respectively) and in 23 non-NET cysts (5 PCs, 7 mucinous cysts, 2 serous cystadenomas, 3 pancreatic cystic lymphangiomas and 6 cysts with inconclusive results) (median values of 8 [7-828] and 2.7 [0.5-35.8] μg/L, respectively) (Fig. 2). Both markers were higher in NETs (p = 0.0015 and p = 0.0045, respectively). Within the non-NET group, the highest levels of CgA (2,105, 140 and 57 μg/L) were found in the pancreatic cystic lymphangiomas and were significantly higher than in the other non-NET cysts (p = 0.0039).
Recovery study
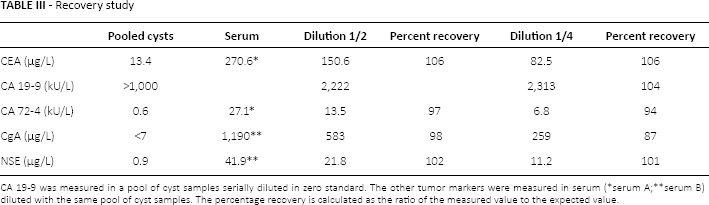
CA 19-9 was measured in a pool of cyst samples serially diluted in zero standard. The other tumor markers were measured in serum (
serum A;
serum B) diluted with the same pool of cyst samples. The percentage recovery is calculated as the ratio of the measured value to the expected value.
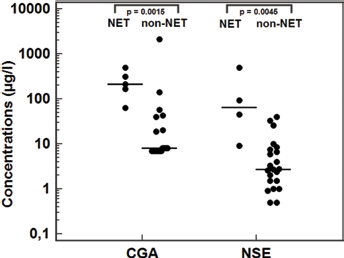
Concentrations of chromogranin A and neuron-specific enolase in cyst fluid from 28 patients with pancreatic cysts. CgA = chromogranin A; NET = neuroendocrine tumor; NSE = neuron-specific enolase.
Discussion
Our study showed that the concentrations of CEA, CA 19-9 and CA 72-4 in cyst fluid were higher in mucinous cysts than nonmucinous ones. CEA alone enabled the 2 types of cysts to be distinguished when a cutoff value of 317 μg/L was used, with a sensitivity of 89% and a specificity of 93%. On the other hand, low concentrations of CEA and CA 19-9 were found in SPNs and NETs, although these have malignant potential. Despite the fact that technological progress in abdominal imaging, including CT, MRI and EUS, has led to better detection of pancreatic cysts, the specificity of these techniques remains limited and dependent on the skill of the operator, and the differential diagnosis of benign and malignant cysts remains difficult. On the other hand, EUS-guided FNA cytology combines high specificity with limited sensitivity due to the small numbers of cells recovered but has the advantage of allowing the analysis of biological markers in the aspirates to yield complementary information. Approximately 0.5 to 1.0 mL is required to run all tests. A number of studies reported incomplete data, particularly for CA 19-9 because, in cases of pancreatic lesions with an extremely low volume of fluid, the authors chose to perform the analysis of CEA first and amylase second (17). In our study, all fluid samples underwent complete analysis; those with a volume of less than 0.5 mL were assayed after dilution in physiological saline for enzyme assays and in a special diluent for immunoassays. In these conditions, CEA was confirmed to be the most accurate marker for diagnosing a mucinous pancreatic cyst. The cutoff values among different laboratories vary between 192 and 800 μg/L due to the use of reagents obtained from different manufacturers (Abbott Laboratories, Beckman Coulter, Roche Diagnostics) and probably also because of the size of the cohort in the study (17, 20, 21).
On the other hand, mucin CA 19-9 is not expressed in people with negative Lewis a blood group, who represent 7% to 10% of the whole population. This situation can be suspected in case of undetectable CA 19-9 in association with the presence of the other mucin, CA 72-4, which, in addition, is more accurate than CA 19-9 for diagnosing a mucinous cyst. Our finding that amylase and lipase levels, which indicate pancreatic duct communication, were higher in PCs than in mucinous cysts confirmed other studies (22, 23). However, the large overlap in the values limited the use of this marker in differentiating these cyst subtypes. Based on the cutoff of CEA at 317 μg/L, one of the unclassified cysts in our study (CEA 7,691 μg/L; CA 19-9 100,815 kU/L; amylase 172,645 U/L) would definitely be categorized as mucinous.
In recent years, there has been growing interest in DNA expression profiling methods (24, 25). Thus, various genetic alterations have been described in association with malignant cysts, including alterations in KRAS, GNAS, TP53 and CTNNB1. A metabolomic approach was also proposed which found glucose and kynurenine to have potential for differentiating mucinous from nonmucinous cysts (26). However, none of these markers has shown superior diagnostic accuracy to CEA.
The remaining types include SPNs and NETs are other cyst types which also have malignant potential. NETs may be difficult to distinguish from more common cystic neoplasms on the basis of morphological features observed by means of CT or EUS. The diagnosis is usually established on cyst fluid (obtained by FNA) that contains plasmacytoid cells with a round to oval, eccentric nucleus, and coarse stippled chromatin. It can be confirmed by histochemistry with anti-CgA or synaptophysin antibodies staining chromatin (8). However, obtaining sufficient cells for diagnostic cytology is often difficult because the number of cells aspirated in pancreatic cyst fluid is often small. On the other hand, little information about biological parameters is available. Thus, very low levels of CEA have been reported in cystic NETs (median 1.25 μg/L) in a series of 12 cases (27). Only one short report mentions an increased level of CgA (138 μg/L) measured in cyst fluid together with low levels of amylase and CEA (28). In the present study we have shown that tumor marker analyses could be reliably achieved in pancreatic cyst fluids provided the matrix effect is controlled, and that analyzing fluid for CgA and NSE may add helpful information in case a NET is suspected but needs further investigation. Unexpectedly, we also found high levels of CgA contrasting with low levels of NSE in cystic lymphangiomas. However, these very rare benign pancreatic neoplasms are typically filled with chylous white fluid rich in triglycerides, which helps the differential diagnosis (29).
In conclusion, in case of nonmucinous cysts particularly with low CEA levels that may erroneously suggest a benign lesion, our results encourage the measurement of CgA and NSE in a larger number of cases to determine the neuroendocrine nature of the cyst and thus provide an aid to the management of these patients.
Footnotes
Abbreviations
Financial support: None.
Conflict of interest: The authors declare they have no conflict of interest related to this article.
