Abstract
Background
Annexin A1 has been implicated in various tumor types, but few studies have investigated its involvement in lung cancer. The purpose of this investigation was to quantify the annexin A1 level in bronchoalveolar lavage fluid (BALF) and analyze its usefulness in lung cancer diagnosis.
Methods
Annexin A1 expression was measured by immunohistochemistry and enzyme immunoassay. The sensitivity and specificity of annexin A1 for distinguishing lung cancer were determined by receiver operator characteristic (ROC) curves.
Results
Tumor tissues, BALF and serum of patients with lung cancer contained higher levels of annexin A1 than those of the control group of patients with benign lung diseases. Moreover, an increased level of BALF annexin A1 was closely correlated with lymphatic invasion and malignant progression of lung cancer. The sensitivity and specificity of BALF annexin A1 for distinguishing lung cancer were 94.2% and 90.2%, respectively.
Conclusions
Increased annexin A1 in BALF was correlated with lymphatic invasion and malignant progression of lung cancer, suggesting that it could be an indicator for discerning lung cancer and predicting outcome.
Introduction
Lung cancer has a high incidence rate in China as a result of ongoing environmental pollution and food safety problems. Although new chemotherapy drugs and molecular targeted drugs have been used in the clinic for many years, the prognosis of lung cancer is still poor. The main reason is that we are still not able to diagnose lung cancer at an early stage, so that many patients will have lost the optimal timing of treatment by the time the diagnosis of this terrible disease has been confirmed. The identification of stable biomarkers is conducive to promoting molecular diagnosis and opening up new targeted therapies for lung cancer (1). Thus, more effective molecular markers and diagnostic tools that can facilitate the diagnosis, prognosis, and therapy of lung cancer are urgently needed (2, 3). However, lung cancer has been short of specific and effective tumor markers so far (4).
Electronic bronchoscopy has been widely applied in the diagnosis and treatment of lung cancer. At present it is considered the main and most effective method for diagnosing patients with a suspicion of a malignancy in the lung. The diagnosis often depends on a tumor tissue biopsy; however, sometimes it is difficult to obtain lung cancer tissues for pathological diagnosis. The bronchoscopy operator may sample bronchoalveolar lavage fluid (BALF) for further testing to help in diagnosis. BALF has been regarded as an alternative resource for detecting biomarkers of lung cancer because of its vicinity to tumor tissues and cells. Compared to biopsy material, it can be obtained noninvasively and thus reduce the complication and operative risk (5). Nowadays, patients suspected of having malignant tumors often undergo electronic bronchoscope examination and bronchoalveolar lavage (BAL) has become a routine method. Research has shown that changes of some proteins in BALF could reflect the lung–cancer-induced immune response in the body. Thus, identification and measurement of specific proteins in BALF may produce useful information and possible clues for distinguishing lung cancer from nonmalignant conditions (6).
In a previous work we reported that elevated annexin A1 in lung cancer tissues was closely related to shorter survival and lymphatic invasion (7). As an intracellular protein, annexin A1 has been found to be coupled with calcium and phospholipids, and to exert a series of crucial biological functions in the survival and growth of cells, such as differentiation, control of apoptosis, and carcinogenesis (8). Moreover, overexpression of annexin A1 in several types of cancer including hepatocellular carcinoma, pancreatic cancer, lung cancer and breast cancer (1, 7, 8) has been found to be a significant marker for those tumors and to correlate with their development and progression. However, the clinical significance of BALF annexin A1 levels in lung cancer patients remains unclear. The purpose of this investigation was to quantify annexin A1 levels in BALF and explore their usefulness in lung cancer diagnosis.
Material and methods
Study participants
From January 2013 to October 2015, we enrolled 127 individuals with pulmonary masses treated at Gansu Provincial People's Hospital, Lanzhou, China and the Affiliated Hospital of Jining Medical University, Jining, China. The study was approved by the institutional review boards of both hospitals, and written informed consent was obtained from each patient before bronchoscopy. All diagnoses were confirmed by histopathological examination and patients were excluded if they had previously received chemotherapy or radiotherapy, or suffered from severe multiple organ dysfunction. All patients underwent lung biopsy and BALF was collected for laboratory tests. Eighty-six patients were diagnosed with lung cancer and were classified according to the TNM staging system (9). Forty-one of the 127 individuals were diagnosed with benign lung disease such as chronic obstructive pulmonary disease, chronic infectious diseases, tuberculosis, and diffuse noninfectious interstitial diseases. We collected blood samples from all 127 patients as an experimental control to BALF.
Immunohistochemistry (IHC)
We applied IHC to measure the expression levels of annexin A1 in tissues with lung cancer and benign lung disease (polyclonal annexin A1 antibody, 1:100 dilution; Boster Biotech Company). We used breast cancer slices with a positive reaction to annexin A1 as positive control, and annexin A1 antibody was replaced with PBS as negative control. The IHC reaction for annexin A1 and immunostaining measurement were performed in accordance with previously described processes and means (7).
Bronchoalveolar lavage
BAL was performed according to a previously described method (6). The bronchoscope operators used 5-10 mL of 2% lidocaine to perform local anesthesia of the airways to be lavaged. They then washed the airways with 0.9% saline solution and used a siliconized container to collect BALF. The fluid was filtered through a nylon filter and centrifuged at 3,000 rpm at 4°C for 30 minutes to isolate the supernatant aliquots. The BALF samples were numbered under double-blind conditions and the supernatant aliquots were immediately placed into an ultra–low-temperature freezer and stored at -80°C (6).
Enzyme-linked immunosorbent assay (ELISA)
All blood specimens were numbered and stored under double-blind conditions. Specimens displaying hemolysis were considered invalid and were removed from the investigation. The concentration of annexin A1 was quantified by an ELISA kit that was produced according to a previously reported method (10) and the experimental process complied with the manufacturer's instructions (code CK-E91728H, Xitang Biotech Company) (10).
Statistical analysis
The expression levels of annexin A1 protein in lung cancer and benign lung tissues were calculated using the chi-square test and Fisher's exact test. Annexin A1 levels in serum and BALF represented continuous variables and were assessed by Student's t-test, the Kruskal-Wallis test and 1-way ANOVA. We applied ROC curve analysis to identify cutoff values of annexin A1 in serum and BALF for discriminating lung cancer from benign lung diseases. We also employed ROC analysis to establish the sensitivity and specificity of annexin A1 in BALF and serum. The statistical software used in this study was SPSS 21.0 (IBM). All tests were 2-sided, and we considered there to be statistical significance when the p value was less than 0.05.
Results
Expression of annexin A1 in lung cancer tissues and tissues with benign lung disease
Some biopsy specimens of the 127 study participants were insufficient for IHC. Fifty-eight lung cancer tissue specimens and 28 specimens of non-lung-cancer tissues qualified for further IHC. The results showed that the expression of annexin A1 in lung cancer tissues was higher (29/58, 50%) than that in normal lung tissues (5/28, 17.9%) (p = 0.002) (Tab. I, Fig. 1).
Relationship between clinical parameters and the expression of annexin A1 in lung cancer tissues (N = 58)
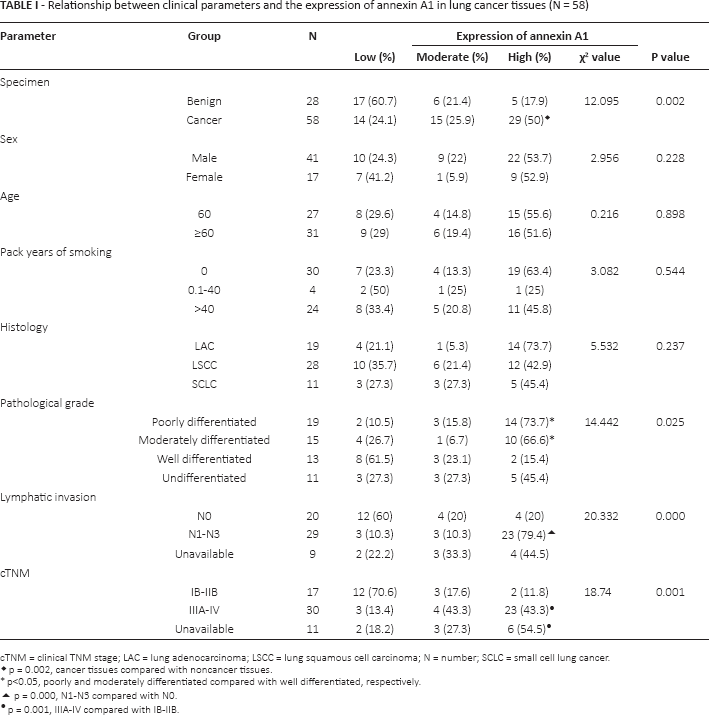
cTNM = clinical TNM stage; LAC = lung adenocarcinoma; LSCC = lung squamous cell carcinoma; N = number; SCLC = small cell lung cancer.
p = 0.002, cancer tissues compared with noncancer tissues.
p<0.05, poorly and moderately differentiated compared with well differentiated, respectively.
p = 0.000, N1-N3 compared with N0.
p = 0.001, IIIA-IV compared with IB-IIB.
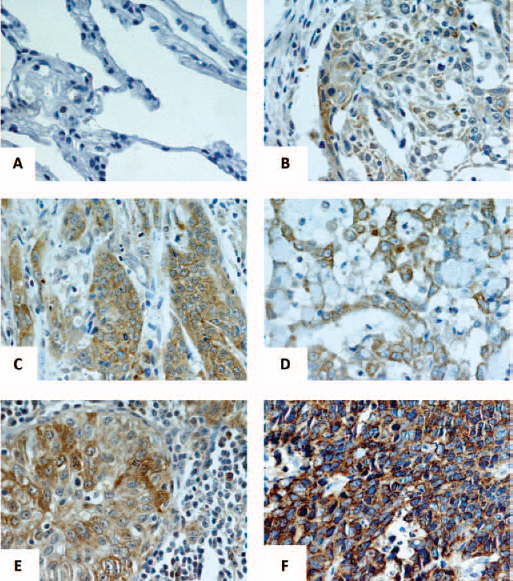
Immunohistochemical analysis of annexin A1 in lung cancer and benign lung disease (IHC ×400). (A) Weak staining of annexin A1 in normal tissue; (B) weak staining of annexin A1 in well-differentiated LSCC; (C) strong staining of annexin A1 in poorly differentiated LSCC; (D) moderate staining of annexin A1 in moderately differentiated LAC; (E) strong staining of annexin A1 in poorly differentiated LAC; (F) strong staining of annexin A1 in SCLC. LAC = lung adenocarcinoma; LSCC = lung squamous cell carcinoma; SCLC = small cell lung cancer.
Correlation of annexin A1 in lung cancer tissues with lymphatic invasion and advanced disease stage
As shown in Table I, poorly and moderately differentiated lung cancer tissues displayed higher annexin A1 expression (73.7% and 66.6%, respectively) than well-differentiated lung cancer tissues (15.4%) (p = 0.025). Lung cancer tissues of patients with lymph node metastasis showed higher annexin A1 expression (79.4%) than those of patients without lymph node metastasis (20%) (p<0.001). Moreover, annexin A1 expression was higher in tissues with stage III-IV lung cancer (43.4%) than in those with stage I-II disease (11.8%) (p = 0.001). The expression of annexin A1 was unrelated to gender, age, smoking and tumor histology (p>0.05).
The levels of annexin A1 in BALF and serum of lung cancer patients and patients with benign lung disease
Annexin A1 levels were measured by ELISA in BALF and serum specimens of 86 lung cancer patients and 41 patients with benign lung disease. The results showed that BALF and serum annexin A1 levels were higher in lung cancer patients than in patients with benign diseases (BALF: 6.75 ± 1.72 ng/mL vs. 1.91 ± 1.02 ng/mL; serum: 5.35 ± 1.27 ng/mL vs. 1.52 ± 1.06 ng/mL) (p<0.0005) (Tab. II).
Correlation between BALF and serum levels of annexin A1 and clinicopathological factors in lung cancer patients
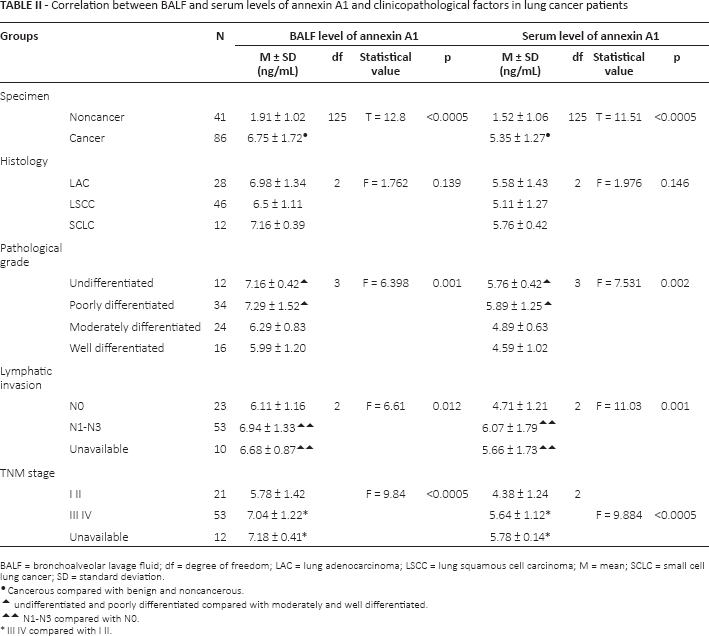
BALF = bronchoalveolar lavage fluid; df = degree of freedom; LAC = lung adenocarcinoma; LSCC = lung squamous cell carcinoma; M = mean; SCLC = small cell lung cancer; SD = standard deviation.
Cancerous compared with benign and noncancerous.
undifferentiated and poorly differentiated compared with moderately and well differentiated.
N1-N3 compared with N0.
III IV compared with I II.
Correlation of annexin A1 in BALF and serum with clinicopathological factors
As shown in Table II, patients with poorly differentiated lung cancer had higher levels of annexin A1 than patients with well-differentiated lung cancer (7.29 ± 1.52 vs. 5.99 ± 1.20 ng/mL in BALF; 5.89 ± 1.25 vs. 4.59 ± 1.02 ng/mL in serum) (p<0.05). Likewise, patients with lymphatic invasion (N1-N3) had higher annexin A1 levels than those without lymphatic invasion (6.94 ± 1.33 vs. 6.11 ± 1.16 ng/mL in BALF; 6.07 ± 1.79 vs. 4.71 ± 1.21 ng/mL in serum) (p<0.05). In addition, lung cancer patients with stage III-IV disease had higher annexin A1 levels than those with stage I-II (7.04 ± 1.22 vs. 5.78 ± 1.42 ng/mL in BALF; 5.64 ± 1.12 vs. 4.38 ± 1.24 ng/mL in serum) (p<0.0005).
Relationship between annexin A1 overexpression in tissue, BALF and serum
We divided the annexin A1 values in BALF and serum into 3 zones and compared them with the 3 levels of tissue expression using nonparametric rank correlation analysis. Spearman's rank correlation coefficient was 0.873 (p = 0.019), indicating a consistent trend between the values of annexin A1 in BALF, serum and tissues.
Determination of cutoff values for annexin A1 in BALF and serum
By observing the changes in the data, we noticed that the lowest value of annexin A1 was in serum of the benign lung disease group (0.19 ng/mL) and the highest value was in BALF of the lung cancer group (16.1 ng/mL). Moreover, the BALF values in patients with lung cancer and benign lung disease nearly intersected at 2-4 ng/mL (Tab. III, Fig. 2A and 2B) and the serum values at 3-4 ng/mL (Tab. III, Fig. 3A and 3B). We deduced that the thresholds of annexin A1 in BALF and serum might be in those areas; the 95% confidence intervals (CIs) were 0.0510 to 0.422 (BALF) and 0.288 to 19.752 (serum).
NLR and PLR of annexin A1 level in BALF and serum groups
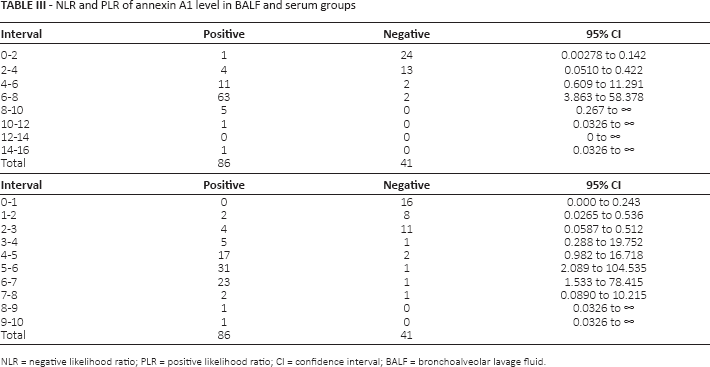
NLR = negative likelihood ratio; PLR = positive likelihood ratio; CI = confidence interval; BALF = bronchoalveolar lavage fluid.
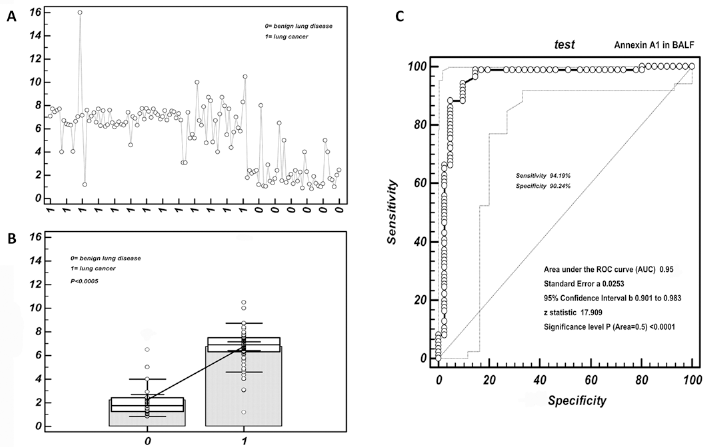
Cutoff value selection of annexin A1 in BALF by ROC curve analysis. (A) Distribution of BALF annexin A1 values in lung cancer patients and patients with benign lung disease. The ordinate represents the expression level of annexin A1 in BALF; the abscissa represents the patient group. (B) Values of BALF annexin A1 were higher in lung cancer than benign lung disease. The ordinate represents the expression level of annexin A1 in BALF; the abscissa represents the patient group. The circles represent the dispersion of the detection value. The steep straight line between the 2 box plots indicates a nearly 3-fold difference between the median values of the 2 patient groups. (C) ROC curves for BALF annexin A1.
Accuracy of annexin A1 in BALF for predicting lung cancer by ROC analysis
As shown in Table IV, the cutoff value of annexin A1 in BALF obtained by ROC analysis was 4 ng/mL. With this threshold, the sensitivity and specificity of annexin A1 in BALF were 94.2% and 90.2%, respectively. The calculated serum threshold was 3.1 ng/mL, with a sensitivity and specificity of 93.02% and 87.80%, respectively. To discern lung cancer, the area under the curve (AUC) of BALF annexin A1 was 0.95, 95% CI 0.901-0.983, Z = 17.909 (p<0.0001); for the serum annexin A1 concentration the AUC was 0.923, 95% CI 0.862-0.963, Z = 12.813 (p<0.0001) (Fig. 2C and 3C).
Cutoff values of annexin A1 in BALF and serum specimens
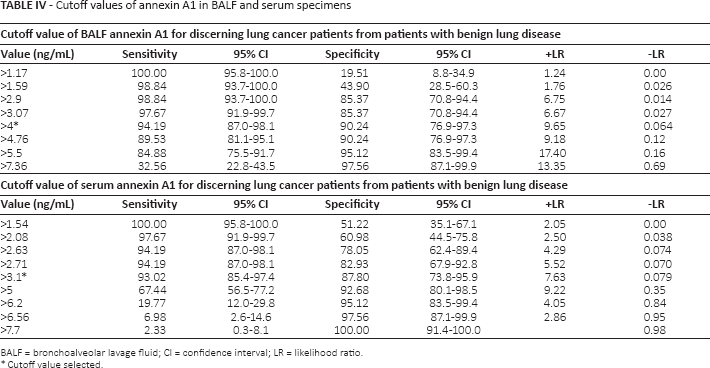
BALF = bronchoalveolar lavage fluid; CI = confidence interval; LR = likelihood ratio.
Cutoff value selected.
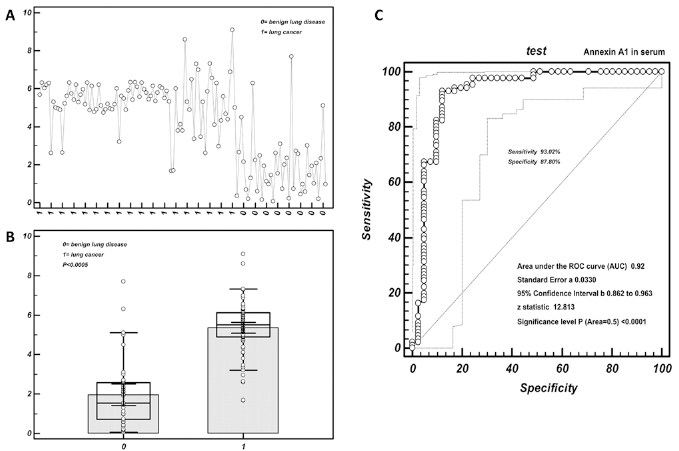
Cutoff value selection of annexin A1 in serum by ROC curve analysis. (A) Distribution of serum annexin A1 values in lung cancer patients and patients with benign lung disease. The ordinate represents the expression level of annexin A1 in serum; the abscissa represents the patient group. (B) Values of serum annexin A1 were higher in lung cancer than benign lung disease. The ordinate represents the expression level of annexin A1 in serum; the abscissa represents the patient group. The circles represent the dispersion of the detection value. (C) ROC curves for serum annexin A1.
Comparison of diagnostic accuracy between BALF and serum annexin A1 levels by ROC analysis
Through observation of the data, we found a strong correlation between BALF annexin A1 and serum annexin A1: the correlation coefficient of the regression model was 0.65 (95% CI 0.533-0.738) (p<0.0001), indicating a linear correlation between the 2 specimen types (p<0.0001; Fig. 4A). The Youden index suggested that BALF annexin A1 had good diagnostic value (Fig. 4B). The combined sensitivity and specificity of BALF and serum annexin A1 in lung cancer detection were 93.5% and 90.2%, respectively, which were higher than the sensitivity and specificity of BALF and serum alone. The difference between the AUCs of annexin A1 in BALF and serum was 0.031, and the Z statistic was 1.376 (p = 0.1681) (Fig. 4C).
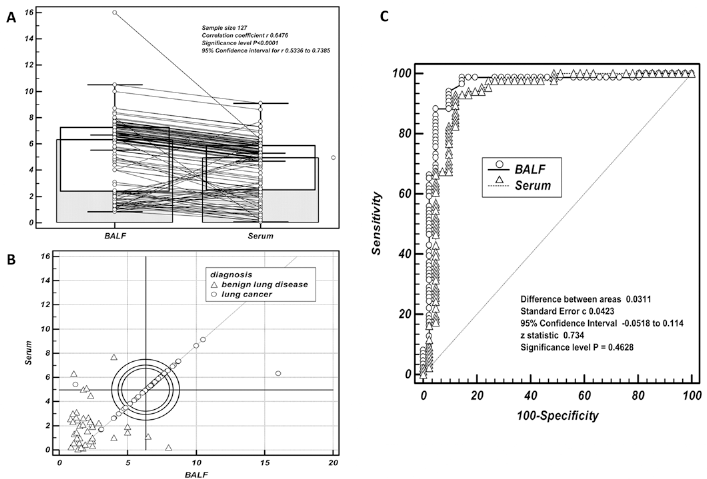
Comparison of diagnostic accuracy between BALF annexin A1 and serum annexin A1 by ROC curve analysis. (A) The correlation coefficient of the regression model of BALF annexin A1 and serum annexin A1 was 0.65 (95% CI 0.533-0.738). There was a linear correlation between the 2 sample types. (B) The Youden index indicated that the values of BALF annexin A1 and serum annexin A1 were more close to 1, and there was good diagnostic value for BALF annexin A1. (C) With benign disease as control, the specificity of BALF annexin A1 surpassed that of serum, but its sensitivity was similar to the sensitivity of serum annexin A1.
Discussion
The Chinese are the largest population in Asia and lung cancer is one of the diseases with the highest incidence rates in this population (11). Electronic bronchoscopy has been widely applied in its diagnosis and treatment, and BAL is regarded as an alternative resource for detecting lung cancer biomarkers (12). Some researchers believe that BALF could be a medium for abnormal proteins from airways where neoplastic changes take place (13, 14). We found in a previous study that elevated annexin A1 in tissues and serum of lung cancer patients correlated with the malignant behavior and prognosis of the disease (7, 10). Annexin A1, a small molecule with a molecular weight of 37 kDa, is involved in a series of biological processes including inflammation, regulation of cell proliferation, signaling of cell apoptosis, and development and progression of malignant tumors (8). Upregulation of its expression has been observed in lung, breast, pancreas, cervix, bladder, renal and pituitary cancer. It was also significantly associated with advanced stages of disease, especially those with an unfavorable prognosis (8). However, the study of the expression level of annexin A1 in BALF and its significance in the development of lung cancer is considerably limited. Our purpose was to clarify the significance of annexin A1 in BALF and its usefulness for lung cancer diagnosis and screening.
At the tissue level we found that annexin A1 protein was upregulated in various histological types of lung cancer compared with benign lung disease, especially in cancer tissues with poor differentiation and advanced stage, which was concordant with the results of a previous investigation (7). This led us to consider that the “ebb and flow” of annexin A1 potentially effects the development and progression of lung cancer. Annexin A1 overexpression has been linked to advanced stages of specific cancers, as well as to differentiation grade and the tendency to metastasize. Furthermore, it has been proposed as an effective differential marker for the detection of these cancers (8). For example, carcinoembryonic antigen, neuron-specific enolase and CYFRA21-1 have been widely used as tumor markers in predicting and discerning malignant lung disease from benign disease (15–19). In the present study, the annexin A1 level in BALF of lung cancer patients was significantly elevated compared with the level in patients with benign lung disease, suggesting that an increase in annexin A1 in BALF correlates with the progression of lung cancer and that it might be a risk factor for lung cancer. Through the consistency test (McNemar's test) we noticed a consistent trend of the annexin A1 level between different sample types (lung cancer tissue, BALF and serum). More importantly, an elevated level of annexin A1 in BALF was associated with more malignant biological characteristics represented by lymphatic metastasis, poor differentiation and high TNM stage. This information suggested that elevated annexin A1 in BALF could be a new diagnostic and predictive tool for lung cancer. In the future, we should further explore its mechanism and accuracy as well as the intrinsic correlation between higher annexin A1 in BALF and the process of lung cancer.
For biomarkers, optimum sensitivity and specificity are crucial. When the diagnosis of a disease is suspected, we need a marker with high specificity to help discern it. Meanwhile, its sensitivity should be such as to allow us to detect it at an early time. This is why we selected the 95th percentile, which was obtained from the cutoff value. In the present study we evaluated the diagnostic ability of annexin A1 in discerning patients with lung cancer from patients with benign lung disease using ROC curve analysis. With a cutoff value of 4 ng/mL, BALF annexin A1 had a sensitivity of 94.2% and a specificity of 90.2%, while the sensitivity and specificity of serum annexin A1 were slightly lower. These results strengthened previous evidence that elevated annexin A1 closely correlated with the presence of lung cancer and suggested that the detection of annexin A1 in BALF of patients with pulmonary masses could be a useful diagnostic approach.
The differential diagnosis of a pulmonary mass is a crucial and challenging process in the clinical workup. Early diagnosis of lung cancer will greatly improve the prognosis of the patient. The results of our research support the usefulness of BALF annexin A1 for the differential diagnosis of lung cancer. Nevertheless, the study has some objective limitations. First, it included only 2 centers. Second, the samples were relatively small, and did not provide adequate evidence to assess early stages of lung cancer (i.e., the difference between stage I and II). In the future we plan to carry out a prospective and large cohort study in multiple centers to clarify these questions.
Conclusion
Increased annexin A1 in BALF correlates with poor differentiation, lymphatic invasion and advanced stage of lung cancer, and there is a concordant trend with serum annexin A1. BALF annexin A1 has a clinically relevant impact on the sensitivity and specificity of lung cancer detection, which indicates that the annexin A1 level in BALF could be considered as a new diagnostic tool for lung cancer.
Footnotes
Financial support: This study was supported by the Technology Research and Development Plan of Gansu Province (0912TCYA016), China.
Conflict of interest: None.
