Abstract
Purpose
The influence of the androgen receptor (AR) CAG repeat polymorphism on breast cancer is controversial. We investigated the combined effects of CAG repeat length and estrogen receptor (ER) status on prognosis in 355 postmenopausal women with primary breast cancer.
Methods
CAG repeat length was determined by the HUMARA test. Relapse-free survival (RFS) and overall survival (OS) according to the X-weighted CAG repeat biallelic mean (XWBM) were investigated by univariate and multivariate analysis.
Results
XWBM was not associated with RFS or OS, but a significant interaction between XWBM and ER status (p = 0.002) was found for OS. ER-negative patients with median XWBM <20 showed lower OS than ER-negative/XWBM ≥20 patients (HR = 0.270; 95% Cl: 0.073-0.999). ER-negative/XWBM <20 patients also had significantly lower OS than ER-positive women, irrespective of CAG repeat length (p<0.001). Accordingly, estimated OS was lowest in ER-negative patients with XWBM <20 (OS: 0.63, 95% CI: 0.41-0.79) and highest in ER-positive patients with XWBM <20 (OS: 0.95, 95% CI: 0.90-0.97).
Conclusions
Our data suggest that short CAG repeat length is associated with increased risk of death in ER-negative disease but is related to better survival when ER is expressed. These findings are in agreement with the hypothesis that AR may stimulate or inhibit breast cancer growth depending on ER status, AR transactivation, and the endocrine-metabolic environment of breast tumors. Evaluation of CAG repeat length together with ER status could help improve the estimation of the risk of death, with possible implications for the optimization of standard breast cancer treatment and implementation of prevention strategies.
Introduction
The relationship between androgens and the development and progression of breast cancer is complex. Epidemiological and clinical evidence (1, 2) shows that high circulating androgen levels are associated with an increased risk of developing breast cancer, the main proposed mechanism being that androgens stimulate breast cancer growth indirectly via aromatization to estrogens or directly via androgen receptors (AR) (3). Many laboratory studies, on the other hand, have shown that androgens have an inhibitory effect on mammary epithelial cells, possibly by competitive blockade of estrogen receptor (ER) response elements, which reduce estrogen-induced stimulation (4). This hypothesis is consistent with the finding that AR expression is associated with a favorable prognosis, particularly for cancers that are also ER positive (5).
AR contains in the N-terminal transactivation domain a polyglutamine tract of variable length encoded by a highly polymorphic CAG repeat microsatellite situated in exon 1 of the AR gene, which is located on the X chromosome at Xq12-13 (6). CAG repeat length can modulate the ability of the receptor to enhance transcriptional events in vitro (7, 8). In particular, it was found that longer CAG repeat length is associated with decreased transcriptional activity of the gene (7, 8). Several studies have investigated CAG repeat length in relation to breast cancer risk, with contrasting or null results (9). A recent meta-analysis suggested that long CAG repeat length might protect against breast cancer onset (10), but the data remain inconclusive, partly because only few studies took into account X-chromosome inactivation (XCI) occurring in women (11, 12). To our knowledge, only Yu et al (13) have investigated CAG repeat length in relation to breast cancer prognosis, finding an inverse association between CAG repeat length and risk of relapse and death.
In this study we aimed to preliminarily investigate the prognostic value of CAG repeat length in postmenopausal women with breast cancer by taking advantage of the TPM (Testosterone Prognosi della Mammella) breast cancer cohort prospectively collected at our institute (14-16).
Methods
Patient cohort
The TPM cohort consisted of 2,030 women (1,340 postmenopausal and 690 premenopausal) with primary invasive breast cancer, recruited from December 2003 to March 2011. All women underwent surgery at the Breast Surgery Unit of the National Cancer Institute of Milan (INT). The inclusion criterion was histologically confirmed non-metastatic breast carcinoma (any T, any N, M0); exclusion criteria were non-epithelial cancer, previous cancer diagnosis (except in situ cervical cancer or non-melanoma skin cancer), and neoadjuvant chemotherapy or hormone therapy. Written informed consent was obtained from all included patients. The study was approved by the Scientific and Ethical Committee of the INT.
At time of diagnosis before any treatment the women provided a fasting blood sample. Samples were processed, divided into aliquots of serum, plasma, buffy coat and red blood cells, and stored at −80°C to form a biological bank. The clinical and pathological characteristics of patients were extracted from the clinical records and entered into a dedicated database. Patient follow-up was performed by consulting the medical records. Information about the vital status of patients who had discontinued their regular checkups at our institution was obtained by telephone interview of patients or their next of kin. Interviews were carried out by expert trained personnel in accordance with a predefined protocol.
This study focused on the first 592 postmenopausal patients, recruited from December 2003 to December 2006, for whom follow-up was completed and ended by December 2012.
Of the 592 initial patients recruited, 20 were excluded because they were not considered postmenopausal: in 9 patients hormone replacement therapy had been stopped only 3 months before recruitment; in 2 information on last menstruation was lacking (1 woman aged <50 years and 1 woman with estradiol >30 pg/mL); 2 had their last menstruation less than 1 year ago; a further 7 had been hysterectomized and estradiol was >30 pg/mL (6 aged <50 years and 1 aged 52 years). A further 217 women were excluded because no buffy coat sample was available. Thus the final number of patients investigated in this analysis was 355. No differences in clinicopathological characteristics were observed between patients with and without CAG determination (data not shown).
The women were followed from study entry to any breast cancer recurrence event, other non-breast primary cancers, death or end of follow-up, whichever occurred first. During follow-up, 50 patients had breast cancer recurrence events and 33 died.
Analysis of CAG repeat length
The analysis of the CAG repeat polymorphism was conducted on DNA extracted from buffy coat according to the standard protocols (QIAamp DNA blood mini kit, Qiagen, Chatsworth, CA, USA). The concentration of extracted DNA was quantified by UV spectrophotometry.
CAG repeat length and XCI were determined by means of the HUMARA assay (17) with XCI ratios in heterozygotes calculated as reported by Manzardo et al (18). The HUMARA test consisted of 2 reactions for every DNA sample: in the first, 250 ng of DNA was digested with 5 U of both HpaII and HhaI restriction enzymes (Fermentas, Vilnius, Lithuania) in a 20-µL reaction volume; in the second, DNA was incubated only with the digestion buffer (Tango buffer, Fermentas) without the 2 enzymes. The digestion process degrades activated (unmethylated) but not inactivated (methylated) DNA. After overnight digestion at 37°C, the sequences of interest of both DNA samples were amplified by PCR reaction.
The following primers were used (17): reverse: 5’-TCCAGAATCTGTTCCAGAGCGTGC-3’; forward: 5’-GCTGTGAAGGTTGCTGTTCTTCAT-3’ (Thermo Fisher Scientific, Ulm, Germany); the forward primer was 5’-labeled with the chromophore 6-FAM. Each PCR amplification was performed in a 25-µL volume containing 200 µM dNTPs, 1.5 mM MgCl2, 1X Buffer Gold, 0.5 µM of each PCR primer and 1.25 U of Ampli Taq Gold DNA polymerase (Applied Biosystems, Bleiswijk, The Netherlands). Reactions were performed using a 2720 GeneAmp thermal cycler (Applied Biosystems) under the following conditions: an initial denaturation step at 95°C for 5 minutes, 40 cycles (each comprising 30 seconds at 95°C, 30 seconds at 65°C and 30 seconds at 72°C), and a final extension at 72°C for 30 minutes. The PCR products were checked on 2% agarose gel. Lastly, 1 µL of PCR product was analyzed by capillary electrophoresis with a genetic automated analyzer (ABI PRISM 3130, Applied Biosystems) that allows the determination of the number of CAG repeats (genotyping).
The percentage of XCI for each AR allele was then calculated using the following formula: (d1*u1)/[(d1*u1) + (d2*u2)], where d1 is the peak area of digested DNA from the first allele and u1 the peak area of undigested DNA from the first allele; d2 is the peak area of digested DNA from the second allele and u2 the peak area of undigested DNA from the second allele (18).
Statistical Analysis
Statistical analysis was performed using as pivotal variable the X-weighted biallelic mean (XWBM), which was determined by combining the number of CAG repeats with the percentage of allele-specific activation as described by Hickey et al (12). In homozygous individuals XWBM is not affected by variation of allele-specific activation and is therefore equal to the mean CAG repeat length (biallelic mean).
Associations of XWBM with clinicopathological characteristics were investigated by the Kruskal-Wallis test. Time from surgery to first relapse and time to death from any cause were used in the estimation of relapse-free survival (RFS) and overall survival (OS), respectively, by the Kaplan-Meier method. The prognostic role of each clinicopathological variable in RFS and OS was investigated by Cox regression modeling with age and XWBM as continuous variables. The relationship between XWBM and outcomes was investigated using a regression model based on restricted cubic splines. We also investigated the interaction between XWBM and ER status using a Cox model that included the main effects and first-order interaction terms for OS and RFS. Finally, the prognostic role of XWBM was evaluated in multivariate fashion by resorting to a Cox model including all of the variables that were statistically significant (α = 5%) in univariate analysis. A more parsimonious final model was then obtained using a backward selection procedure that retained only the variables reaching the conventional level of significance of 5%. The analyses were performed with SAS (version 9.2; SAS Institute Inc., Cary, NC, USA), adopting a significance level of alpha = 0.05.
Results
The patient and tumor characteristics are summarized in Table I. No statistically significant associations were found between XWBM and the considered clinicopathological characteristics (data not shown).
Patient characteristics
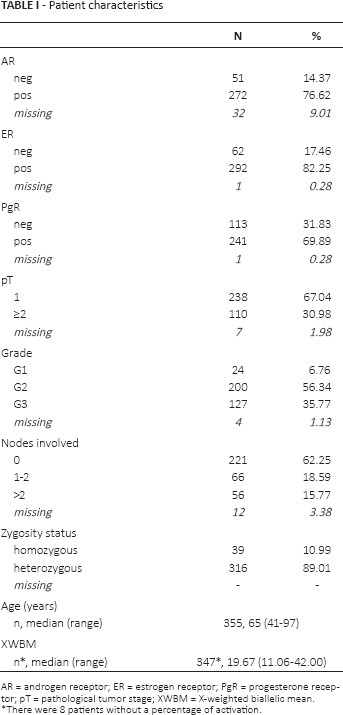
AR = androgen receptor; ER = estrogen receptor; PgR = progesterone receptor; pT = pathological tumor stage; XWBM = X-weighted biallelic mean.
There were 8 patients without a percentage of activation.
At a median follow-up of 70 months (range 0-102), the probability of OS was 0.90 (95% CI 0.86-0.93) and the probability of RFS 0.86 (95% CI 0.82-0.90). In univariate analysis, XWBM was not associated with OS or RFS. ER status, tumor size, tumor grade, number of involved nodes and age were significantly associated with OS. The same variables except age showed significant prognostic value for RFS (Tab. II).
Univariate Cox analysis – Overall and relapse-free survival
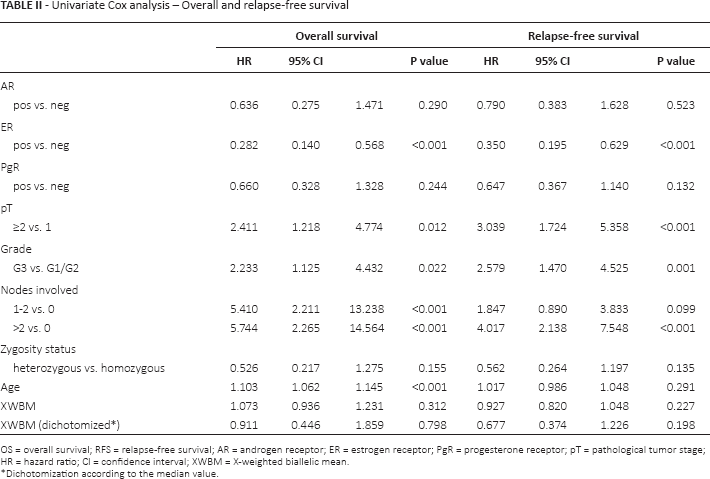
OS = overall survival; RFS = relapse-free survival; AR = androgen receptor; ER = estrogen receptor; PgR = progesterone receptor; pT = pathological tumor stage; HR = hazard ratio; CI = confidence interval; XWBM = X-weighted biallelic mean.
Dichotomization according to the median value.
The first-order interaction term between XWBM and ER status was statistically significant only for OS (Tab. III). Taking XWBM as a continuous variable, we observed a decreased risk of death for longer XWBM values in ER-negative patients and the opposite in ER-positive patients (data not shown). To further investigate the interaction and make the interpretation of the results easier, we dichotomized XWBM according to its median value and we considered the joint variable “XWBM/ER status” obtained by combining the modalities XWBM and ER status in the following categories: XWBM<20/ER-negative, XWBM≥20/ER-negative, XWBM<20/ER-positive, and XWBM≥20/ER-positive.
Bivariate Cox regression analysis with first-order interaction term (OS) – XWBM continuous
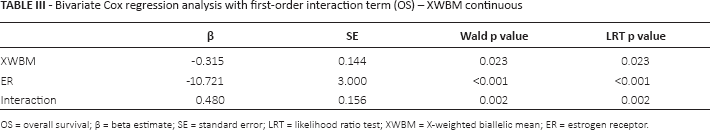
OS = overall survival; β = beta estimate; SE = standard error; LRT = likelihood ratio test; XWBM = X-weighted biallelic mean; ER = estrogen receptor.
Univariate Cox analysis, taking XWBM<20/ER-negative as the reference category, showed that ER-negative patients with XWBM<20 had poorer OS than XWBM≥20/ER-negative patients, with borderline statistical significance (p = 0.05). Conversely, ER-negative patients with XWBM<20 had statistically significantly poorer OS with respect to ER-positive patients irrespective of their XWBM status (Fig. 1). Accordingly, the estimated survival in ER-negative/XWBM<20 patients was lowest (OS: 0.63, 95% CI 0.41-0.79), while ER-positive patients with XWBM<20 had the best result in terms of survival (OS: 0.95, 95% CI 0.90-0.97) (Fig. 1). These results indicate that short CAG repeat length is associated with poorer OS when ER status is negative but with better survival when ER status is positive. This finding was confirmed in the multivariate analysis, which we performed, given the small number of events per variable, for exploratory purposes only (Tab. IV).
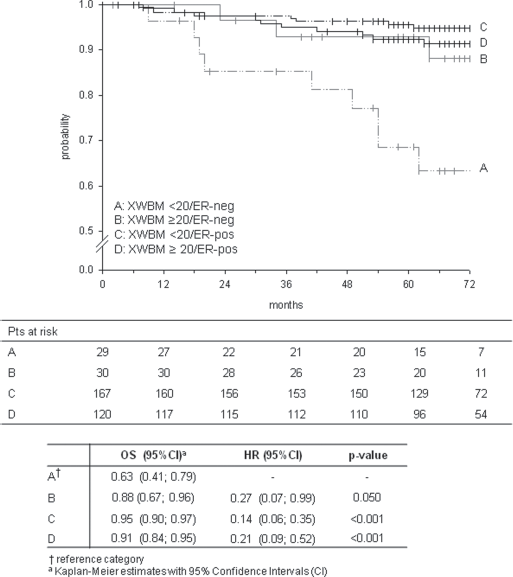
Five-year probability of overall survival in relation to the joint variable XWBM/ER status, together with the univariate Cox results.
Multivariate Cox regression analysis (OS): final model - XWBM/ER
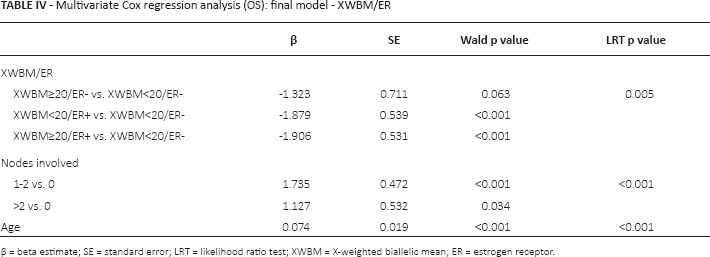
β = beta estimate; SE = standard error; LRT = likelihood ratio test; XWBM = X-weighted biallelic mean; ER = estrogen receptor.
Discussion
This study investigated the role of CAG repeat length in breast cancer prognosis according to ER status. The AR gene is located on chromosome X and is subject to inactivation, so for a better simultaneous estimate of both length and specific activation we calculated the XCI ratios and the XWBM.
No association was observed between the patients’ clinicopathological characteristics and CAG repeat length, which is in agreement with the findings of other authors (19, 20). Similarly, we did not see a significant association between the number of CAG repeats (on a continuous or dichotomized scale) and patient prognosis.
However, we did observe a significant interaction between XWBM and expression of ER in the OS analysis, indicative of a different, opposite influence of CAG repeat length on death risk in the 2 patient subgroups. A decreased risk of death was related to long CAG repeat sequences in the ER-negative subgroup (OS probability: 0.63 [95% CI 0.41-0.79] and 0.88 [95% CI 0.67-0.96] with XWBM<20 and XWBM≥20, respectively), but in ER-positive patients we observed the opposite trend, with short CAG repeat length being associated with a better prognosis (OS probability 0.95 [95% CI 0.90-0.97] and 0.91 [95% CI 0.84-0.95] for XWBM<20 and XWBM≥20, respectively).
To our knowledge this is the first study assessing the combined influence of CAG repeat length and ER status on breast cancer prognosis. Several studies have investigated the influence of AR length polymorphism on the onset of breast cancer. These studies were recently summarized in a meta-analysis by Hao et al (10), which showed that long CAG repeat length could have a protective effect, i.e., reduce the risk of breast cancer in a woman's lifetime. Yu et al (13) found that long CAG repeats were also associated with a better breast cancer prognosis. These data are broadly consistent with our findings, although differences in study design, such as the fact that Yu et al did not examine XCI and did not stratify for ER status, make it difficult to compare the results.
Our data, suggesting a different effect of CAG repeat length on breast cancer prognosis as a function of ER status, are consistent with the emerging hypothesis that androgens and the AR pathway act in a bidirectional manner on breast cancer growth, i.e., proliferative or antiproliferative depending on the balance of steroidogenic enzymes (aromatase/5α-reductase), sex hormones (androgens/estrogens), steroid receptors (ER/AR), and more generally on the endocrine-metabolic environment of breast cancer cells (for reviews, see ref. 21, 22). We speculate that, in ER-positive cancer, an AR with shorter CAG repeat length (which is an index of higher transactivational activity) more effectively represses estrogen-induced transcriptional events, thereby exerting a protective effect. Conversely, in ER-negative tumors, increased AR transcriptional activity due to shorter CAG repeat length may stimulate breast cancer proliferation via direct AR-mediated mechanisms. Experimental studies showed that, in the absence of ER expression, AR is able to bind and regulate ER-cis-regulatory elements, mimicking ER proliferative transcriptional events (4, 23). In addition, AR can promote the proliferation of ER-negative tumors by activating growth factor pathways (4, 24).
Although our data are consistent with the new evidence on the 2-way action of ARs, it is necessary to take into account some limitations of the study. X chromosomes are randomly inactivated during early embryogenesis, resulting in an inactivation pattern that varies among tissues. Thus the XCI pattern we observed in blood DNA might not completely overlap the XCI pattern in breast cancer. However, several studies showed that the correlation of XCI between tissues ranges from 0.5 to 0.8 in the same individual (25). Furthermore, Kristiansen et al (26) described a comparable pattern of XCI in breast cancer tissue and peripheral blood cells of young breast cancer patients (26). These observations provide a rationale for XCI analysis in alternative tissues when the appropriate tissue is not available or when large cohorts are studied. An additional limitation emerged from the observation that repeat length polymorphisms may be unstable in cancer, and also XCI is a reversible epigenetic process that can be altered in cancer, often producing considerable intratumoral genetic heterogeneity. As a result, the breast cancer prognosis should be influenced by the genotype of cancer cells (somatic) more than by the genotype of the individual (constitutional) (27).
To conclude, our finding that the prognostic role of CAG repeat length in breast cancer seems to depend on ER status sheds some light on the complex and still unclarified mechanisms of AR activity in tumor growth. If our data are confirmed, the evaluation of CAG repeat length together with ER status could help improve the estimation of individual patient risk of death, with possible implications for the optimization of standard breast cancer treatment and implementation of prevention strategies.
Footnotes
Abbreviations
Acknowledgment
We thank Dr. Giorgio Secreto, who inspired the present study, Dr. Franco Berrino for useful suggestions, Dr. Giuseppina Saragò and Dr. Matteo Galli for their contribution to the research, Ms. Patrizia Mondini for technical assistance, and the patients of the TPM cohort for their cooperation.
Financial support: This work was supported by the Italian Association for Cancer Research (ref. 47/03), the Italian Ministry of Health (RF-INT-2005-157276), and funds obtained through an Italian law that allows taxpayers to allocate 0.5 percent of their income tax payment to a research institution of their choice.
Conflict of interest: The authors declare that they have no conflict of interest.
