Abstract
Purpose
The use of trastuzumab (Herceptin) to target HER2 has been applied in breast carcinoma and gastric carcinoma (GC). Previous studies have tested trastuzumab's effectiveness by assessing HER2 expression or HER2 amplification by means of immunohistochemistry (IHC) or fluorescence in situ hybridization (FISH). In this work we aimed to evaluate automated FISH and IHC technologies for HER2 detection in GC biopsies to be used in routine pathology practice.
Methods
The study used an Oracle HER2 IHC System and an LSI HER2/CEP17 Dual Probe on an automated Bond™ system (Leica Microsystems). One hundred GC biopsies were evaluated including 44 intestinal type, 38 diffuse type and 18 indeterminate type according to Lauren's classification.
Results
The overall concordance rate between the automated FISH and IHC methods was 94% (κ = 0.87), as 6 samples were scored as equivocal (4 in IHC and 2 in FISH). Moreover, HER2 positivity was significantly different between the 3 types of GC (p<0.05), being more frequent in intestinal-type GC (23%) than in the other 2 histological types (5% and 0%). Finally, the HER2/CEP17 FISH ratio was significantly different (p<0.01) between disomic and polysomic samples, being higher in polysomic samples (mean 1.633 ± 0.509) than in disomic samples (mean 1.231 ± 0.675).
Conclusions
Automated HER2 testing of GC biopsies using the Leica Bond system was useful and efficient. This method allowed us to improve normal routine procedures, minimizing time and costs as well as handling and observation errors.
Introduction
Gastric cancer (GC) is one of the most common malignancies worldwide (1-2-3). Although in the last few decades its mortality has decreased considerably, it remains the second leading cause of cancer-related death on a global level (3, 4). GC is also associated with poor prognosis and late diagnosis mainly because symptoms are uncommon until the later stages of the disease (4). As other types of carcinomas, GC carcinogenesis seems to be a multistep process involving numerous genetic and epigenetic alterations, including gene amplification (4, 5).
Overexpression of human epidermal growth factor receptor 2 (HER2) at the messenger RNA or protein level occurs in about 20% of patients with early stage breast cancer (6) and has been identified in other types of carcinomas as well, including GC (7-8-9). The monoclonal antibody trastuzumab (Herceptin) is being used in treatment of breast and gastric carcinomas (3, 4, 9, 10). This humanized antibody binds to HER2, thereby preventing receptor dimerization and enabling immune recognition of tumor cells through antibody-dependent cell-mediated cytotoxicity, and causing endocytic degradation of cell surface-localized HER2 receptors (10-11-12-13-14). Trastuzumab binds directly to HER2 protein in gastroesophageal carcinomas and produces strong clinical benefit when combined with chemotherapy; this has led to an improvement of the overall survival rate (15-16-17).
Evaluation of HER2 is critical for selecting GC patients who could benefit from trastuzumab therapy (3, 4). Testing for HER2 amplification and/or overexpression is now considered routine practice in pathology laboratories for breast and gastric carcinoma therapy selection (3, 4, 18). HER2 testing is usually done by immunohistochemical assessment of HER2 protein or by fluorescence in situ hybridization (FISH) analysis of the HER2 gene (19-20-21-22). Analytical variability in HER2 immunohistochemical or FISH testing can be minimized by the use of standardized tests, by interlaboratory quality control assessment, and also by the use of automated procedures (19-20-21-22) such as the automated Bond™ system of Leica Microsystems.
In the present study, we sought to examine the suitability of the Bond system for automated HER2 detection by immunohistochemical and FISH techniques in GC biopsies for daily routine pathology practice.
Methods
Tumor Samples
One hundred formalin-fixed and paraffin-embedded GC biopsy specimens consisting of at least 2 fragments collected in each tumor by endoscopy were retrieved between 2010 and 2011 from the archives of the Institute of Anatomical Pathology at the Faculty of Medicine of the University of Coimbra, Portugal. This study was approved by the local ethics committee (Faculty of Medicine, University of Coimbra) as a retrospective study.
The GC samples were clinically identified as belonging to gastric antrum carcinomas and were selected according to the availability of malignant cells, with at least 60 malignant cells in each specimen. The GC cases were classified according to Lauren's criteria as intestinal type, diffuse type and indeterminate type.
Histology
Hematoxylin-eosin staining was performed using standard histology techniques. Stained sections were used to select areas with invasive carcinoma for subsequent HER2 immunostaining and HER2 FISH. To correlate HER2 immunostaining and HER2 FISH results at the cellular level, areas of interest were marked to compare the results. In all cases the expression of cytokeratin (MNF116) was evaluated to reinforce the identification of tumor cells in order to sustain FISH microscopic examination. This process also allowed the identification of cytokeratin-negative cells such as lymphocytes and stromal/fusiform cells in the GC biopsies. Tumor stroma inflammation was then scored to correlate with HER2 expression/copy number as follows: predominance of background lymphocytes (3+); balance between fusiform cells and lymphocytes (2+); predominance of stromal fusiform cells (1+).
HER2 Assessment by Automated IHC
From all tissue samples, 5-µm serial sections were placed on positively charged slides for immunohistochemistry (IHC) and FISH. The FDA-approved automated Bond Oracle™ HER2 IHC System (Leica Biosystems, Newcastle Upon Tyne, United Kingdom) was used for IHC staining of HER2 according to the manufacturer's instructions. IHC detection of HER2 was assessed in all samples and scored by conventional IHC criteria and according to the score recommended by Rüschoff (2): 0 (no reactivity or no membranous reactivity in any tumor cell); 1+ (tumor cell cluster with faint/barely perceptible membranous reactivity irrespective of the percentage of tumor cells stained); 2+ (tumor cell cluster with weak to moderate, complete, basolateral or lateral membranous reactivity irrespective of the percentage of tumor cells stained); 3+ (tumor cell cluster with strong, complete, basolateral or lateral membranous reactivity irrespective of the percentage of tumor cells stained). Samples given a 2+ score were considered equivocal and samples with a 3+ score were considered positive for HER2 overexpression according to the current recommendations (2).
HER2 Assessment by automated FISH
HER2 FISH analysis was carried out in all cases using the LSI HER2/CEP17 Dual Probe on the same automated Bond system (Leica Biosystems) according to the manufacturer's instructions. Slides were counterstained with 4’, 6-diamidine-2’-phenylindole dihydrochloride (DAPI) in anti-fade solution and examined using an automated CytoVision platform (Leica Biosystems). The complete tissue section was scanned to detect any subpopulations of amplified cells. HER2 gene amplification status was evaluated by counting signals in 20 non-overlapping tumor cells with the highest gene counts. The samples were analyzed using the standard criteria for HER2 positivity suggested by Rüschoff et al (2). A HER2: CEP17 ratio higher than 2.2 was defined as positive for HER2 amplification; a ratio between 1.8 and 2.2 was considered equivocal; a ratio lower than 1.8 was considered negative. In equivocal cases, we counted 20 additional tumor cells and considered the sample positive if the ratio was 2.0 or higher and negative if the ratio was lower than 2.0. Polysomy 17 was defined as the occurrence of 3 or more copy numbers of centromeres for chromosome (Chr) 17 per cell, according to Rüschoff et al (2).
All gastric biopsies where GC heterogeneity could not be achieved were analyzed by 2 independent technicians and validated by a pathologist in both FISH and IHC procedures. Procedure validation was done by analysis of standard control samples.
Statistical Analysis
All statistical analyses were performed using the SPSS statistical software for Windows, v. 22.0 (SPSS, Chicago, IL, USA). Correlations between clinicopathological parameters and HER2 status were analyzed using Fisher's exact test and the chi-square test. Intermethod agreement and comparisons of sensitivity and specificity between scoring systems were analyzed by calculation of concordance rates and Cohen's kappa coefficients. P values <0.05 were considered statistically significant.
Results
Patients’ Clinicopathological Characteristics
In this work we examined a total of 100 GC biopsies classified according to Lauren's classification: 44 of the intestinal type (44%), 38 of the diffuse type (38%), and 18 of the indeterminate type (18%). The patients’ ages ranged from 36 to 87 years (mean 67.65 ± 12.97 years), and the sample consisted of 58% men and 42% women. The histological characteristics, gender, age and inflammation status of the 100 tumor biopsies are presented in Table I.
Clinicopathological data distribution according to HER2 FISH status
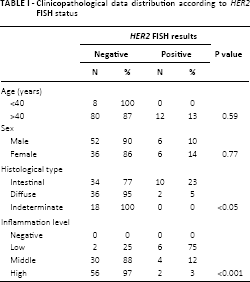
HER2 positivity was significantly different between the 3 histological types of GC (p<0.05): it was most frequent in the intestinal type (23%), while the diffuse and indeterminate types showed 5% and 0% positivity, respectively (Tab. I). Age and gender distribution showed no association with HER2 gene amplification, although HER2 amplification was only verified in the >40-year age group (Tab. I).
HER2 Gene Amplification Status by Automated FISH
The 100 GC biopsies were analyzed using the automated FISH method. We found that of the 44 intestinal-type GC cases, 34 (77.3%) were negative for HER2 amplification (ratio HER2/Chr17<1.8) and 10 (23.7%) showed HER2 amplification (ratio HER2/Chr17>2.2). Of the 38 cases of diffuse-type carcinoma, 36 (95%) were scored as negative for HER2 amplification (ratio HER2/Chr17<1.8) and 2 (5%) as positive (ratio HER2/Chr17>2.2). All 18 cases with indeterminate-type GC were considered negative for HER2 amplification after a second counting (ratio HER2/Chr17<2.0) (Tab. I), although in the first counting we considered 2 samples equivocal (ratios HER2/Chr17 between 1.8 and 2.2) (Tab. II).
Automated FISH HER2/CEP17 ratio results
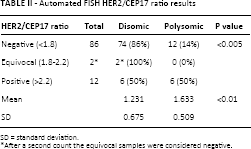
SD = standard deviation.
After a second count the equivocal samples were considered negative.
Taking the chromosome 17 polysomy results into consideration, we found a significant correlation (p<0.005) between chromosome 17 polysomy and HER2 amplification (Tab. II). Polysomy (Fig. 1A) was present in 18 cases, 12 of which were considered HER2 FISH negative with a ratio <2. The remaining 6 cases that presented polysomy were considered HER2 FISH positive (ratio ≥2) (Tab. II). On the other hand, disomy (Fig. 1B) was present in 82 cases, 74 of which (90.2%) were HER2 FISH negative (ratio <2) and 6 were considered HER2 FISH positive (ratio ≥2) (Tab. II). The remaining 2 cases were equivocal but after the second counting were judged as negative (Tab. II). Furthermore, the HER2/CEP17 ratio was found to be significantly different (p<0.01) between disomic and polysomic samples, being higher in polysomic samples (mean: 1.633 ± 0.509) than in disomic samples (mean: 1.231 ± 0.675) (Tab. II).
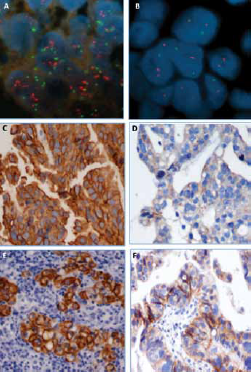
Automated HER2 status testing by FISH, IHC and MNF116. Examples of negative and positive cases. (
HER2 Protein Expression Status by Automated IHC
All 100 GC biopsies were analyzed using automated IHC. We found that of 44 cases of intestinal-type carcinoma, 34 (77.3%) were negative for HER2 overexpression (30 were scored 0 and 4 scored 1+), 4 (9.1%) were positive/equivocal (2+), and 6 (13.6%) were positive (3+) (Fig. 1C and 1D); of the 38 cases of diffuse-type carcinoma, 36 (95%) scored 0 (negative) and 2 (5%) had a score of 3+ (positive); all 18 cases with indeterminate-type carcinomas (100%) were considered negative for HER2 overexpression (14 with a score 0 and 4 with a score 1+).
In addition, we found that the tissue inflammation level was negatively correlated (p<0.001) with HER2 positivity, being higher (3+; Fig. 1E) in the HER2-negative group (97% of cases) and lower (1+; Fig. 1F) in the HER2-positive group (75% of cases) (Tab. I).
Concordance Analysis between Automated HER2 FISH and Automated HER2 IHC
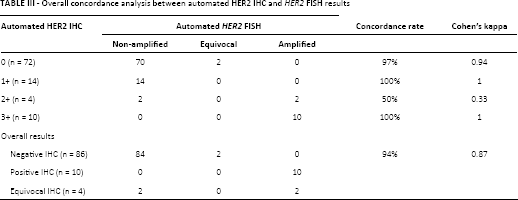
Both methods were successfully performed on all samples (Fig. 1) and we did not observe any FISH or IHC antibody failures. The overall concordance rate between the 2 methods was 94% (kappa coefficient [κ] 0.87) (Tab. III). Of the 84 cases with a negative IHC score (0 and 1+), 82 were considered FISH negative (concordance rate 97%, κ = 0.94) due to the presence of 2 equivocal cases in automated FISH. The 10 positive cases with automated IHC (3+ score) showed total equivalence with the automated FISH results (concordance rate 100%, κ = 1) (Tab. III). Of the 4 equivocal results (2+ score) in automated HER2 IHC, 2 showed HER2 amplification and the remaining 2 were considered negative (50% concordance level) (Tab. III).
Overall concordance analysis between automated HER2 IHC and HER2 FISH results
Discussion
Many studies defend that HER2 status could be important to determine the type of therapy that should be used in patients with GC (2, 5). In spite of all the handling problems, manual IHC and FISH are the most commonly used methods to evaluate HER2 status in the laboratory routine (1, 5). The European Medicines Agency and the College of American Pathologists recommend testing first with IHC studies and subsequent FISH for cases that are indeterminate (2+ immunohistochemical score) (8, 17). An IHC-first screening protocol demands high concordance between the IHC and FISH assays. In this work we tried to evaluate a recently FDA-approved automated system and its impact on the general results of our routine lab procedures. The automated systems demonstrated good quality results overall. HER2 protein overexpression was demonstrated in 10%-14% of GC biopsies, and HER2gene amplification was demonstrated in 12%. Although some studies found higher positivity levels among their subsets, and the levels of positivity can in some cases be explained by HER2 expression heterogeneity of GC in tumor samples (23), our results are in accordance with the normal range of positivity (7% to 38%) (2, 4). The overall concordance rate between the results of automated IHC and FISH was 94% (κ = 0.88). The concordance rate between the 2 methods showed a lower level (50%) only in samples with IHC 2+ scores, which is due to the 2+ score generally being considered equivocal (2, 3, 18). These results suggest that automated IHC is a good predictor of HER2 gene amplification and that the FISH test should only be done in IHC 2+ cases (2, 4). Our results support the idea that a complete, fully automated test such as the Oracle HER2 IHC system improves the daily routine, minimizing the risk of handling errors. Since other works using manual procedures showed higher levels of equivocal results and lower concordance rates (4, 5), we can say that automated procedures are without any doubt the best choice for routine practice.
In this study we also found an association between HER2amplification status and histological type of GC, namely higher positivity in the intestinal type (23% vs. 5% and 0%). These results are in agreement with previous studies indicating that HER2 overexpression is strongly associated with differentiated or intestinal-type GCs, which generally have a better prognosis than undifferentiated or diffuse-type cancers (2, 3, 7, 24, 25). Those studies also showed that the highest rates of HER2 overexpression were observed in patients with intestinal-type rather than diffuse or indeterminate-type GC (2, 3, 7, 24, 25). Beltran Gárate et al (26) showed that 70% of tumors that overexpressed HER2 were intestinal-type tumors and Hofmann et al (24) stated that of the 10.7% of tumors making up the IHC 3+ group in their study, 83.3% were of the intestinal type.
Generally the assessment of chromosome 17 polysomy did not contribute to amplification prediction. Most samples were found to be disomic (82%), but 50% of the HER2-positive samples were polysomic. Although some authors consider polysomy to be a major factor for IHC 2+ equivocal results (2-3-4, 18, 19, 24), in our work we did not find any correlation between the 2 conditions. Moreover, we found that polysomy was common in both HER2-positive (33%) and HER2-negative (67%) samples, whereas disomic samples were prevalent in the HER2-negative group (83%). Finally, the HER2/CEP17 ratio was higher in polysomic samples (1.633 vs. 1.231); this result is in agreement with previous studies that reported HER2 status to be inversely correlated with polysomy and disomy (3, 4, 18, 19, 24).
Another interesting finding of our study was the inverse correlation between HER2 positivity and the level of inflammation. It seems that HER2 positivity inhibits or is inhibited by the level of inflammation, being higher (3+) in HER2-negative samples and lower (1+) in HER2-positive cases. This issue has not been yet clarified, although some authors mention that the presence of CD8+ T lymphocytes is correlated with good prognosis in HER2-positive carcinomas including breast carcinoma and GC (27, 28). These results corroborate the importance of stromal immune processes for cancer growth and progression and their role in early carcinogenesis. Nevertheless, this assumption needs further study to be properly clarified.
In conclusion, our study demonstrated that automated HER2 testing by IHC and FISH using the Bond system of Leica Microsystems has high concordance levels and good performance rates in GC biopsy samples. This method allowed us to improve the routine procedures by minimizing the handling time, reducing personnel costs, decreasing the rate of equivocal samples, diminishing handling errors, and improving the HER2 status evaluation procedures overall. Another finding of our study is that the use of cytokeratin immunoexpression for neoplastic cell identification could be a simple technique to replace stromal typing. Therefore, such immunostaining could be applied in future studies with a wider range of cases for the purpose of immunotherapy prescription.
Footnotes
Acknowledgment
The authors acknowledge Portuguese Leica Biosystems services for technology support.
Financial support: None.
Conflict of interest: None of the authors have any competing interests related to this study.
