Abstract
Background
The advent of noninvasive urine-based markers as well as other novel modalities has yielded improved diagnostic accuracy. However, the new markers failed to reach higher sensitivity and specificity. We therefore evaluated the potential role of epithelial membrane antigen (EMA) and nuclear matrix protein 52 (NMP-52) singly and combined as noninvasive biomarkers for the detection of bladder cancer (BC).
Methods
A total of 160 individuals including 66 patients with BC, 54 patients with benign urologic disorders and 40 healthy volunteers were investigated. Urinary EMA at 130 kDa and NMP at 52 kDa were identified, purified and quantified by Western blot, electroelution and enzyme-linked immunosorbent assay (ELISA). The diagnostic performance of each biomarker and their combination were compared using area under receiver operating characteristic curves (AUC).
Results
Mean urinary EMA, 2.42 µg/mL, and NMP-52, 17.85 µg/mL, were significantly elevated in patients with BC compared to controls, 1.18 and 3.44 µg/mL, respectively (p<0.0001). The combined use of these markers yielded values which were increased 4.4- and 13.7-fold in the benign and malignant disease groups, respectively, with respect to the normal group. The values of EMA and NMP-52 were significantly higher in patients with higher-grade tumors than those with lower-grade tumors (p<0.0001). Moreover, this combination could predict all BC stages and grades with 0.91 AUC, 94% sensitivity and 80% specificity.
Conclusions
EMA and NMP-52 in combination could be promising noninvasive biomarkers for BC detection.
Introduction
Globally, bladder cancer (BC) is estimated to be the ninth most common malignancy, accounting for 3.3% of all malignancies. It is the second genitourinary cancer in incidence and mortality (1-3). The highest incidence rates are found in industrialized countries such as the United States, Canada and the European Union, while the lowest rates are found in developing countries in Africa and Asia (4, 5). Interestingly, high mortality rates (16.3 per 100,000) were found among Egyptian males. These rates are twice as high as the highest rates in Europe and over 4 times higher than in the United States (2). Only about 20%-25% of tumors are muscle invasive (MIBC, T2+) or metastatic at diagnosis, while the majority (75%-80%) of patients present with non-muscle-invasive tumors (NMIBC, stages <T2) (6-8).
Cystoscopy remains the gold standard procedure for BC diagnosis and monitoring. It is an unpleasant invasive diagnostic approach and expensive, rendering the surveillance program not well received by patients. The gold standard noninvasive BC diagnostic test is urine cytology, which also has its limitations: it is laborious and has suboptimal sensitivity, and there may be interobserver variability in low-grade tumors (1, 9, 10). In the face of the mentioned limitations of cystoscopy and cytology, investigators are searching for novel and better ways for BC diagnosis which should be noninvasive, sensitive, cost-effective and easy to perform (11, 12).
The use of urine samples in this context is a major advantage. Urine can be obtained noninvasively and abundant amounts of sample can be repeatedly obtained. Thus, researchers developed many new urine-based BC tests that have been approved by the US Food and Drug Administration (FDA) (6, 13). However, more urinary diagnostic tests are needed to reach higher sensitivity and specificity (12).
Nuclear matrix proteins (NMPs) are an important part of the structural framework of the nucleus (12). It has been demonstrated that the NMP-22 concentration is higher in BC cell lines than in urothelium from normal bladder (14, 15). In this respect, Attallah et al (16) identified a urinary NMP marker at 52 kDa (NMP-52) in the urine of BC patients. Epithelial membrane antigen (EMA) is a protein that exists on the exterior cell surface of the body (17). In diagnostic tumor histopathology, EMA has been suggested to be an indicator of epithelial differentiation (18-20). However, to date studies using EMA for urine analysis related to BC have been scarce. The current study was started to study EMA and NMP-52 levels in urine of BC patients compared with the levels in patients with benign bladder conditions and healthy individuals. We assessed the clinical utility of their urine concentrations as diagnostic biomarkers for the detection of BC by means of enzyme-linked immunosorbent assay (ELISA).
Materials and Methods
Patients and Samples
Voided urine samples of 160 individuals enrolled in this study were collected at the Urology and Nephrology Center, Mansoura University, Egypt. The gold standard method for identification of BC was urine cytology. In case of suspicious findings, a bladder biopsy was taken and the specimen was examined using histopathological techniques. The study participants were classified into 3 groups: 1) 66 BC patients having urothelial cell carcinoma or squamous cell carcinoma (including 42 men [63.6%] and 24 women [36.4%] with a mean age of 60.6 ± 10.6 years [range 44-81 years]); 2) 54 patients with noncancerous bladder cystitis (benign group) having inflammatory smears or hematuria (including 38 men [70.4%] and 16 women [29.6%] with a mean age of 58.6 ± 11.2 years [range 45-70 years]); 3) 40 healthy individuals as control group (26 men and 14 women, age range 45-65 years, mean age 56.2 ± 8.2 years). The BC patients were subdivided according to tumor stage into 17 with NMIBC (stages <T2) and 49 with MIBC (stages T2+), and according to differentiation grade into 15 with low-grade tumors (G1) and 51 with high-grade tumors (G2-3). Exclusion criteria were a history of another urological malignancy (kidney or ureter), recent urological surgery, and pelvic malignancy.
Fully informed consent was prospectively obtained from all study participants. The study protocol conformed to the ethical guidelines of the 1975 Helsinki Declaration.
Sample Collection
A single, naturally voided, first morning midstream urine sample was obtained prospectively from all patients. The collected samples were centrifuged at 2500-4000 g for 15-20 minutes and separated into supernatants and pellets. The cellular sediment, the pellet, was eliminated and the supernatant was used for subsequent analysis or stored at −20°C with no additions or treatment until tested.
Urinary Protein Separation using gel Electrophoresis
According to the method of Laemmli (21), urine proteins were separated in 0.75-mm-thick, 12% or 16% vertical slab gels. Randomly selected urine samples were mixed with the sample buffer (0.125 M Tris base, 4% sodium dodecyl sulfate, 20% glycerol, 10% mercaptoethanol, and 0.1% bromophenol blue as a tracking dye). A mixture of standard proteins (Sigma Chem Co., St Louis, MO, USA) ranging from 39.2 to 215.0 kDa was run in parallel. The protein pattern was determined by Coomassie blue staining.
Western blot
According to the method of Towbin et al (22), urine samples randomly selected from each set (healthy, cystitis and BC) separated as described above were electrotransferred onto a nitrocellulose membrane (0.45-µm pore size; Sigma). Western blot using EMA was run according to the methods of Attallah et al (23). A similar procedure was used for NMP-52 detection using the specific anti-NMP-52 antibody (ABC Diagnostics, New Damietta, Egypt) and the diluted anti-rabbit IgG alkaline phosphatase conjugate (Sigma). The dilutions of the antibodies and conjugates were adjusted to eliminate the background, i.e., the presence of the target antigen at low concentration.
Gel Electroelution
Using the method described by Attallah et al (23), both EMA and NMP-52 bands were electroeluted separately from unstained preparative gels. The protein content of purified EMA and NMP-52 was determined (24) and the remainder was stored at −20°C until use.
Quantitation of Urinary EMA and NMP-52 using ELISA
Voided urine samples were tested for EMA using the protocol of Attallah et al (23). The optical density (OD) at 405 nm was read using a ∑960 microplate autoreader (∑960, Metertech, Inc., Taipei, Taiwan). Color intensity was proportional to the amount of the target EMA concentration present in the urine sample. In a similar manner, the concentration of NMP-52 in each sample was measured using the specific anti-NMP-52 antibody and anti-rabbit IgG alkaline phosphatase conjugate. Urine samples from healthy volunteers and patients with noncancerous bladder cystitis were used to set the cutoff limit of the ELISA. A standard curve was determined by plot absorbance readings (0.10-0.70) of appropriately serially diluted fractions of the purified antigen within the range of 0.6 to 37.5 µg/mL for EMA and 1.1 to 110 µg/mL for NMP-52. To confirm the accuracy and reproducibility of these assays, using 10 samples the intra-assay variation (test precision) was determined in 3 repeats within 1 experiment. Interassay variation was investigated in 3 different experiments performed on 3 different days. The requirement for precision was acceptable if the coefficient of variation (%CV) did not exceed ±15%, either for intraday or interday assays (25).
Statistical Analysis
All statistical calculations were done with the Statistical Package for the Social Sciences (SPSS), v.15.0 (SPSS Inc., Chicago, IL, USA) and the GraphPad Prism package, v.5.0 (GraphPad Software, San Diego, CA, USA). Study participants’ characteristics were descriptively summarized and reported as mean ± standard error of the mean (SEM) or standard deviation (SD). To test the significance of differences between mean EMA and NMP-52 concentrations in different groups we used univariate analyses: Student's t-test and ANOVA. To test these markers in combination, logistic regression analysis was used. The regression coefficients in a logistic regression model were not estimable. Consequently, to develop a simplified score, and based on diagnostic performance, these coefficients were dropped from the model. Receiver operator characteristic (ROC) curves were used to determine the best cutoff values for optimal prediction of BC. The cutoff points were selected according to the point on the curve closest to the (0, 1) point (the minimal (1-sensitivity)2 + (1-specificity)2) (26). Common performance indicators of the candidate markers (sensitivity, specificity, efficiency, and positive and negative predictive values) were derived from a 2 × 2 contingency table.
Results
Identification of urinary EMA and NMP using Western Blot
The descriptive data on age, gender and clinicopathological data are shown in Table I. EMA was identified at 130 kDa in the urine of patients with BC. At the same molecular weight, urine samples from patients with benign cystitis showed a faint immunoreactive band. No reactive band was observed in urine from healthy individuals under this condition (Fig. 1A). Similarly, a highly immunoreactive band at 52 kDa was detected using the anti-NMP-52 antibody in the urine of BC patients. In contrast, faint bands in the urine of patients with nonmalignant bladder cystitis were observed and no bands were detected in urine of healthy individuals (Fig. 1B).
Characteristics of patients and healthy volunteers
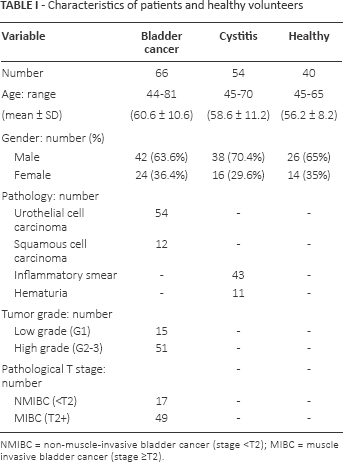
NMIBC = non-muscle-invasive bladder cancer (stage <T2); MIBC = muscle invasive bladder cancer (stage ≥T2).
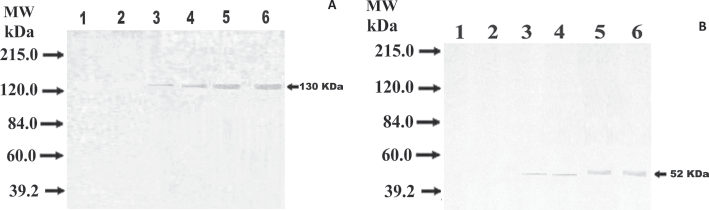
Western blots of urine samples randomly selected from bladder cancer patients showing the detection of
Urinary EMA and NMP-52 Concentrations and their Combination for BC Prediction
EMA and NMP-52 were purified separately from urine samples of BC patients using electroelution technique from preparative slab gels. To quantify EMA and NMP-52, their concentrations were determined in all of the urine samples using ELISA and their specific antibodies. For EMA, the intra-assay and interassay %CVs were 1.5% and 3.5%, respectively. For NMP-52, the intra-assay and interassay %CVs were 2% and 3.5%, respectively. The difference in urinary concentration of EMA, NMP-52 and their combination in patients with and without BC indicated potential diagnostic value. A ROC analysis of NMP-52, EMA and their combination gave AUCs of 0.877, 0.836 and 0.909, respectively (Fig. 2A-B-C) to discriminate BC urine samples from samples of normal individuals and cystitis patients combined. These values rose to 0.968, 0.928 and 0.995, respectively, when cancer patients were compared only to normal individuals (Fig. 2D-E-F). The optimal urinary EMA, NMP-52 and combination thresholds for cancer versus noncancer were 1.8, 2.5 and 4 µg/mL, respectively. Use of the EMA cutoff gave 90% sensitivity for all BC patients with a specificity of 80%. The sensitivity for NMIBC and MIBC was 85% and 92%, respectively. On the other hand, use of the NMP-52 threshold gave 87% sensitivity for all BC patients with a specificity of 83%. The sensitivity for NMIBC and MIBC was 85% and 88%, respectively. Remarkably, when urinary EMA was combined with NMP-52 for identifying all BC stages or grades, higher sensitivities and specificities were reached. The performance characteristics of urinary EMA, NMP-52 and their combination are shown in Table II.
Summary of data generated for bladder cancer patients compared with noncancer controls (54 patients with benign cystitis and 40 healthy individuals)
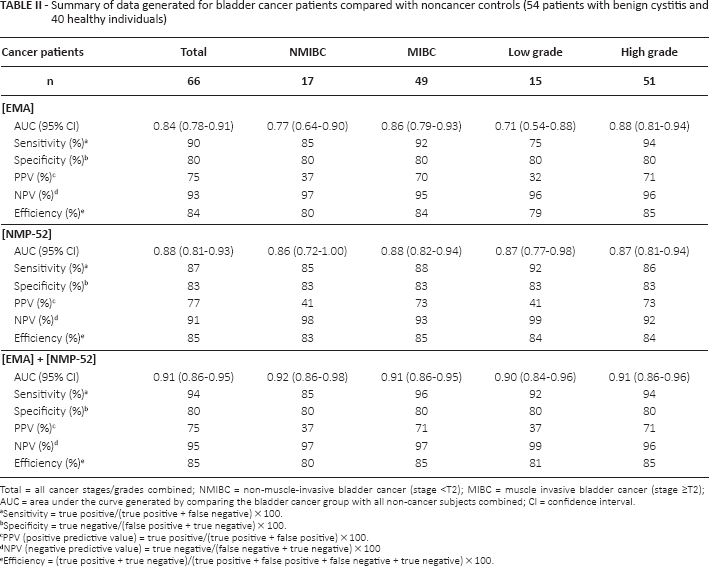
Total = all cancer stages/grades combined; NMIBC = non-muscle-invasive bladder cancer (stage <T2); MIBC = muscle invasive bladder cancer (stage ≥T2); AUC = area under the curve generated by comparing the bladder cancer group with all non-cancer subjects combined; CI = confidence interval.
Sensitivity = true positive/(true positive + false negative) × 100.
Specificity = true negative/(false positive + true negative) × 100.
PPV (positive predictive value) = true positive/(true positive + false positive) × 100.
NPV (negative predictive value) = true negative/(false negative + true negative) × 100
Efficiency = (true positive + true negative)/(true positive + false positive + false negative + true negative) × 100.
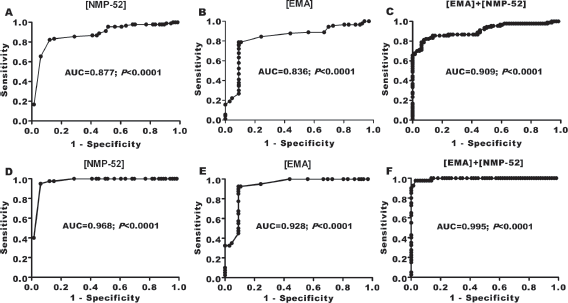
ROC analysis for EMA, NMP-52 and their combination to calculate the best cutoff point to discriminate bladder cancer urine samples from either
Urinary levels of EMA, NMP-52 and their Combination between Investigated Groups
The distribution of urinary EMA and NMP-52 levels (Fig. 3A and B) and their combination (Fig. 4A) among the study groups is presented as box plots. In BC patients, the concentrations (mean ± SEM) of EMA, NMP-52 and their combination were significantly (p<0.0001) higher (2.42 ± 0.15, 17.85 ± 2.78 and 20.24 ± 2.84 µg/mL, respectively) than those of healthy individuals (0.85 ± .08, 0.70 ± 0.12 and 1.55 ± 0.15 µg/mL) and noncancer patients (1.18 ± 0.08, 3.44 ± 0.93 and 4.62 ± 0.94 µg/mL). There was no significant difference (p = 0.71) between EMA+NMP-52 values in NMIBC (19.8 µg/mL) and MIBC (22.2 µg/mL). However, the values of patients with higher-grade tumors (22.1 µg/mL) were significantly (p<0.0001) higher than those of patients with lower-grade tumors (12.7 µg/mL) (Fig. 4B). The combined use of EMA and NMP-52 yielded values which were increased 4.4-fold and 13.7-fold in the benign and malignant disease groups, respectively, compared with the normal control group (Fig. 4C). The distributions of urine EMA + NMP combination values have a sigmoid pattern, showing the likelihood of patients having BC. The increase in this likelihood was related to EMA + NMP at concentrations greater than 4 µg/mL (Fig. 4D).
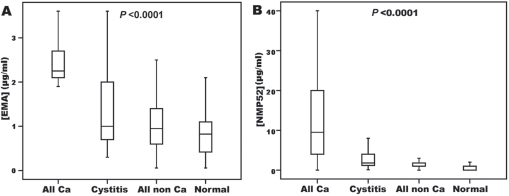
Box plots for
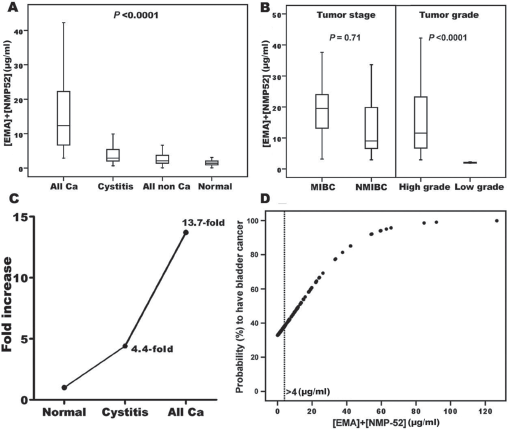
Box plots showing the distribution of EMA + NMP-52 values (
Discussion
On premalignant and malignant cells tumor antigen expression can be detected using monoclonal antibody staining (27). In our study, EMA and NMP-52 were identified in urine of BC patients using specific monoclonal antibodies and Western blot at 130 kDa and 52 kDa, respectively. In contrast to urine of the control group, intense immunoreactive bands appeared in the urine of patients with different types of BC. These results further suggest that EMA and NMP-52 could be used as potential biomarkers for the diagnosis of BC. Therefore, we purified the 2 target antigens and developed a direct ELISA for the quantitation of EMA and NMP-52 separately in urine of patients with BC. To overcome the possible interfering effect of dieresis, the levels were corrected by dividing each concentration of the biomarkers by those of urinary creatinine. Our results revealed a marked increase in the mean ± SEM (µg/mL) of EMA, NMP-52 and their combination in BC patients versus the group with benign bladder disease and the group of normal individuals (p<0.0001). The most likely explanation for these results would be that EMA is expressed at a very low level on normal adenocytes, while the immune system recognizes EMA as a glycoprotein antigen on cancer cells (28, 29). NMP-52 is probably released from the nuclei of tumor cells during apoptosis (16).
Using cystoscopy as the gold standard for urinary sample categorization, ROC analyses rendered an area of 0.836 and 0.877 for EMA at a 1.8 µg/mL cutoff and NMP-52 at a 2.5 µg/mL cutoff, respectively. In the evaluation of urinary EMA as a diagnostic biomarker for BC, EMA was detected in all types of BC with an overall sensitivity of 90% and a specificity of 80%. Conversely, NMP-52 could be utilized as a diagnostic tool to distinguish patients with BC from controls with a sensitivity of 87% and a specificity of 83%. In comparison, urine cytology has a high diagnostic specificity (78%-100%), but lacks robust sensitivity (12.2%-84.6%), especially for low- and intermediate-grade tumors (8, 29). The presence of NMP-22 in urine represents the most sensitive protein-based noninvasive test for early-stage BC. It has a sensitivity of 46%-48% for stages <T2. Since its sensitivity for stages <T2 does not exceed 50%, its diagnostic utility for NMIBC is limited (8). A number of other investigational urine markers have been described in recent reviews (13, 30-33). However, to date no marker has reached widespread use. Although several urinary markers have shown higher sensitivity than cytology, most suffer from low specificity. Another limitation is that every setting (e.g., screening/early detection and surveillance) has different requirements. Finally, each marker has to prove its cost-effectiveness. Interestingly, the combined use of EMA and NMP-52 revealed higher diagnostic value (AUC of 0.909) at a 4 µg/mL cutoff with higher sensitivity (94%) and specificity (80%). Higher-grade tumors were associated with higher mean values of the urinary EMA+NMP-52 combination than lower-grade tumors (p<0.0001). Since low-grade tumors undergo less apoptosis than more advanced tumors (34), the NMP-52 release might also be expected to be less. The overexpression of EMA by carcinomas is usually hypoglycosylated and is related to greater tumor aggressiveness (35). Very low urinary EMA+NMP-52 levels (1.5 µg/mL) were found in the healthy control samples. Similarly, in the noncancer samples there were low levels of EMA+NMP-52 (4.5 µg/mL). A low baseline level is an advantage that may allow a clear distinction between a healthy and disease state.
Future studies are needed to clarify the prospective biological behavior of these markers in different pathological types of BC. To our knowledge, this study is among the first to use EMA as a urine marker for BC. Taken together, our results highlight the potential utility of the combined use of EMA and NMP-52 as a urinary biomarker for BC.
Footnotes
Acknowledgment
The authors would like to thank the staff of the Biotechnology Research Center for their involvement in the experimental work.
Financial support: None.
Conflict of interest: The authors have stated that they have no conflicts of interest.
