Abstract
Background
Ductal carcinoma in situ (DCIS) is a heterogeneous disease that has not been investigated as widely as invasive breast cancer. Thus, the search for biomarkers capable of identifying DCIS lesions that may recur or progress to invasive cancer is ongoing. Although conventional steroid hormone receptors, cell proliferation and other important tumor markers have been extensively studied in invasive tumors, little is known about the role played by androgen receptors (ARs), widely expressed in breast cancer, in DCIS.
Methods
We performed a retrospective study in a series of 43 DCIS patients treated with quadrantectomy only and followed up for a period ranging from 5 to 13 years, to evaluate the prognostic relevance of conventional biomarkers (estrogen receptor [ER], progesterone receptor [PgR], Ki67, human epidermal growth factor receptor 2 [HER2]) and AR.
Results
Our findings showed that AR and ER were not independent prognostic variables and that an AR/ER ratio cutoff of 1.13 showed a sensitivity of 75% and a specificity of 94% in predicting in situ relapse or progression to the invasive phenotype. Moreover, while the variables considered singly showed area under the curve (AUC) values ranging from 0.52% to 0.77%, the AR/ER ratio reached a very high AUC (0.92%).
Conclusions
These preliminary results highlight the potentially important role of AR and ER and, in particular, of their ratio, as prognostic indicators of DCIS evolution.
Introduction
Breast cancer is the most frequent malignancy and the second cause of death from cancer in women (1). It is detected as an invasive ductal lesion in up to 80% of cases and as a lobular lesion in 5% to 15%. The high incidence of invasive breast cancer has led to intensive research to better identify biological profiles corresponding to different prognostic risks that can provide useful information to plan tailored therapy. An important breakthrough was recently obtained by the discovery of biological subgroups of lesions such as triple negative (estrogen receptor negative [ER-], progesterone receptor negative [PgR-], human epidermal growth factor receptor 2 negative [HER2-]) or triple unfavorable (ER-, PgR-, Ki67 >20%) (2, 3) that denote tumors with different aggressiveness and which differ in their response to conventionally used treatments and drugs (2).
The past few years have seen a renewed interest in the study of in situ breast cancers, and the substantial increase in the diagnosis of small tumors (from 5% to 20%) has led to screening programs now being acknowledged as an important diagnostic tool. However, a close examination of results reveals that there has been a large overestimation (false-positive cases) of breast cancer diagnosis, resulting in 3% to 14% of women being submitted to unnecessary treatment for tumors that will never develop (4). This limiting aspect of screening, supported by results from important studies conducted in this area (5, 6), has led to greater research efforts to identify the biological features of early lesions.
We studied conventional biomarkers currently determined in invasive cancers (2) to see which, if any, could accurately identify ductal carcinoma in situ (DCIS) destined to have a malignant evolution. Women with in situ breast cancer are mainly treated by either total mastectomy (with or without sentinel node biopsy) or conservative surgery (usually without sentinel node biopsy) and almost always by radiotherapy to complete the therapeutic efficacy of surgery. However, radiation treatment is not always effective, and in addition to being a heavy economic burden for National Health Systems, it occasionally causes long-term toxicity and psychological problems in patients.
Steroid hormone receptors, cell proliferation and other important tumor markers are conventionally used to study the biological profiles of invasive tumors. Androgen receptors (ARs), albeit widely expressed in breast cancer with uncertain significance (7), have not been extensively studied in DCIS. In the present work we evaluated the real prognostic impact of standard biomarkers (ER, PgR, Ki67 and HER2) and AR on DCIS recurrence/evolution in a case series treated with surgery alone.
Materials and Methods
This retrospective study was carried out in a series of 43 patients with DCIS diagnosed between 2001 and 2009 (median follow-up of 8 years) during screening at the Cancer Prevention Unit and followed at the Breast Surgical Unit of Morgagni-Pierantoni Hospital, Forlì. All patients were aged ≥18 years, had a histological diagnosis of DCIS treated with quadrantectomy only, and were followed up for a period ranging from 5 to 13 years. Patients with infiltrating carcinoma (IC) at diagnosis were excluded. Recurrent disease in patients was defined as a DCIS or IC lesion occurring more than 12 months after surgery.
The original hematoxylin-and-eosin stained sections were reviewed by 2 pathologists responsible for selecting pathological inclusions representative of tumor tissue. The study protocol was reviewed and approved by the IRST Ethics Committee, and patients gave their written informed consent for their biological material to be used for research purposes.
Biomarker Determination
Four-micron sections of neutral buffered formalin-fixed, paraffin-embedded tissue were mounted on positive-charged slides (Bio Optica, Milan, Italy), deparaffinized with xylene and rehydrated in decreasing concentrations of ethanol to distilled water. Endogenous peroxidase activity in rehydrated deparaffinized sections was blocked using 3% hydrogen peroxide solution; biomarkers were determined by immunohistochemistry. ER and PgR determinations were performed according to European Quality Assurance guidelines (8). Moreover, positive and negative breast tissue was used as intra-assay and interassay control for all biomarkers.
For AR, ER, PgR, Ki67 and HER2 determination, tissue sections were treated with epitope retrieval solution (0.01 M citrate buffer, pH 6.0) in a water bath at 98.5°C for 40 minutes, followed by a 20-minute cooling period at room temperature. The sections were treated with 3% bovine serum albumin in phosphate-buffered saline (PBS) for 20 minutes, after which they were incubated at room temperature with the different primary antibodies (2, 3). ER, PgR, Ki67 (Leica, Novocastra, Newcastle, UK), HER2 (Dako, Carpinteria, CA, USA) and AR (Roche Ventana, Tucson, AZ, USA) antibodies were used. For ER, PgR, Ki67 and HER2 detection, tissue sections were incubated for 60 minutes with antibodies diluted 1:80, 1:40, 1:100 and 1:350, respectively, in antibody diluent with background reducing components (Dako Corporation). AR antibody was ready to use and thus not diluted. The sections were then rinsed with PBS, incubated with universal biotinylated secondary antibody for 15 minutes, washed in PBS and incubated with streptavidin-peroxidase conjugate for 15 minutes (LSAB+kit; Dako Corporation). Sections were rinsed again in PBS, and the immunoreaction was developed using diaminobenzidine hydrogen peroxidase chromogen solution (Dako Corporation).
The sections were then counterstained with hematoxylin and washed several times with cold tap water. Biomarker positivity was detected and semiquantitatively quantified as the percentage ratio between the immunopositive and total number of tumor cells.
All samples were evaluated by 2 independent observers. A disagreement of more than 10% of positive cells, observed in 15%-22% of cases for the different biomarkers, was resolved by consensus after joint review using a multihead microscope. The conventional biomarkers were classified on the basis of the most recent St. Gallen and ASCO-CAP guidelines (2013) (9, 10).
Statistical Analysis
Nonparametric ranking statistics (median test) were used to analyze the relationship between the median values of biomarkers. Spearman's correlation was used to investigate the relationship between the different biomarkers considered as continuous variables. The accuracy of single or combined biomarkers, considered as continuous variables, was measured using the area under the curve (AUC) to evaluate the differences in diagnostic accuracy. In the receiver operating characteristic (ROC) curves, true positive rates (sensitivity) were plotted against false positive rates (1-specificity) for all classification points. All p values were based on 2-sided testing, and statistical analysis was carried out using SAS software, version 9.3 (SAS Institute, Cary, NC, USA).
Results
All patients were submitted to surgery alone and followed up for a maximum of 13 years. Five patients relapsed during this period, 1 at 3 years after diagnosis, 2 after 7 years and 2 after 10 years. All 5 markers (AR, ER, PgR, Ki67 and HER2) were analyzed in the entire case series. Statistical analysis showed that the different markers, when considered singly, were not predictive of relapse; ER (Tab. I) was the only variable whose median value was at the limit of statistical significance (p = 0.056). Proliferative activity, evaluated as level of Ki67 or HER2, both of which are acknowledged as important indicators of aggressiveness in invasive tumors, showed no prognostic relevance in DCIS (Tab. I).
Marker expression in tumor cells of relapsed and nonrelapsed patients
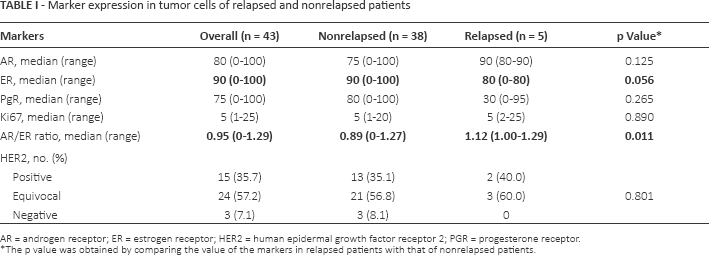
AR = androgen receptor; ER = estrogen receptor; HER2 = human epidermal growth factor receptor 2; PGR = progesterone receptor.
The p value was obtained by comparing the value of the markers in relapsed patients with that of nonrelapsed patients.
The analysis of AR and ER expression (Fig. 1) showed that, when considered singly, neither was indicative of relapse (AR, p = 0.125; ER, p = 0.056) (Tab. I). It also revealed that the AR/ER ratio in relapsed patients was significantly different from that of nonrelapsed cases (p = 0.011) and, at the best cutoff of 1.13, showed a sensitivity of 75% and a specificity of 94% in predicting in situ relapse or progression to invasive carcinoma. An AR/PgR ratio cutoff of 1.00 showed a sensitivity of 75% and specificity of 53%, while a cutoff of 3.00 had a sensitivity of 50% and a specificity of 84% (data not shown). Moreover, while AUC values of single variables ranged from 0.52% to 0.77%, the AR/ER ratio reached a very high AUC (0.92%) (Tab. II).
Carcinoma in situ of the breast:
Area under the curve values for markers in tumor cells
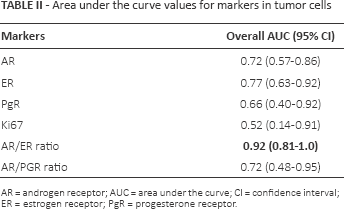
AR = androgen receptor; AUC = area under the curve; CI = confidence interval; ER = estrogen receptor; PgR = progesterone receptor.
Discussion
Despite an enthusiastic and promising start to the use of screening for the detection of small in situ breast cancers, the analysis of results from different studies has led to disappointing, somewhat alarming conclusions. A problem of overdiagnosis has emerged, involving, in some cases, unnecessary treatments, and causing a negative psychological and social impact on patients and a considerable economic burden on health services. This prompted us to review the conventionally used biological characterization of invasive tumors which has been shown to provide important clinical information. We planned a similar study of biomarker characterization in the less widely investigated area of ductal carcinoma in situ (7, 11, 12). The case series was limited because of the difficulty in finding patients treated with surgery alone, the majority of whom are also treated with adjuvant radiotherapy. The impact of radiation on the natural history of the tumor has yet to be determined. Few patients relapsed, an indication of the low percentage of DCIS that are destined to recur or progress when treated with surgery alone. We chose to evaluate 5 variables: proliferative activity, HER2 amplification and 3 hormonal markers (AR, ER and PgR). The proliferative activity of the cell population, a source of such important information on invasive tumors (13), and which is used as a prognostic variable and also to select the type and intensity of drugs for tailored therapy (2), did not provide any useful clinical information on DCIS progression. Similarly, HER2 status, whether negative, positive or equivocal, did not influence the evolution of DCIS.
Conversely, interesting results were obtained on 2 of the hormonal markers. Little information is available on AR status in DCIS, and its prognostic significance in invasive tumors is still very much open to debate (14, 15). A recent study by Cochrane et al concluded that an AR/ER ratio >2 was an independent predictor of disease-free and disease-specific survival (15). In particular, the authors suggested that a high AR/ER ratio may influence breast cancer response by increasing the risk of tamoxifen failure. We observed a prognostic value for ER which improved when the marker was combined with AR. In particular, the AR/ER ratio was highly accurate in predicting disease recurrence, identifying aggressive tumors with a potentially malignant evolution in 0.92% of cases.
In addition to good accuracy, these markers have the advantage of being determined by immunohistochemistry, a widely used laboratory method that is less expensive than clinical instrumental determinations.
In conclusion, our preliminary results suggest that AR and ER may play an important role in identifying DCIS that will relapse or progress and could thus furnish important biological information when planning therapy.
Footnotes
Acknowledgment
The authors thank Ursula Elbling for editing the paper.
Financial support: This study was funded by Italian Health Ministry project no. GR-2009-1594885.
Conflict of interest: None.
