Abstract
Background
The glycoprotein MUC1 is overexpressed and underglycosylated in cancer cells. MUC1 is translated as a single polypeptide that undergoes autocleavage into 2 subunits (the extracellular domain and the cytoplasmic tail), and forms a stable heterodimer at the apical membrane of normal epithelial cells. The MUC1 cytoplasmic tail localizes to the cytoplasm of transformed cells and is targeted to the nucleus.
Aims
To study the expression of the MUC1 extracellular subunit in cell nuclei of neoplastic breast, head and neck, and colon samples.
Materials and Methods
330 primary tumor samples were analyzed: 166 invasive breast carcinomas, 127 head and neck tumors, and 47 colon tumors; 10 benign breast disease (BBD) and 40 normal specimens were also included. A standard immunohistochemical method with antigen retrieval was performed. Nuclear fractions from tissue homogenates and breast cancer cell lines (ZR-75, MDA-MB-231, MCF7, and T47D) were obtained and analyzed by Western blotting (WB). The anti-MUC1 extracellular subunit monoclonal antibody HMFG1 was used for immunohistochemistry.
Results
37/166 breast cancer specimens, 5/127 head and neck cancer specimens, 2/47 colon cancer samples, and 3/10 BBD samples showed immunohistochemical staining at the nuclear level. No nuclear reaction was detected in normal samples. By WB, breast and colon cancer purified nuclear fractions showed reactivity at 200 kDa in 3/30 breast and 3/20 colon cancer samples as well as purified nuclear fractions obtained from breast cancer cell lines.
Conclusions
This study shows that the MUC1 extracellular domain might be translocated to the cell nucleus in breast, head and neck, and colon cancer as well as BBD.
Introduction
MUC1 is a large transmembrane glycoprotein expressed by epithelial cells; it is overexpressed and underglycosylated in cancer cells. MUC1 is translated as a single polypeptide that undergoes autocleavage into 2 subunits (the extracellular domain, MUC1-N, and the cytoplasmic tail, MUC1-C) in the endoplasmic reticulum (1). It forms a stable heterodimer at the apical membrane of normal epithelial cells (2, 3). The MUC1-N terminal subunit has a protein core mainly composed of a variable number of tandem repeats (VNTR). Each tandem repeat consists of a sequence of 20 amino acids which contains 5 potential glycosylation sites in Ser and Thr (4, 5). Normal epithelium presents long carbohydrate chains, while tumor cells display an altered pattern of glycosylation (6, 7). With transformation and loss of polarity, MUC1 is found in the cytosol and over the entire plasma membrane of carcinoma cells (8, 9). Early research into MUC1 focused on the MUC1-N mucin component and led to development of the CA 15-3 assay to monitor circulating levels of this subunit as a tumor marker (9).
Subsequent research focused on the MUC1-C transmembrane subunit as the potential link between its overexpression and carcinogenesis; it consists of a 58-amino-acid extracellular domain, a 28-amino-acid transmembrane domain, and a 72-amino-acid cytoplasmic tail (10). The MUC1 cytoplasmic tail interacts with a variety of proteins involved in neoplasia and cell adhesion such as the epidermal growth factor receptor (EGFR), erbB2, erbB3, erbB4, c-Src, protein kinase C delta (PKCd), Grb2, β-catenin, GSK3β, and p120ctn (11-17). The MUC1 cytoplasmic domain has proved sufficient to induce transformation; it also localizes to the cytoplasm of transformed cells and is targeted to the nucleus and mitochondria (18, 19). Furthermore, it was found to be associated with apoptosis resistance in response to oxidative stress (20).
It is frequently assumed that the MUC1 extracellular subunit does not enter the nucleus. In a previous study (21) where we used Western blotting (WB) with the anti-VNTR MUC1 C595 monoclonal antibody (MAb), we detected the MUC1 extracellular domain in nuclear fractions of primary breast cancer samples: a double band at 180 kDa was observed along with a smear reaction from 50-60 kDa up to 200 kDa. Based on these observations, we performed the present study with the aim to detect extracellular MUC1 in the cell nuclei of neoplastic tissues.
Materials and Methods
Tissue specimens
A total of 330 primary tumor samples were analyzed: 166 invasive breast carcinomas, 127 head and neck squamous cell carcinomas (HNSCC), and 47 colon adenocarcinomas. Forty normal tissues were included as controls: 20 mammary samples, 10 colon mucosa specimens, and 10 head and neck mucosa specimens. Ten benign breast disease (BBD) samples were also studied: 5 fibroadenoma (FA) specimens, 3 atypical hyperplasia (ADH) specimens, and 2 nonproliferative disease (NPF) specimens.
Each tissue was studied using routine procedures. Specimens were fixed for histopathological diagnosis and immunohistochemistry (IHC) analysis. Before formalin fixation, a fraction of the tumor sample was rinsed with sterile Hank's balanced solution and processed for preparation of subcellular fractions for SDS-PAGE and WB analysis.
The procedures followed were in accordance with the Helsinki Declaration. Informed consent was obtained from all patients included in this study. The study was approved by the Ethics Approving Committee, Faculty of Medical Sciences, National University of La Plata.
Cell lines
The breast cancer cell lines T47D, MDA-MB-231, MCF7, and ZR-75 were grown in RPMI 1640 medium (Sigma, USA) supplemented with 10% fetal calf serum (PAA, Austria) and 100 units/mL penicillin and 100 μg/mL streptomycin (Sigma, USA) at 37°C in a humidified atmosphere of 95% air and 5% CO2.
Antibody
We used the monoclonal antibody HMFG1; it was developed against the epithelial component of the human milk fat globule membrane, which reacts with the PDTR epitope present in the VNTR of MUC1-N (22, 23).
Methods
Immunohistochemical analysis
The technique was performed following standard procedures, with modifications (21). Briefly, 4-µm-thick sections were deparaffinized and rehydrated; antigen retrieval was performed in 10 mM sodium citrate buffer at 100°C for 5 minutes with incubation overnight at 4°C with MAb HMFG1, which was employed undiluted. After 3 washes with phosphate buffer, peroxidase-conjugated goat antimouse IgG (Dako, USA) diluted 1:400 was added and incubated for 60 minutes. The slides were counterstained with hematoxylin and coverslipped with mounting medium.
Negative controls were incubated with phosphate-buffered saline (PBS) instead of MAb. Staining of the cytoplasm, plasma membrane, and nucleus was evaluated. Sections were examined by light microscopy and the antibody staining patterns were scored in a semiquantitative manner. The staining intensity was graded as negative (-), low (+), moderate (++), or strong (+++) (21).
The number of low-power (×10) optical fields in a specimen that were positively stained was expressed as a percentage of the total number of optical fields containing tissue.
Preparation of subcellular fractions
Fractions were prepared from breast, colon, and head and neck tumors according to Price et al (23) and Boisvert et al (24). Samples were exhaustively dissected, necrotic areas were discarded, and tumors were cut into 1-mm pieces and extensively washed with 10 mM Tris, pH 7.2. Immediately after, mechanical homogenization was performed with a Polytron (Kinematica, Switzerland) in an ice bath employing the same buffer with 0.01 M protease inhibitor cocktail (Sigma, USA). Homogenates were filtered through a metallic mesh and centrifuged at 600×g at 4°C for 15 minutes, obtaining a supernatant which corresponds to the cytoplasmic and extranuclear membranes, and a pellet which corresponds to the nuclear subcellular fraction (21). This pellet was resuspended in 0.75 mL of 0.88 M sucrose, 0.5 mM MgCl2 followed by centrifugation at 2800×g for 10 minutes at 4°C. The final pellets (purified nuclear fractions) were resuspended in PBS and the protein content was measured by means of the Bradford method. Finally, fractions were aliquoted and stored at −70°C.
Subcellular fractionation of cell lines
Cells were grown in 10-cm plastic dishes until subconfluence, harvested by trypsinization and then washed 3 times with cold PBS at 300×g for 5 minutes at 4°C. The cells were subsequently incubated at 4°C with 2 mL of 10 mM HEPES pH 7.9 (Sigma, USA), 10 mM KCl (Merck, Germany), 1.5 mM MgCl2 (Merck, Germany), and 0.5 mM DTT (Sigma, USA) supplemented with a protease inhibitor cocktail (Sigma, USA). Cells were transferred to a dounce homogenizer (Sigma, USA) and 10 strokes were applied while cell lysis was verified under a phase contrast microscope. Homogenized cells were centrifuged at 300×g for 5 minutes at 4°C; thus we obtained a supernatant (cytoplasmic and extranuclear membrane fraction) and a pellet (nuclear fraction) which was applied to a sucrose cushion to obtain the purified nuclear fraction.
Final pellets (purified nuclear fractions) from tumors as well as cell lines were dialyzed against 1.41 M PBS at 4°C for 48 hours, lyophilized, and stored at −70°C. Samples were analyzed by SDS-PAGE and WB.
SDS-PAGE and western blotting
The isolated fractions were analyzed under reducing conditions in SDS-PAGE in a discontinuous buffer system (4% for the stacking gel and 7% for the resolving gel). Fractions were suspended in sample buffer and heated at 100°C for 5 minutes; on each lane the protein content loaded was 40 µg/25 µL. After electrophoresis, gels were electrophoretically transferred onto nitrocellulose membranes, which were blocked with PBS/5% skimmed milk (blocking buffer), washed and incubated with undiluted MAb HMFG1. After overnight incubation at 4°C, the blots were rinsed 6 times for 5 minutes in PBS. Then peroxidase-conjugated goat antimouse IgG (Dako, USA) diluted 1:2000 was added. Nitrocellulose sheets were developed employing enhanced chemiluminescence.
Results
Thirty-seven of 166 (22%) breast cancer specimens reacted at the nuclear level. The tumor stage distribution was 12/37 (32%) stage I, 15/37 (40.5%) stage II, and 10/37 (27%) stage III. Estrogen and progesterone receptor expression was 84% and 81%, respectively, whereas HER2/neu expression was found in 9/35 samples (26%).
Despite nuclear reactivity, 100% of breast cancer specimens reacted with the anti-MUC1-N MAb HMFG1; most samples showed nonapical, cytoplasmic MUC1 staining with a strong intensity, while several samples showed an apical pattern with a linear, cytoplasmic or mixed reaction. Figure 1A shows the HMFG1 reactivity in a breast carcinoma specimen; the nuclei exhibit strong reactivity as does the cytoplasm of most cells.
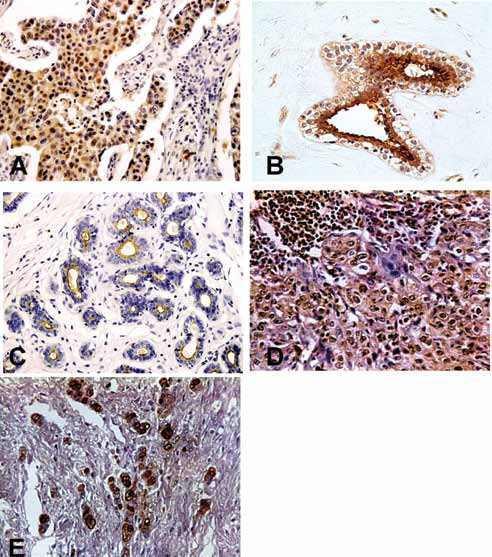
A) Positive staining of an invasive ductal carcinoma for MAb HMFG1. A strong nuclear and cytoplasmic reaction is depicted. B) Duct with intact lumen belonging to an fibroadenoma sample incubated with MAb HMFG1; a strong reaction involves the nucleus, apical plasma membrane, and apical cytoplasm of epithelial cells. C) Normal mammary sample incubated with MAb HMFG1; reaction is found in the apical plasma membrane. D) Larynx carcinoma section incubated with MAb HMFG1; most neoplastic cells show strong reactivity at the nuclear and cytoplasmic level. E) Intense positive nuclear staining for MUC1-N of an undifferentiated colon carcinoma detected with MAb HMFG1.
We also found nuclear reactivity in 3 of 10 BBD samples including 1 FA, 1 ADH and 1 NPF. Figure 1B shows a positive nuclear reaction in the FA specimen. In most BBD specimens, HMFG1 stained the apical part of the cytoplasm and plasma membranes, while in normal samples only apical reactivity was detected with no nuclear staining (Fig. 1C).
Forty percent of HNSCC and 95% of colorectal cancer specimens reacted positively with HMFG1. In most HNSCC and colorectal cancer samples, MUC1 reactivity was mainly restricted to the cytoplasm and the plasma membranes were negative; the intensity varied from moderate to strong. Also, 5/127 (4%) HNSCC specimens and 2/47 (4%) colorectal cancer samples showed a nuclear HMFG1-positive reaction. In Figure 1D and 1E the reactivity is shown. HMFG1-positive HNSCC tumors were located in the larynx (4/5) and pharynx (1/5). Patients with laryngeal tumors were at stage II (1/4) or stage III (3/4) with moderate (1/4) and poor (3/4) histological grade. The patient with pharynx cancer presented at stage IV with a well-differentiated tumor, while patients with colorectal adenocarcinomas were in stages II and III.
Confirmation of nuclear localization was obtained by analyzing nuclear fractions by WB with HMFG1. In 3 of 30 breast cancer samples and 3 of 20 colorectal cancer samples, typical MUC1 extracellular domain reactivity at about 200 kDa was detected. Figure 2 shows a representative experiment of a WB reaction on purified nuclear fractions obtained from breast cancer (lane 2) and colon cancer (lane 3) samples.
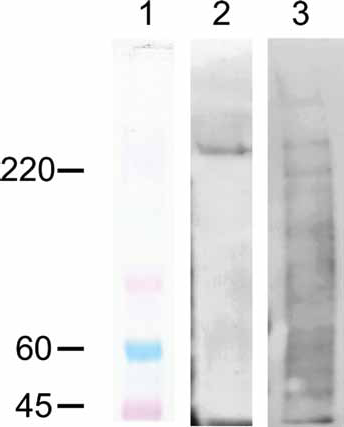
Western blot of purified nuclear fractions obtained from a breast cancer (lane 2) and a colon cancer (lane 3) incubated with MAb HMFG1. A >220 kDa band is detected for both samples; the colon cancer sample also shows several low-molecular-weight bands. In lane 1 the molecular weight standard is depicted.
Figure 3A shows WB results obtained in 4 breast cancer cell lines (T47D, MDA 231, ZR75, and MCF7). The subcellular fractions assessed included cytoplasmic and extranuclear membrane fractions (lanes 2-5) as well as purified nuclear fractions (lanes 7-10). The reaction with anti-MUC1-N shows the typical bands at about 200 kDa. Figure 3B shows the reactivity of the purified nuclear fractions (lanes 7-10) with a nuclear marker (lamin A).
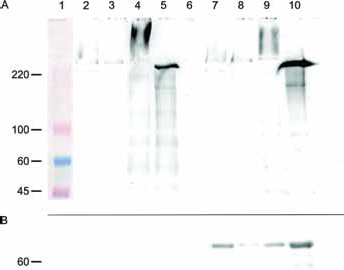
Western blot of subcellular fractions belonging to breast cancer cell lines (T47D, MDA-MB-231, MCF7, and ZR-75). A) Incubation with MAb HMFG1. Lane 1 shows the molecular weight standard; lanes 2-5 correspond to supernatants of nuclear pellets (cytoplasmic and membrane fraction), while lanes 7 to 10 show purified nuclear pellets. In lane 6 no sample was run to clearly separate the specimens. In lanes 2-4 and 7-10 the characteristic MUC1 bands at approximately 220 kDa are shown. Other bands as well as smear reactivity are also shown in lanes 2, 4, 5, 7, 9, and 10. B) Incubation with an antibody against lamin (nuclear marker); purified nuclear fractions (lanes 7-10) show a reaction at >60 kDa.
Eleven nuclear fractions derived from HNSCC as well as those belonging to 2 normal breast tissue samples were not reactive (data not shown).
Discussion
In previous studies we have studied MUC1 expression in breast (8, 21, 25, 26), head and neck (27, 28), and colon cancer samples (29, 30), and we found that some specimens showed nuclear reactivity with anti-MUC1-N MAbs. In the present study, using IHC and WB, we found that the anti-MUC1 extracellular domain MAb HMFG1 reacts at the nuclear level in these 3 tumor types, while BBD samples were also reactive. Some samples that showed nuclear positivity with IHC also showed reactivity when WB analysis was used; this indicates that the 2 techniques both provide information although they are not interchangeable.
In line with other reports (28, 31-33), when performing SDS-PAGE and WB analysis of subcellular fractions we obtained in some samples typical bands of about 200 kDa and several bands of lower molecular weight as well as smear reactivity at the stacking gel. As Lo-Guidice et al (31) pointed out, these smear reactions may be due to the presence of MUC1 with different grades of glycosylation, while the bands of low molecular weight could correspond to protein degradation.
Many immunohistochemical studies have been performed on normal epithelial and tumor tissues using well-characterized antibodies against the MUC1-N domain, which recognize epitopes within its VNTR region; although antibody reactivity often varies depending on the degree of O-linked glycosylation, MUC1 overexpression has been largely described in tumor samples (9, 34, 35). On normal epithelia, MUC1 VNTR is extensively O-glycosylated with long branched glycans, whereas on tumor cells it is markedly underglycosylated with simpler and shorter glycan chains affecting many functions of MUC1 (6, 7). The pattern of expression differs between normal and neoplastic cells, being restricted to the apical part of normal cells while tumor samples show a mixed, nonapical reaction (2, 9, 36, 37).
In the case of invasive breast cancer samples, we have found different percentages of reactivity depending on the MAb employed; HMFG1 was the most reactive MAb since almost 100% of specimens stained, although the MAbs HMFG2 and C595 also had high reactivity while SM3 was less reactive (8, 21, 25, 26). In colorectal cancer we found high expression of MUC1-N with C595 and HMFG1 (25, 29, 30), while the HNSCC MUC1-N expression percentages detected with these MAbs were lower (27, 28). These results are confirmed in the series considered in the present report, although the percentage of positivity varied, being 100% in breast cancer, 94% in colorectal cancer, and 40% in HNSCC.
Different mechanisms may be involved in tumor MUC1 overexpression, such as regulatory transcriptional events, epigenetic control (38), and gene amplification (39). Furthermore, the kinetics of membrane trafficking either in the de novo pathway or the endosomal recycling pathway may be altered: endocytosis and intracellular accumulation of full-length MUC1 without its degradation or cleavage in tumor cells have been reported (40, 41).
In general, immunohistochemical and immunocytochemical studies using anti-MUC1-N MAbs failed to detect any nuclear localization. Kumar et al (42) considered that this failure may be due to low magnification images, which preclude adequate assessment of nuclear staining, and also to lack of mention or discussion by the authors; however, in some cases nuclear staining is evident from photomicrographs such as in our own reports in breast cancer (8, 21, 37, 43), BBD (44), colon cancer (29), and larynx carcinoma (27) employing anti-MUC1 extracellular domain MAbs. On the other hand, in a study on the importance of MUC1 in the intestinal mucosal barrier to infection, McAuley et al (45) reported translocation of the MUC1 extracellular domain to the nucleus in Campylobacter jejuni-treated HeLa cells as an unexpected finding, and considered a possible role for p53 in MUC1 stabilization and recycling during stress.
Thirkill et al (46) observed MUC1-N MAb B27-29 nucleus-associated staining in normal macaque trophoblast cultures, while progesterone treatment of endometrial cancer cell lines changed their MUC1-N profile with nuclear reactivity (47). Kumar et al (42) consistently found nuclear MUC1-N in all cell types and tissues studied, which comprised the MCF7 cell line, 2 human trophoblast-derived choriocarcinoma cell lines (BeWo and Jar), the COS7.MUC1 cell line, primary cell cultures of macaque trophoblast and normal mammary epithelia (240LB, passage 5-6), and macaque colon sections. They reported intense MUC1-N nuclear staining in the MCF7 and Jar tumor cell lines; this nuclear expression was heterogeneous for MCF7 and primary cultures of normal human mammary epithelial cells. Employing another anti-MUC1-N antibody, DF3, they found weaker nuclear staining in MCF7 and BeWo cells and no nuclear staining in trophoblasts. Their findings demonstrated that MUC1-N was located in nuclear speckles (interchromatin granule clusters) and closely associated with the spliceosome protein U2AF65. Their results suggested that MUC1-N and MUC1-C have dissimilar intranuclear distribution patterns.
By chromatin immunoprecipitation and real-time PCR, Cascio et al (48) found that MUC1/22TR upregulated IL-6 and TNF-α expression by binding to their promoter regions in an NF-κB p65-dependent manner in both MUC1-transfected cells and human breast cancer cells that express endogenous MUC1. They proposed that tumor-specific short O-glycans or no glycans on the tandem repeats of the VNTR region reduce the size of the molecule and its 3-dimensional configuration, allowing its binding to transcriptional coactivators such as p65 and trafficking to the nucleus, although the specific MUC1 sequence that interacts with p65 was not identified. These authors considered that their findings are not expected to occur in normal epithelial cells, where MUC1 is strictly polarized to the luminal surface and long, branched oligosaccharides conceal the VNTR protein and increase the size of the molecule.
MUC1 protein sequence (NP_001191215.1) analysis using the cNLS Mapper resource (http://nls-mapper.iab.keio.ac.jp) (49, 50) identifies 2 predicted bipartite nuclear localization signals (NLSs), one located at the extracellular domain (MUC1-N: APKPATVVTGSGHASSTPGGEKETSATQRS) and the other located at the cytoplasmic tail (MUC1-C: RRKNYGQLDIFPARDTYHPMSEYPTYHTH). Among the MUC1 transcript isoforms described in the NCBI GenBank, 14 present NLSs both in the extracellular domain and cytoplasmic tail while the other isoforms only present NLSs at MUC1-C. In this sense, the MUC1-N NLSs are only encoded by the MUC1 isoforms with a full-length exon 2 sequence, while the MUC1 isoforms without MUC-N NLSs are characterized by a shorter exon 2 mRNA sequence. Interestingly, Ng et al (51) have reported a single-nucleotide polymorphism at the 5′ end of exon 2 (rs4072037) implicated in the control of the alternative splicing event of this exon sequence; it can lead to a 9-amino-acid insertion affecting the signal peptide region of both full-length MUC1 transcripts and those lacking the tandem repeat domain (51, 52). Such evidence raises the possibility that alternative splicing events involving the 5′ end of the exon 2 region could drive the translocation of the MUC1 extracellular domain to the nucleus by inclusion of an NLS.
The longer transcript was reported to be more abundant in cancer tissues (52-55) with variations of the different splice forms according to tumor location, although the overrepresentation of these transcripts may be affected by inflammation as well (51). These variations may also explain the differential MUC1-N detection at the tumor locations considered in our study: breast, colon, and head and neck.
Investigating the possible functions of nuclear MUC1-N localization, Kumar et al (42) found the MUC1 extracellular domain in nuclear speckles and proposed that the dependence of this localization on RNA suggests a possible role in the assembly and/or organization of these structures.
The present study was performed in human neoplastic tissues. It demonstrates that the MUC1 extracellular domain subunit is reactive at the nuclear level in primary breast, HNSCC, and colon tumors as well as BBD. BBD reactivity comprised entities with and without an increased risk of breast cancer development. We did not find reactivity in normal tissue samples.
The MUC1 extracellular domain protein core may be involved in transcriptional and posttranscriptional processes associated with stress conditions, although it should not be underestimated that it may account for normal phenomena as well. Thus, the complexity of the MUC1 molecule highlights new biological events and may mediate different functions depending on the context.
Footnotes
Abbreviation
Acknowledgments
We gratefully acknowledge the MAb HMFG1 kindly provided by Prof. Joyce Taylor Papadimitriou and Prof. Joy Burchell (King′s College, London, UK) and anti-lamin A provided by Prof. Ana Ves Losada (INIBIOLP, La Plata, Argentina).
We thank Dr. W. Servi, Dr. A. Barbera, Dr. F. Terrier, Dr. A. Cretón and Dr. A. Pereyra for their kind collaboration with patients′ samples, and Miss María Emilia Cermignani for English correction of the manuscript.
Financial support: This study was supported by the National University of La Plata, Argentina.
Conflict of interest: The authors declare that they have no financial relationship that may lead to a conflict of interest in relation to the submitted manuscript. Prof. María Virginia Croce, Dr. Marina Isla Larrain and Dr. Martín E. Rabassa are members of the research career of the Comisión de Investigaciones Científicas de la Provincia de Buenos Aires (CIC/PBA).
