Abstract
Background
Neoadjuvant chemotherapy has been increasingly utilized in the treatment of breast cancer patients. However, there are no established surrogate markers predicting the response to subsequent adjuvant therapy and clinical outcome of patients. In particular, whether primary or lymph nodes metastasis should be evaluated for these analyses has remained unknown. Therefore, in this study, we first evaluated the differences in biomarkers between primary and metastatic cancer tissues in the patients undergoing neoadjuvant chemotherapy. We then correlated the findings with the clinical outcomes of these patients.
Methods
We examined 49 patients receiving neoadjuvant chemotherapy and subsequent surgery with lymph node metastasis. Estrogen receptor (ER), progesterone receptor (PgR), human epidermal growth factor receptor 2 (HER2) and Ki-67 were all immunohistochemically evaluated in core needle biopsy samples from primary and metastatic tumors following chemotherapy.
Results
No statistically significant differences in these markers were detected between the primary tumor and metastatic lymph nodes following therapy, but the Ki-67 labeling index was significantly higher in metastatic lymph nodes than in primary tumor (p = 0.017). The patients associated with luminal A type carcinoma in their lymph nodes following chemotherapy demonstrated significantly better clinical outcomes (disease-free survival: p = 0.0045, overall survival: p = 0.0006) than those who were not.
Conclusion
These data indicate that subtype classification following chemotherapy, in the metastatic lymph nodes rather than primary tumor could predict long-term outcomes of patients undergoing neoadjuvant chemotherapy.
Keywords
Introduction
Estrogen receptor (ER), progesterone receptor (PgR), human epidermal growth factor receptor 2 (HER2) and Ki-67 status are being routinely studied as surrogate markers for systemic treatment (1). Epoch-making gene expression studies by Perou et al and Dawood et al identified 4 major breast cancer subtypes (2, 3), and Ki-67 values were also included in the subtype classification after the St. Gallen International Breast Cancer Conference of 2011 (4). In addition to these subtypes, tumor size, lymph node status, histological type, pathological grade and pTNM stage are still all considered important prognostic factors in selecting medical adjuvant therapy. Among these factors, the presence of metastatic lymph nodes is generally considered the most important, but the status of axillary lymph node dissection was reported to exert little effect on overall survival of the patients (5–7).
Systemic primary neoadjuvant chemotherapy (NAC) is usually administered to breast cancer patients with locally advanced tumors, allowing surgery in otherwise nonoperable breast cancer and increasing the success rate for breast-conservative surgery in the patients with large operable breast cancer (8, 9). Identification of the factors predicting the subsequent clinical outcome of these patients and types of adjuvant treatment following NAC has also become pivotal. Pathological complete response (pCR) of primary breast cancer has been reported to be associated with higher disease-free survival (DFS) and overall survival (OS) rates among those receiving NAC (8–9–14). Axillary lymph node pCR was also proposed to predict the long-term clinical outcome of patients despite the presence of a residual primary tumor (11), but little availability of pretreatment tissue from lymph nodes has made it difficult to assess the response rate in the specimens after treatment. Falk et al recently reported a comparison of the subtypes between primary and synchronous lymph nodes metastasis (15), but the comparison between primary and metastatic tumors following NAC and subsequent correlation of the findings with clinical outcomes of patients have not been reported. Therefore, in this study, we first evaluated the differences in biomarkers between primary and metastatic breast cancer in patients following NAC. We then correlated the findings with clinical outcomes for these patients to analyze the significance of subtype-based evaluation of primary and metastatic lesions following NAC.
Materials and Methods
Patients
Fifty-six cases were included of invasive ductal carcinoma in Japanese patients who underwent mastectomy and lymph node dissection between 2004 and 2010 at Tohoku University Hospital (Sendai, Japan). All of the patients had undergone NAC and were pathologically diagnosed with lymph node metastasis postoperatively. In 7 patients, insufficient metastatic lymph node tissues were available for examination after sectioning. Therefore, the primary tumors and the corresponding lymph node metastases of the remaining 49 patients were examined in this study. Among these 49 patients, 35 had corresponding core needle biopsy specimens obtained before NAC, available for this study. Clinicopathological features of the cases examined are summarized in Table I.
Clinicopathological factors of patients examined in this study

The patients ranged in age from 31 to 80 years (median 49 years) and had a median tumor diameter of 23 mm (2-80 mm). Table II shows the NAC regimen and total dose (mg) per body weight (kg) for each patient. Of the patients, 46/49 (94%) underwent chemotherapy with an anthracycline-based regimen, 19 of whom received anthracycline and taxane. Two patients (4%) received taxane alone as anthracycline was contraindicated due to a history of congestive heart failure and an ejection fraction of <60%. One patient (2%) received taxane and neoadjuvant trastuzumab because the patient refused anthracycline and had a HER2-positive tumor based on the evaluation of core needle biopsy. No patient underwent neoadjuvant endocrine therapy. There were no significant correlations between the types of NAC regimens (the inclusion or otherwise of taxane) and clinical outcomes for the patients.
Neoadjuvant chemotherapy (NAC) regimen for each patient, and total dose (mg)/body weight (kg)
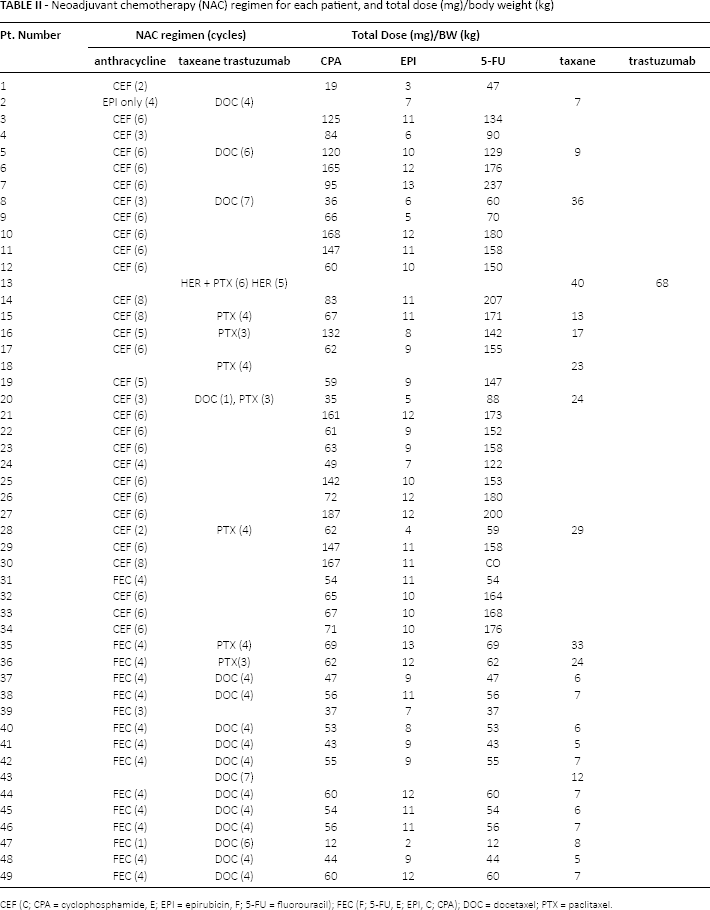
CEF (C; CPA = cyclophosphamide, E; EPI = epirubicin, F; 5-FU = fluorouracil); FEC (F; 5-FU, E; EPI, C; CPA); DOC = docetaxel; PTX = paclitaxel.
Immunohistochemistry
After evaluation of histopathological features, the primary tumor, metastatic lymph node tissue and core needle biopsy specimens were evaluated for ER, PgR and Ki-67 by immunohistochemistry (IHC) using standard techniques and for HER2 expression by both IHC and fluorescent in situ hybridization (FISH).
Tissue sections (4-μm thick) were deparaffinized, and endogenous peroxidase activity was blocked by immersing the slides in 0.3% hydrogen peroxidase for 30 minutes. Antigen retrieval was performed by heating the slides in an autoclave at 121°C for 5 minutes in citric acid buffer (2 mM citric acid and 9 mM trisodium citrate dehydrate, pH 6.0). The sections were then incubated with 10% normal rabbit serum or goat serum and were then incubated for 16 hours at 5°C with the primary antibodies. Dilutions of the primary antibodies were as follows: ER, 1:50; PgR, 1:50; HER2, 1:1,000; and Ki-67, 1:300. The sections were subsequently incubated with biotinylated rabbit antimouse IgG or goat anti-rabbit IgG (Histofine Kit; Nichirei, Tokyo, Japan) and subsequently with horseradish peroxidase-conjugated streptavidin (Nichirei). The antigen–antibody complex was then visualized with 3, 3′-diaminobenzidine (DAB) solution (1 mM DAB, 50 mM Tris-HCL buffer, pH 7.6, and 0.006% H2O2) and counterstained with hematoxylin.
The following antibodies were used: ERalpha (6F11; Leica Microsystems, Wetzlar, Germany), PgR (1A6; Chemicon International Inc., Temecula, CA, USA), HER2 and Ki-67 (K5204, MIB1; DakoCytomation Co. Ltd, Kyoto, Japan).
Analysis of Immunohistochemistry
ER and PgR immunoreactivity was detected in the nuclei of carcinoma cells. Results were evaluated according to the procedure of Allred et al (16); a score between 1 and 5 represented the percentage of carcinoma cells with positive immunoreactivity and was added to a score between 1 and 3, which represented relative immunointensity, to give a maximum total score of 8. A total score of ≥3 was categorized as ER- or PgR-positive.
HER2 immunoreactivity was scored in accordance with the American Society of Clinical Oncology (ASCO)/College of American Pathologists (CAP) 2007 guidelines (17). An algorithm defining positive, equivocal and negative values for HER2 protein expression and gene amplification was employed: a positive HER2 result was defined as an IHC staining score of 3+ (uniform, intense membrane staining of >30% of the invasive tumor cells), weak-to-moderate staining of the entire membrane in >10% of the tumor cells was considered equivocal (2+), and an IHC staining score of 0 or 1+ was considered negative. FISH analysis was performed for all HER2 IHC equivocal cases and was defined as positive if the HER2/CEP17 ratio was ≥2.2.
Ki-67 immunoreactivity was detected in the nuclei of the cells, and the labeling index (LI) was obtained. The fraction of proliferating cells was based on a count of at least 1,000 tumor cells at hot spots of the specimens, following evaluation in low-power fields, and the Ki-67 LI value was expressed as the percentage of positive cells in each case.
Classification of Subtypes
In this study, breast cancer subtypes were classified in accordance with the St. Gallen recommendation based on IHC analysis of ER, PgR, HER2 and Ki-67 (4). The following subtypes were defined: luminal-type (ER+ and/or PgR+), HER2 type (ER− and PgR−, and HER2+) and triple negative (TN; ER− and PgR−, and HER2−). The luminal-type was further subdivided into types A (HER2− and Ki-67 <14.0%) and B (HER2+ and/or Ki-67 ≥14%) (4). OS and DFS were calculated from the date of diagnosis.
Statistical Analysis
Statistical analyses of ER, PgR and HER2 immunoreactivity and of the tumor site were performed using the chi-square test. Spearman's rank test (bivariate Pearson's correlation analysis) and the Mann-Whitney test were used to compare the Ki-67 status. OS and DFS curves of the patients examined were generated according to the Kaplan-Meier method, and statistical significance was calculated using the log-rank test. All tests were 2-tailed, and p value ≤0.05 was considered significant. The statistical analyses were performed using JMP Pro software version 10.0.2 (SAS Institute, Cary, NC, USA).
Results
There were significant differences between clinical outcome and tumor grade of the patients (p = 0.02, Mann-Whitney test), but no significant differences were detected between other clinicopathological factors and clinical outcomes of the patients (age, p = 0.9; pTNM, p = 0.08; tumor size, p = 0.44; number of metastatic lymph nodes, p = 0.43; response evaluation, p = 0.44).
ER, PgR and HER2 Status
When ER, PgR and HER2 status were dichotomized as positive and negative, there were some discrepancies between the primary tumor and metastatic lymph nodes of the same patient detected after NAC (Tab. III). ER status was discordant in 8% (4/49; 1 patient lost ER positivity, and 3 patients gained positivity; p<0.0001, by Fisher's test, p = 0.32, by McNemar's exact test) and PgR status in 22% (11/49) of the patients (7 patients lost PgR positivity and 4 patients gained positivity; p = 0.06, by Fisher's test; p = 0.37, by McNemar's exact test). HER2 status was discordant in 6% (3/49) of the patients (1 patient lost HER2 positivity and 2 patients gained HER2 positivity; p<0.0001, by Fisher's test; p = 0.33, by McNemar's exact test).
Summary of ER, PgR and HER2 status in the primary tumor and the metastatic lymph node of the patients examined in this study
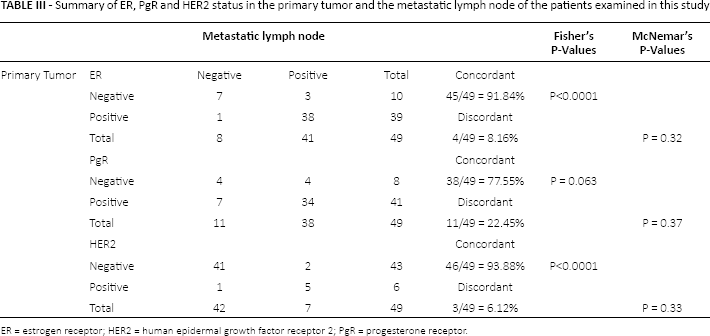
ER = estrogen receptor; HER2 = human epidermal growth factor receptor 2; PgR = progesterone receptor.
Ki-67 LI between the Primary Tumors and Lymph Node Metastasis Following NAC
There was a significant positive correlation between the Ki-67 LI of the primary tumor and metastatic lymph nodes in the same patient (Spearman's rank test p = 0.006). However, there was a significant difference between the Ki-67 LI of the primary tumor and metastatic lymph nodes (14.2% ± 2.2% vs. 18.1% ± 2.8%, respectively; p = 0.017, Mann-Whitney test). Table IV summarizes the correlation between the Ki-67 LI and clinical outcomes of the cases studied. The mean Ki-67 LI of the primary tumors of those with and without recurrence was 21.8% ± 3.1% and 8.4% ± 2.7%, respectively. No significant differences were detected between recurrence and the Ki-67 LI of the primary tumor (p = 0.0804, by Mann-Whitney test), but significant differences were noted between recurrence and the Ki-67 LI of the metastatic lymph node (p<0.0001, by Mann-Whitney test). In addition, there were significant differences in Ki67 LI of both primary and metastasis between the patients who survived and those did not (primary tumor p = 0.0203, lymph node metastases p = 0.0019; Mann-Whitney test).
Summary of ki-67 labeling index (%) and recurrence or survival of patients studied
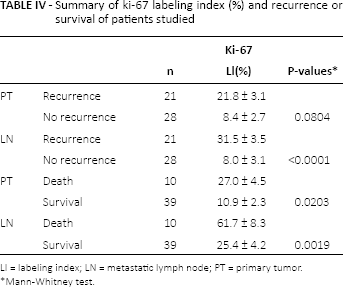
LI = labeling index; LN = metastatic lymph node; PT = primary tumor.
Mann-Whitney test.
Subtype Classification
Table V summarizes the subtype classifications of the primary and metastatic lymph tumors. The distribution of subtypes was as follows: luminal A 57% (28/49), luminal B 37% (18/49), HER2 2% (1/49) and TN 4% (2/49) for primary tumor and luminal A 53% (26/49), luminal B 41% (20/49), HER2 4% (2/49) and TN 2% (1/49) for metastatic tumors. No significant differences were detected between the primary tumor and metastatic lymph node subtypes. However, there were differences between primary and metastatic tumors in 18 patients.
Summary of results for subtypes of primary tumor and metastatic lymph nodes

Fisher's exact test: p = 0.0146; McNemar test: p = 0.9739.
HER2 = human epidermal growth factor receptor 2; TN = triple negative.
Correlations between Subtype Classifications and Clinical Outcome of Patients
OS and DFS curves of the patients are summarized in Figure 1. Statistical analyses were limited to the luminal A and B subtypes because few were categorized as TN or HER2 in this study. Luminal A subtype in metastatic nodes was significantly associated with better survival and favorable recurrence (log-rank test: OS p = 0.0006; DFS p = 0.0045) and that in primary tumors was also significantly associated with better survival of the patients, but no significant correlation was detected between subtypes and recurrence of the patients (log-rank test: OS, p = 0.0162; DFS, p = 0.557) (Fig. 2).
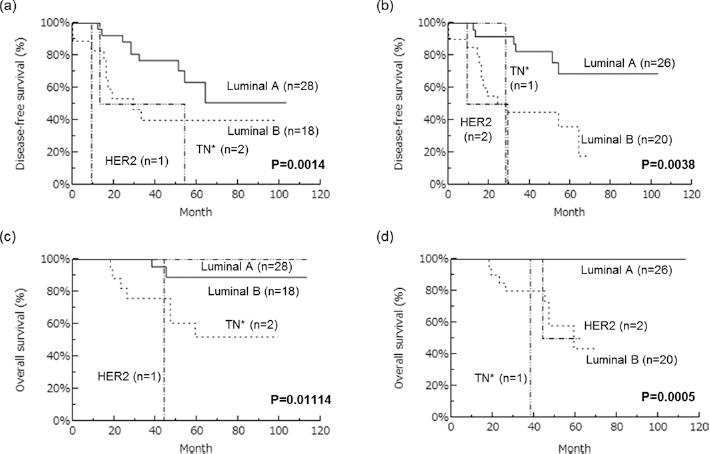
Disease-fee and overall survival (DFS and OS) curves of patients according to subtype classification by Kaplan-Meier method. a) Disease-free survival according to the subtypes of primary tumor. b) Disease-free survival according to the subtypes of metastatic lymph node. c) Overall survival according to the subtypes of primary tumor. d) Overall survival according to the subtypes of metastatic lymph node. HER2 = human epidermal growth factor receptor 2; TN = triple negative.
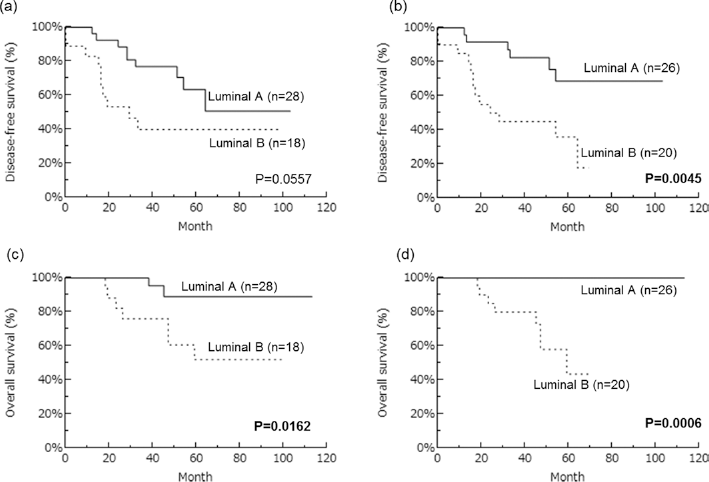
Disease-free and overall survival (DFS and OS) curves of patients with luminal A or luminal B subtype by Kaplan-Meier method. a) Disease-free survival in primary tumor. b) Disease-free survival in metastatic lymph node. c) Overall survival in primary tumor. d) Overall survival in metastatic lymph node. Statistical analysis was evaluated by the log-rank test; p values <0.05 were considered significant and are shown in boldface. HER2 = human epidermal growth factor receptor 2; TN = triple negative.
Discussion
Postoperative or adjuvant systemic chemotherapy for breast cancer patients has come to be more frequently based on the biomarker profiles of the primary tumor – i.e., the expression of ER, PgR and HER2. However, it is also true that the major targets of those systemic therapies are carcinoma cells in metastasis including micrometastasis (18). Usually classification of breast cancer, including newly defined subtypes, has been performed in primary tumor, although many studies have reported a significant correlation between the primary and metastatic lesions (18–22).
The process of breast cancer progression to metastasis has recently been evaluated based upon the epithelial–mesenchymal transition (EMT) and mesenchymal–epithelial transition (MET) concepts, and EMT is generally considered one of the main mechanisms in this process (23–25). In EMT, epithelial cells acquire their invasive potential by becoming less cohesive mesenchymal-like cells, increasing their invasive properties and subsequently gaining access to vascular systems. These circulating tumor cells can then reach and invade distant organs and form metastatic lesions. MET is involved in the last process to maintain the metastatic lesions (26).
Breast cancers are heterogeneous tumors with many different subclones, and intratumoral heterogeneity has been well known in both primary and metastatic tumors (27–29). Therefore, in this study, we compared the status of surrogate markers between primary and metastatic tumors among patients who had undergone NAC. Results for ER, PgR and HER2 in our present study were similar to those for 4 other studies done in patients without NAC, which demonstrated that the difference between primary tumor and metastatic lymph node was 3%-7% (ER), 10%-16% (PgR) and 2%-10% (HER2), respectively (19–22). It is therefore important to note that even after chemotherapy, ER, PgR and HER2 status of the primary and metastatic tumors was almost identical, but complete concordance was not detected in all patients, which was suggestive of discordant responses to the chemotherapy between these 2 different groups of carcinoma cells in the same patients. However, despite these differences, NAC exerted little effect on the expression patterns of biomarkers in carcinoma cells between the primary tumor and the metastatic lymph nodes. In addition, discordant ER, PgR and HER2 status of the primary tumor between paired core needle biopsy specimens and surgical specimens after NAC was 13.9%, 33.4% and 8.1%, respectively. These differences did not reach statistical significance, which is consistent with results of other reported studies (30).
In our present study, Ki-67 LI values were significantly different between primary and metastatic lymph nodes using the Mann-Whitney test (p = 0.017) – i.e., the Ki-67 LI of the metastatic lymph nodes was significantly higher than that of the primary tumor. This finding suggests that carcinoma cells with greater proliferation potential are present in the metastatic lymph nodes as opposed to the primary tumor. Of interest, no significant differences in Ki67 LI were reported between primary and metastatic tumors in the patients not receiving NAC (15, 19, 22, 31). This discrepancy may be the influence of NAC on the proliferation potential of tumor cells, but this awaits further investigation for clarification.
There is controversy with regard to the clinical significance of pCR as a predictive marker of the eventual effectiveness of NAC (8–14, 32). Ki-67 has been used as a surrogate marker for clinical response and a prognostic factor after NAC (33–36), but its significance has also remained controversial. For instance, the Ki-67 LI value before NAC was reported as a significant predictive factor of the effectiveness of NAC and the degrees of its decrement were reported to be correlated with clinical response and DFS of patients (34, 35). However, Matsubara et al reported that the changes of Ki-67 LI in carcinoma cells between pre- and post-NAC were by no means a prognostic factor in the luminal A patients (33). In this study, the eventual clinical outcome of the patients was significantly different among those yielding different Ki67 LI before and after the chemotherapy. The mean Ki-67 LI value among the patients with and without recurrence was increased by 9.2% ± 4.4% and decreased by 8.6% ± 4.4%, respectively, differences that were statistically significant (p = 0.023, by Mann-Whitney test). These results were all consistent with those in previously reported studies mentioned above (34, 36).
The pCR of a metastatic lymph node was reported to be significantly associated with a better prognosis, even in the presence of a residual primary tumor (11). Numerous studies have investigated the differences in the biomarker profiles of primary tumors and metastatic lymph nodes among patients receiving NAC, but few have evaluated their prognostic potential in metastatic lymph nodes. We therefore determined whether biomarker status in metastatic lymph nodes following NAC could predict eventual clinical outcomes of patients better than that in primary tumor. The patients with recurrence had significantly higher Ki67 LI in metastatic lymph nodes than those who did not, a difference that was, however, not detected in primary tumor. These findings did indicate that the Ki-67 LI status of metastatic lymph nodes is more significantly correlated with clinical outcomes of patients receiving NAC than that of the primary tumor.
A previous prognostic analysis also compared subtype classifications of primary tumors and metastatic lymph nodes in patients without NAC, and there were no significant differences (15). Therefore, it was usually concluded that subtypes could usually be maintained in the process of metastasis in the great majority of breast cancer patients (15).
However, it is also true that patients with luminal A in the primary tumor and with a different subtype in a lymph node metastasis were associated with a significantly adverse clinical outcome (15). In our present study, luminal A metastatic lymph nodes were significantly associated with better survival and favorable recurrence among the patients examined in this study (log-rank test: OS, p = 0.0003; DFS, p = 0.0071). However, when studying the correlation of the subtypes with clinical outcomes of the patients in their primary tumor, luminal A subtypes were also significantly associated with better survival, but no significant correlation between subtype and recurrence rate was noted (log-rank test: OS, p = 0.0162; DFS, p = 0.557). Of interest, the number of patients studied was rather limited in our present study, but none of the patients who died had luminal A metastatic lymph nodes.
Among the patients who had discrepant subtypes between primary and metastatic tumors, the patients with luminal A in primary tumor tended to have a worse prognosis, whereas those with luminal A in metastatic lymph node had a better prognosis (Fig. 3). These findings were also consistent with results of previous studies of patients without treatment (15). Patients with luminal A classifications are generally considered to have a good prognosis. However, in our present study, this was only applicable to the metastatic lymph nodes. Therefore, lymph node biomarker profiles could provide better prognostic indicators than primary tumors following NAC. Intratumoral heterogeneity may account for the differences between primary and metastatic lesions as described above. In our present study, significant differences in DFS according to metastatic lymph node subtype but not the primary one were detected. This finding suggests that some clones are present in high-grade and low-grade forms in metastatic lymph nodes before NAC and that only high-grade clones survive in the metastatic sites after NAC, but this awaits further investigation for clarification.
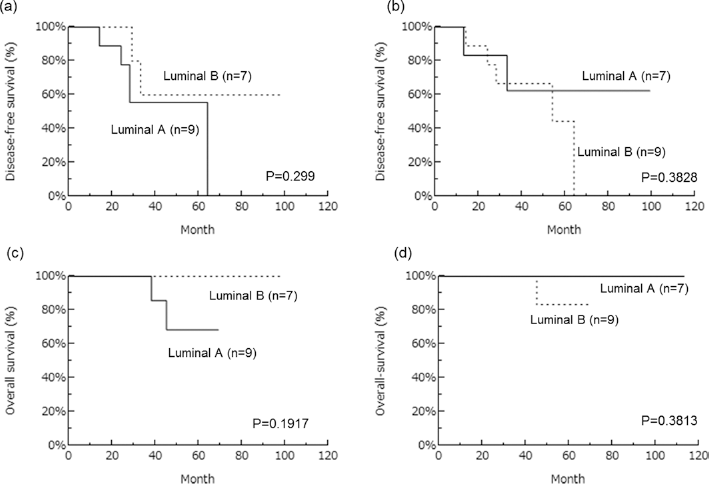
Disease-free and overall survival (DFS and OS) curves of the patients who had different subtypes between the primary and metastatic tumors by Kaplan-Meier method. a) Disease-free survival in primary tumor. b) Disease-free survival in metastatic lymph node. c) Overall survival in primary tumor. d) Overall survival in metastatic lymph node. There were no significant differences between clinical outcome and subtypes.
At present, there has been no established clinical strategy for postoperative patients who have undergone NAC and been diagnosed with lymph node metastases postoperatively. However, the roles of tumor biomarkers and of subtype classifications in synchronous lymph node metastases have remained undefined. Evaluation of their biomarker profiles by immunostaining is comparatively easy. Our findings indicated that the metastatic lymph node subtype of patients who have undergone NAC is an effective prognostic indicator. Further stratification of patients after surgery may thus extend treatment options.
Footnotes
Acknowledgements
We appreciate the skillful scientific assistance of Ms. Yayoi Takahashi, Ms. Erin Iwabuchi and Ms. Syuko Hata, MT (Department of Pathology, Tohoku University Hospital).
Financial support: This work was supported by JSPS KAKENHI Grant Number 26460639.
Conflict of interest: We declare that there is no conflict of interest that could be perceived as prejudicing the impartiality of the research reported.
