Abstract
Thyroid malfunction is more common in individuals with Down syndrome (DS) than in the general population. It has been hypothesized that thyroid may influence cancer risk. Individuals with DS are at greater risk of developing leukemia than the general population, while solid tumors especially breast cancer (BC) are rare. BC patients have higher levels of circulating thyroid-stimulating hormone (TSH) and prolactin (PRL), both regulated by the thyrotropin-releasing hormone (TRH), a hypothalamic tripeptide. This study was aimed at investigating the status of TRH functional polymorphisms in subjects with DS and BC. Unrelated families with DS probands (n=180), individuals with BC (n=99) and ethnically matched controls (n=216) were recruited. Genomic DNA isolated from peripheral blood was subjected to PCR amplification followed by DNA sequence analysis. Data obtained were analyzed by population- and family-based statistical analysis. Among 30 studied sites, only 2 (rs7645772 and rs13097335) were polymorphic. Case-control analysis showed a lack of any significant association with DS, while the rs13097335 GG and GT genotype frequency was significantly different in the BC samples. A paternal-biased transmission of the G allele was observed in female DS probands. It may be concluded that rs13097335 may have a protective role toward the development of BC.
Introduction
Normal thyroid function is essential for brain development as well as physical growth. Function of the thyroid gland is monitored by 2 principal molecules, thyrotropin-releasing hormone (TRH) and thyroid-stimulating hormone (TSH), while tetraiodothyronine and triiodothyronine are released by the thyroid gland to regulate functions such as intelligence, emotional status, behavior and cognition governed by the central nervous system (1). Thyroid also regulates neural activity and the rate of metabolic reactions. Further, thyroid hormone promotes growth hormone actions, nervous system development, target cell responsiveness to catecholamines etc. (2).
Association between thyroid dysfunction and Down syndrome (DS) is a well-established fact. A significant link has been described between DS and autoimmune thyroid disease, usually hypothyroidism (3). Mild elevation in plasma TSH is extremely prevalent, with a prevalence rate in individuals with DS of 80%-90% in early infancy and 30%-50% thereafter (4). Thyroid dysfunction in DS neonates may affect the already compromised brain development and somatic growth of children with DS.
Due to trisomy of the human chromosome 21 in DS, genetic overdosage of a number of genes is a distinct possibility. Change in the euploid gene expressions is also speculated to contribute to DS-related abnormalities (5). Intellectual disability, immunological imbalance, hormonal alteration, congenital heart disease, early onset of Alzheimer's-like diseases, childhood acute leukemia etc. are very common in individuals with DS (6).
DS patients are prone to acute leukemia, while solid tumors, especially breast cancer (BC), are rare (7). Reports on cancer incidence and death rates in subjects with DS have clearly shown a strikingly lower rate of breast neoplasms in comparison with non-DS subjects (7). Considering the substantial increase in life expectancy of the DS population over the past 5 decades, breast neoplasms are estimated to be 10-25 times less frequent in subjects with DS than in the general population (8).
There has been a lot of discussion over the possible existence of a relationship between thyroid dysfunction and BC. Studies have shown that patients with BC have altered thyroid functions and increased serum levels of TSH (9). Recent epidemiological studies have also implicated a role for prolactin (PRL) in conferring a risk of BC; elevated circulating PRL is associated with a higher risk of development of tumors that express estrogen receptors (10). TRH promotes release of TSH as well as PRL. In humans, TRH stimulates PRL release by a direct action on the pituitary gland (11). TRH, the first identified hypothalamic releasing factor, is a tripeptide synthesized by the hypothalamic paraventricular nucleus neurons (12). TRH functions as a neurotransmitter/modulator in the central and peripheral nervous system, but its major function is in the maintenance of thyroid hormone homeostasis via regulation of TSH biosynthesis and secretion (13).
As an important factor in the regulation of PRL and TSH, alteration in TRH has a potency to significantly affect disease pathophysiology. Studying functional single nucleotide polymorphisms (fSNPs) in candidate genes is important to find out their association with disease phenotypes. The TRH gene is located at human chromosome subband 3q13.3-q21. The first large transcript, prepro-TRH, containing multiple copies of TRH progenitor sequence, is enzymatically cleaved to produce TRH (13). The transcriptional unit is 3.3 kb in size and contains 3 exons interrupted by 2 introns of approximately 1,050 and 650 bp, respectively. Exon1 encodes the 5′ untranslated region of the mRNA, exon2 the putative signal sequence and the initial portion of propeptide, exon3 encodes for the remainder of the propeptide, which contains 6 copies of the TRH sequence. The predicted human preproTRH peptide structure has 242 amino acids (14). In the present study, fSNPs present in the intron2 and in a portion of exon3 of the TRH gene were analyzed in a group of eastern Indian individuals diagnosed with DS and BC to understand their role in the disease etiology.
Materials and Methods
Functional assessment of SNPs
Risk conferred by the fSNPs studied was analyzed computationally by FastSNP (http://fastsnp.ibms.sinica.edu.tw) and F-SNP (http://compbio.cs.queensu.ca/F-SNP/). Predicted function of the fSNPs was collected from SNPnexus (http://snp-nexus.org/index.html).
Subjects recruited
Unrelated nuclear families (N=180) with DS probands (106 complete parent-offspring trios, 61 duos – 54 without father and 7 without mother – and 13 single proband with DS) were recruited from the outpatient department of Manovikas Kendra, Kolkata, based on diagnoses according to the criteria of the Diagnostic and Statistical Manual of Mental Disorders, 4th Edition (15). Age range of the probands was 8 months to 27 years (mean ± SE: 7.7 ± 0.51). All of the DS cases recruited were confirmed for trisomy of the 21st chromosome by karyotyping. Adjacent normal tissues from malignant BC patients (n=99) were collected from the Chittaranjan National Cancer Research Institute, Kolkata. Ethnically matched healthy control individuals (n=216) (mean age 8.9 ± 0.7) were also recruited. All of the individuals were engaged for the study after obtaining informed written consent for participation. The study protocol was approved by the institutional human ethics committee of Manovikas Kendra.
Genomic DNA isolation and genotyping
Peripheral blood samples were obtained from participating families with DS probands and controls in the study. Genomic DNA was isolated from peripheral blood of the affected subjects, their parents and healthy controls, using standard techniques (16). Genomic DNA from postoperative normal tissue, adjacent to malignant BC tissue, was isolated following the same protocol.
A 601-bp sequence of the TRH gene including 30 fSNPs was amplified by PCR using the forward primer 5′-TGAGGCTCCCAGAGATCAAT-3′ and reverse primer 5′-TTTCAGGCATCAGGTCTTCC-3′. PCR amplicons were subjected to DNA sequence analysis in an ABI prism 3130 Genetic Analyzer (Applied Biosystems, Hitachi, Tokyo, Japan) using a Big Dye sequencing kit version 3.1 followed by analysis using Sequence analysis software version 5.2 (Applied Biosystems, California, USA).
Statistical analysis of genotype data
Difference in allelic and genotypic frequencies for the fSNPs studied in different study groups were compared with controls using a simple r x c contingency table (http://www.physics.csbsju.edu/stats/contingency_NROW_NCOLUMN_form.html). Minor allele frequency (MAF) of the studied fSNPs was also compared with that of 4 major populations reported in the Ensembl 1000 genomes project, Phase 1 (Population from Utah with ancestry from western and northern Europe [CEPH], Han Chinese from Beijing, China [HCB], Japanese from Tokyo, Japan [JPT] and Yoruba from Ibadan, Nigeria [YRI]). An odds ratio calculator (http://www.hutchon.net/ConfidORnulhypo.htm) was used for measuring allelic odds ratios. The transmission pattern of the risk allele from parent to probands with DS was analyzed by the Unphased program (version 2.404) (17). Power of all chi-square tests was calculated by Piface (18).
Results
Details for the fSNPs studied are given in Table I. Among 30 fSNPs analyzed, only 2, rs7645772 and rs13097335, were found to be polymorphic in the studied eastern Indian population (IND). All statistical analysis was executed for these 2 fSNPs only. Genotype frequencies for both the fSNPs were in Hardy-Weinberg equilibrium for all groups, except for rs13097335 in the father of probands with DS (p=0.022).
Details Of Fsnps Studied In The Trh Gene
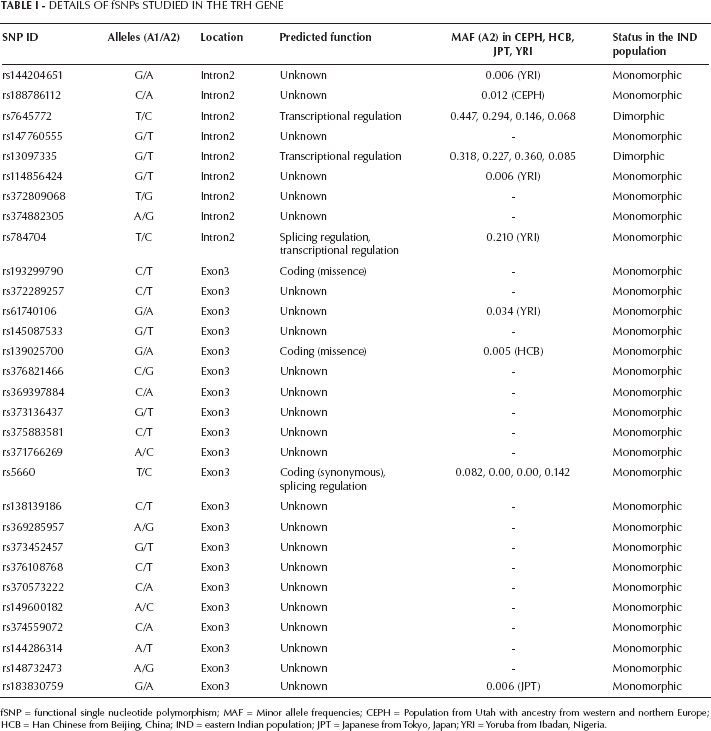
fSNP = functional single nucleotide polymorphism; MAF = Minor allele frequencies; CEPH = Population from Utah with ancestry from western and northern Europe; HCB = Han Chinese from Beijing, China; IND = eastern Indian population; JPT = Japanese from Tokyo, Japan; YRI = Yoruba from Ibadan, Nigeria.
Comparative analysis of MAF of the studied IND population versus that of different populations reported in the 1000 genome phase 1 revealed no significant difference with the CEPH and HCB for both fSNPs; rs7645772 MAF showed significant difference with both JPT and YRI populations (p=0.005 and p=0.0001, respectively), while rs13097335 significantly differed from the YRI population only (p=0.0001) (Tab. II).
Comparative Analysis Of Allelic Frequencies In Control And Populations Reported In The 1000 Genomes Project
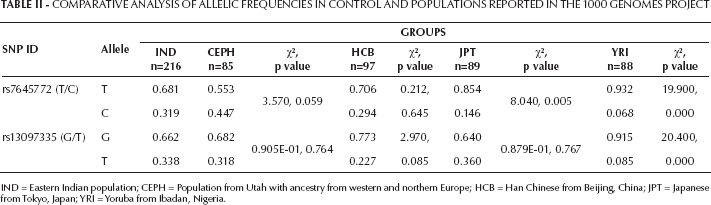
IND = Eastern Indian population; CEPH = Population from Utah with ancestry from western and northern Europe; HCB = Han Chinese from Beijing, China; JPT = Japanese from Tokyo, Japan; YRI = Yoruba from Ibadan, Nigeria.
Case-control analysis failed to show any significant difference in allelic frequency for either of the fSNPs (Tab. III). No significant differences were found in genotypic frequency in probands with DS and their parents when compared with control (Tab. IV).
Allelic Frequencies In Ind Control Individuals, Families With Ds Probands And Subjects With Bc
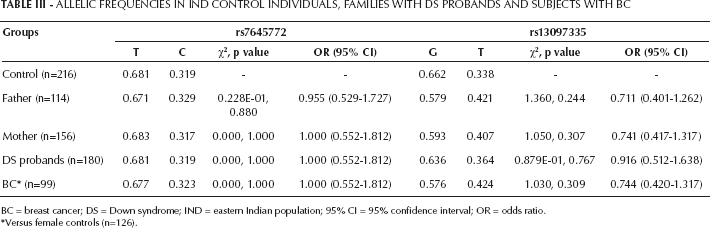
BC = breast cancer; DS = Down syndrome; IND = eastern Indian population; 95% CI = 95% confidence interval; OR = odds ratio.
Versus female controls (n=126).
Genotypic Frequencies In The Control Individuals, Families With Ds Probands And Subjects With Bc
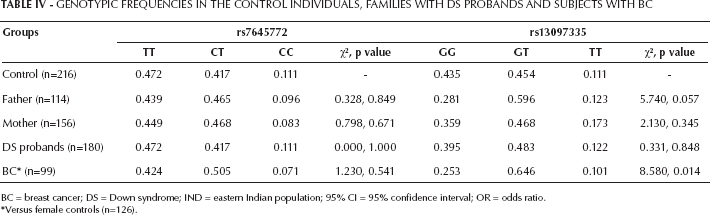
BC = breast cancer; DS = Down syndrome; IND = eastern Indian population; 95% CI = 95% confidence interval; OR = odds ratio.
Versus female controls (n=126).
No significant difference in allelic frequency was noticed either for BC samples for both fSNPs when compared with female controls (Tab. III). But a significant difference in genotypic frequency for rs13097335 (χ2=8.58, p=0.014) was observed in BC samples when compared with female controls (Tab. IV).
The Family-based Transmission Disequilibrium Test [TDT] (Supplementary Table - TDT analysis in unrelated nuclear Families with DS probands. Available online at www.biological-markers.com) showed a biased transmission of rs13097335 G allele from both parents to the DS probands (likelihood ratio statistic [LRS] = 5.183, p=0.023). Further analysis showed that this significantly biased transmission of the G allele to probands is paternal in nature (LRS=8.878, p=0.003) and more specific for the female probands (LRS=6.738, p=0.009). On the other hand, rs7645772 failed to show any bias in transmission from the parents to probands.
Discussion
The present study was aimed at identifying possible involvement of TRH fSNPs with DS and BC. Among the 30 fSNPs of TRH studied in the IND population, only 2 SNPs (rs7645772 and rs13097335), both situated in the intron2, were dimorphic. Intronic SNPs are important regulators for transcription and can modify the splicing pattern of a gene thus likely having profound effects on the phenotype with relevance for disease risk. Both rs7645772 and rs13097335, with a splice distance of 169 and 102 respectively, showed a predicted function in transcriptional regulation with an FS score of 0.101 (F-SNP).
No remarkable change in allelic and genotypic frequencies for rs7645772 was observed in the present study for samples from unrelated families with DS probands and BC as compared with controls. Thus this site may not have any significant contribution in the etiology of DS as well as BC in this population.
Although there was a mild decrease in the G allele and GG genotype of rs13097335 in the parents and DS probands as compared with the controls, we could not find any statistically significant association. However, in the case of BC, rs13097335 genotypic frequency significantly differed due to a decrease in the GG genotype with a concomitant increase in the GT genotype in comparison with the female controls (χ2=8.58, p=0.014, power = 74.94%). A statistically insignificant decrease in rs13097335 G allele was also observed in the BC samples. TDT analysis showed a bias in transmission of the G allele from father to probands with DS (LRS=8.878), and more specifically to the daughters with DS (LRS=6.738). It may be concluded from these data that the GG genotype and G allele, transmitted preferentially to female DS probands and encoding for a regular TRH gene, may confer protection for BC. It has been reported earlier that only 25% of fetuses with milder developmental phenotype are born alive with DS, while about 75% of trisomy 21 conceptions are lost in utero (reviewed in (19)). On the basis of the current study, we may speculate that the higher proportion of fetuses aborted in utero may harbor the GT/TT genotypes and are removed to confer some advantage. However, it would also be intriguing to find out why this may happen. This preliminary investigation of the TRH gene for the first time showed an association with DS, and further work involving comparative expression analysis may give us deeper insights on the subject.
Individuals with DS have a decreased risk of solid tumors, while BC, nephroblastoma, neuroblastoma and medulloblastoma are very rare (7). Presence of 3 copies of chromosome 21 in DS reduces the incidence of solid tumors (19). In transgenic mice, a modest increase in expression of Down syndrome candidate region-1 gene conferred significant suppression of tumor growth by reducing angiogenesis, a step crucial for vascularization (20). Protection from BC in DS individuals, the most common malignant neoplasm in women, thus may be conferred by the genes, especially those acting as tumor suppressor, located at chromosome 21 (19). Additionally, based on the present study, it can be hypothesized that the rs13097335 G allele, showing paternal overtransmission in female DS probands and present at significantly lower frequencies in individuals with BC, may have a role in this “paradox.” Further study is warranted to confirm whether rs13097335 G allele has a protective role in the development of BC in DS.
List of Abbreviations
breast cancer
Population from Utah with ancestry from western and northern Europe
Down syndrome
functional single nucleotide polymorphism
Han Chinese from Beijing, China
eastern Indian population
Japanese from Tokyo, Japan
Likelihood ratio statistics
minor allele frequency
prolactin
Transmission disequilibrium test
thyrotropin-releasing hormone
thyroid-stimulating hormone
Yoruba from Ibadan, Nigeria
