Abstract
Angiogenesis, a hallmark of cancer, has been studied to be a potential marker for diagnosis, prognosis and therapy in breast cancer. To evaluate tumor angiogenesis, histological assessment has been a common approach and counting tumor microvessels after visualizing them by immunohistochemistry has been in use for a long time. With recent advances in digital pathology and image analysis, other characteristics of tumor vasculature can also be evaluated. In this article we briefly review the potentials of image analysis in assessing tumor microvessel morphologically that might be helpful in defining a better angiogenesis marker than other common markers like vessel count.
Rapidly dividing malignant cells are in need not only of oxygen and nutrients to fuel the engine for tumor growth and dissemination, but also of a route for carbon dioxide and metabolic waste to be disposed. This cannot be achieved without forming new blood vessels, a phenomenon called angiogenesis, which has been accepted to be one of the hallmarks of cancer (1). However the tumor vascular network is aberrant and there is no hierarchy of arteriole-capillary-venule as seen in normal tissues (2). Angiogenesis takes place when the balance between pro and anti-angiogenic factors is shifted toward pro-angiogenic factors. Breast cancer is among those malignancies with a growth that is evidently angiogenesis-dependent (3). Therefore, angiogenesis has been the focus of research for many years, and clinical investigators have approached angiogenesis in breast cancer from different aspects (e.g., diagnostic, prognostic, and therapeutic). Serological as well as histological expression levels of angiogenic growth factors, such as the vascular endothelial growth factor (VEGF) family and their receptors (VEGFRs), have been shown to have, to some extent, a prognostic value in breast cancer (4). The expression level of angiogenic factors can only be a surrogate marker for tumor vascularity (5). In order to estimate the degree of tumor angiogenesis as a technically easy-to-evaluate surrogate marker, histological examination of the tumor microvasculature has been practiced since long ago (6). The use of antibodies against endothelial markers, such as CD31, CD34, and CD105, for immunohistochemistry (IHC), allows to examine the distribution pattern of the microvessels in different sections of the tumor. The microvessel density (MVD) determined by this method has been shown to be strongly correlated with the expression of angiogenic factors in blood and tumor tissue of patients with breast cancer (7). For measuring MVD the researcher normally selects 3 to 5 different highly vascular areas (hot spots) under a low magnification, and then counts the number of spots stained for a specific endothelial marker under a 200X magnification. Some investigators suggested that the Chalkley method, employing a grid with 25 points on hot spots, should be used as the standard method rather than the observer-dependent quantification method (8). Yet, MVD quantified with either of these methods can only reflect the number of structures (consisting of endothelial cells) present in a given area, but fails to provide any information on how these microvessels functioned in vivo. To the best of our knowledge, the actual imaging modalities, such as dynamic contrast enhanced magnetic resonance imaging (DCE-MRI), that are being used in daily clinical practice, at their best can extract vessels with a few-hundred micrometers diameter in breast cancer (9). Other imaging modalities, such as diffuse optical spectroscopic imaging (DOSI), can distinguish the well-oxygenated areas from the hypoxic areas (10). Although new imaging techniques (for instance intravital microscopy) have much improved the visualization of small-sized vascular networks in fields like ophthalmology, in oncology the use of these advanced technologies is still limited to animal models (11). Microtomography or microCT, as well as casting the vascular network by injecting Microfil, or even serial sectioning of the whole tumor and IHC staining for endothelial markers, all techniques that have produced much information in animal models, are of limited use in routine clinical practice. Therefore, at this point of time in the clinical setting it is inevitable to use ex vivo histological data of tumor microvasculature in conjunction with imaging methods in order to simulate tumor vasculature architecture and computationally model the vascular network. As noted before, most of the tumor microvessels are structurally and functionally abnormal and can contribute to higher intratumoral pressure due to their leakiness. Consequently, these newly formed microvessels can collapse or get less perfused under the high pressure of the surrounding solid tissue, as they are immature and lack the supporting mural cells. Maturation of newly shaped microvessels is a multistep procedure and is functionally characterized by the recruitment of pericytes that cover the endothelial tube, control endothelial cell proliferation, and change their phenotype from activated to quiescent (12). Normalization of tumor vasculature, which is believed to be the mechanism of efficacy of some antiangiogenic therapies, is a result of pruning immature vessels that are structurally and functionally aberrant and lack pericytes. Normalized vessels are defined as vessel with less dilation, better morphology, and more coverage by pericytes and basement membrane (13). Some characteristics of the microvessels visualized by IHC on routine 4-µm thick sections might be helpful in theoretically assuming whether a given microvessel had a normal function in vivo or not.
Presence or absence of lumen. As discussed earlier, immature microvessels are more likely to collapse. Also, tissue processing and fixation might result in shrinkage of lumen. However, in order not to mistake a longitudinal section of a vessel wall with a microvessel without lumen, other morphological characteristics of the structure, such as the number of endothelial cells forming the structure and their alignment, should be taken into consideration.
Lumen shape. It is known that mature microvessels are more likely to have a regular geometry, while immature microvessels have elliptical-like lumen shapes. Here again, attention should be paid to the morphology of endothelial cells in order not to mistake a diagonally sectioned microvessel with an immature one.
Presence or absence of pericytes. Pericytes are known to play an important role in microvessel maturation and stabilization through cell-to-cell interaction with endothelial cells and paracrine effects. Routine IHC methods allow 2 or at most 3 markers to be stained simultaneously on a single histological section. When double-stained for pericytes and endothelial cells at the same time, the microvessels with or without mural cells can be differentiated. This method also provides the possibility to evaluate whether the pericytes are in close contact with the endothelial cells or not.
Presence or absence of red blood cells (RBCs). Although the absence of RBCs cannot rule out the possibility of perfusion in vivo, due to tissue manipulation after excision, their presence can be, at least to some extent, a strong evidence for selecting the perfused microvessels.
Distribution of microvessels in relation to hypoxia markers. Hypoxia is recognized to initiate key regulatory mechanisms of angiogenesis in cancer. Hypoxia markers, such as hypoxia-inducible factor 1 alpha (HIF-1α) or carbonic anhydrase IX (CA IX), have been studied to find the prognostic and predictive values of hypoxia in breast cancer. Simultaneous analysis of these markers, together with the microvessel distribution pattern, can help to better estimate the maturity level of the vascular network adjacent to the hypoxic area.
Digital pathology or computer-assisted image analysis, even if not accepted by many pathologists as diagnostic tools, have been well integrated in research. Virtual (or digital) slides are created from classical glass histology slides using scanners. Image analysis systems can then be used to modify the chromogen color used for IHC into a pseudocolor. This technique provides the opportunity to use serial 4-µm sections, using one section for each marker and staining all sections with the same chromogen (usually DAB), thus allowing to create a single slide after overlaying the serial virtual slides (Fig. 1). This helps to overcome the technical difficulty of obtaining effective stainings by double or triple IHC staining. Digital slides can then be modified, analyzed in part or as a whole, with image analysis software such as ImageJ (http://rsb.info.nih.gov/ij/) or Image Pro-Plus (Medical Cybernetics, USA). Stained structures are selected, and hue, saturation, and intensity of staining are optimized so that most of the stained structures in the slide, with the least nonspecific staining, are highlighted. These software can calculate the morphological characteristics of these structures, such as perimeter, diameter, and surface area of each structure. Roundness of vessel lumen can also be calculated with the following equation (14):
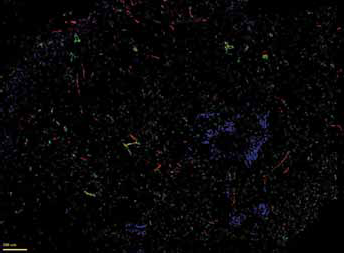
An overlay of pseudocolored CD31 (red), smooth muscle actin (SMA as pericyte marker, green) and CA IX (blue) on serial 4-µm histological sections. Scale bar = 100 µm.
Roundness of lumen = perimeter2/ (4π × area)
If calculated with the above equation, the roundness of a perfect circle is equal to 1 and that of a line is equal to infinity.
After determining these properties for all cross-sectioned microvessels, the distribution of these structures in any given area can be mapped based on selected criteria such as lumen shape or lumen surface area.
Several computational and mathematical methods, such as fractal morphometry, have been described for modeling tumor angiogenesis (15). It is beyond the scope of this article to discuss these modeling approaches; however, we would like to point out that some of the information that can be obtained by digital pathology and image analysis might be useful in computational modeling of tumor angiogenesis. Hypothetically, in normal tissue, blood supply is distributed homogenously so that all cells have access to oxygen and nutrients. Some have proposed that the total vessel lumen area (TVLA) per tissue area is representative of this distribution (16); however, gas and nutrient exchange takes place through capillary walls, and thus this parameter can be translated as total vessel perimeter (TVP). After virtual slides are created, the region of interest (ROI) or the whole slide can be segmented to smaller-size boxes (e.g. 500 µm2 equivalent to a 200X magnification area under light microscopy or 100 µm2, 100 µm being the maximum distance cells can survive away from blood supplying oxygen). Any given parameter, such as MVD, TVLA, or TVP (per box) can be calculated in each box and the whole slide can get mapped based on these box data. Figure 2 shows a simplified representation of 2 same-size tissue area boxes. In this example, by conventional quantification and MVD score, A results to be less angiogenic; however, based on TVLA both areas have the same blood supply. TVP in B is twice as that in A, which might mean twice more oxygen accessibility in A. Image analysis makes it possible to calculate all these parameters simultaneously, and this provides researchers with more options to test and try which of these are the most clinically relevant parameters and which can be a fair representative of the angiogenic profile of each tumor.
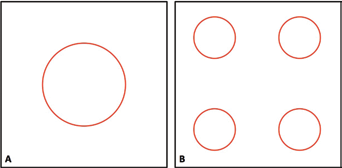
Simplified representation of same-size tissue areas with circles representing cross-sections of microvessels. Diameter of circle in A = 2 × diameter of circles in B; MVDA = 1; MVDB = 4; total vessel lumen area (A) = total lumen vessel area (B); total vessel perimeter (A) = 1/2 × total vessel perimeter (B).
Given the fact that in routine diagnostic pathology tissue paraffin blocks are made from every 5-mm interval of the excised mass, with the proposed method it is possible to analyze different areas of the whole tumor. Together with imaging modalities, such as DCE-MRI or microbubble contrast enhanced ultrasonography that can provide blood flow dynamics in vivo, this can help finding a more reliable histological angiogenic marker than conventional MVD.
In our preliminary study, we showed that the total number of CD31 stained vascular structures (MVD) in representative normal versus malignant epithelial mammary tissues was not significantly different (17). However, malignant tissue-associated CD31 positive structures had far more irregular shapes and smaller lumen area than normal tissues. Although these findings are not far from our expectation, they confirm the usefulness of digital pathology in providing more microvessel characteristics in a much wider scale than conventional vessel count in hot zones.
Further analyses are being undertaken in order to verify the reliability of image analysis-derived data in distinguishing tumor vascularity and oxygenation in vivo.
In conclusion, the morphological characteristics of tumor microvessel cross-sections, analyzed by image analysis on virtual slides from different sections of tumors, can shed light on finding new angiogenic profiles for breast cancer.
