Abstract
Aims
Inhibition of angiogenesis is an effective treatment option for metastatic colorectal cancer. Predictive biomarkers to select patients who are most likely to benefit from this therapeutic strategy are lacking. We conducted a pilot, retrospective biomarker study in a cohort of metastatic colorectal cancer patients treated with bevacizumab. The objectives of this study were to evaluate the prognostic value of biomarker expression in metastases and to compare their expression in paired tumor specimens.
Materials and methods
Eligible patients were treated with a bevacizumab-containing therapy; from these patients, tumor tissue from metastases was available. PTEN, PI3K p110a, c-MET, and CAIX were analyzed by immunohistochemistry.
Results
Forty-two patients received bevacizumab, 13 (31%) with first-line and 29 (69%) with second-line chemotherapy. Expression of CAIX, PI3K p110a, and c-MET in metastases did not predict objective response. PTEN loss was associated with response to treatment (p=0.02) and this association remained significant after adjusting for prognostic variables (p=0.006). However, no association with survival outcomes was found. In 32 patients (76%) with available paired specimens, we observed an equal expression between primary tumors and corresponding metastases in 75% of cases for CAIX in epithelial tumor cells, 56% for CAIX in stromal cells, 63% for PTEN, and 87% for c-MET.
Conclusion
PTEN loss in metastases appears to be associated with response to bevacizumab-based therapy. However, larger studies are necessary to confirm the potential role of the PI3K/AKT/mTOR pathway in modulating the therapeutic effect of bevacizumab. Tumor heterogeneity should be taken into consideration when analyzing tumor tissues for biomarker studies.
Introduction
Angiogenesis has been demonstrated to play a crucial role in cancer growth and progression (1–2–3), and targeting angiogenesis has become an established therapeutic option in several malignancies. Bevacizumab (Avastin; Genentech Inc, South San Francisco, CA, US) is a recombinant, humanized monoclonal antibody that exerts anti-angiogenic activity by binding to and inhibiting the vascular endothelial growth factor (VEGF), a key mediator in tumor angiogenesis (4, 5). In metastatic colorectal cancer (mCRC), adding bevacizumab to standard first and second-line chemotherapy resulted in a significant improvement in response rates (RR), progression-free survival (PFS), and overall survival (OS) (6–7–8–9–10–11).
Studies investigating predictive factors of responsiveness or resistance to bevacizumab have provided inconsistent results and have largely failed (12–13–14). This failure is likely due to the complexity of angiogenesis and to the intricate network of intracellular signaling pathways underlying tumor response to hypoxia. Indeed, although VEGF plays a key role in promoting tumor cell survival, growth, and invasiveness, several cellular types, growth factors, and intracellular mediators are involved in tumor angiogenesis (5, 15). Moreover, several concerns on biomarker studies still remain, including the lack of validation methodology for biomarker analysis and the use of old samples from primary tumors that are no longer representative of the actual tumor biology (16).
The VHL/HIF-1 axis is not the only mediator of the hypoxia-induced angiogenic response (17, 18). The PI3K/AKT/mTOR pathway via RHO/RHO kinase and the transcriptional factor c-Myc was shown to activate HIF-1-independent, angiogenic pathways (19). Moreover, preclinical data showed that inhibition of PI3K resulted in downregulation of VEGF (19).
c-MET is a receptor tyrosine kinase with high binding affinity for the hepatocyte growth factor (HGF). In human cancers, c-MET and HGF are often deregulated and associated with poor prognosis (20, 21). Even more interestingly, c-MET was shown to promote tumor invasiveness and metastasis (22–23–24) especially following drug-induced hypoxic conditions (25).
Carbonic anhydrase IX (CAIX) is an enzyme involved in the regulation of intracellular and extracellular pH (26). CAIX expression has been shown to correlate with poor outcome in several malignancies. CAIX is upregulated in CRC (27–28–29–30–31) and this upregulation may represent a marker of tumor metabolic adaption during hypoxia and may favor tumor proliferation in acidic conditions.
Based on these data, we performed a pilot, retrospective biomarker study investigating both the primary and metastatic tumor expression and prognostic value of PTEN, PI3K subunit p110a, c-MET, and CAIX in a small cohort of mCRC patients treated with a bevacizumab-containing therapy.
Materials and Methods
This was a multicenter, retrospective study conducted in 2 Italian centers: Humanitas Cancer Center/Humanitas Research Hospital (Rozzano, Milan), and Humanitas Gavazzeni (Bergamo). The study was approved by the institutional review boards.
Patients were eligible for this study if they had: (i) histologically confirmed CRC; (ii) received a bevacizumab-containing therapy for the treatment of metastatic disease; (iii) regular, on-treatment radiological assessment for evaluation of tumor response; (iv) archived tumor tissue from metastatic sites available for biomarker analysis.
Patient medical records were used to collect baseline clinicopathological characteristics including gender, age, site of primary tumor, disease-free survival (DFS), number and sites of metastases, levels of carcinoembrionic antigen (CEA), carbohydrate antigen (CA) 19-9, alkaline phosphatase (ALP), and lactate dehydrogenase (LDH), as well as chemotherapy regimen used in combination with bevacizumab.
Tumor assessment was performed with CT scan every 2-3 months as per clinical practice in the institutions participating in the study. PET or bone scans were not routinely performed and were requested only when clinically indicated. For the purpose of this study, baseline and on-treatment scans were systematically reviewed by 2 radiologists who were blinded to the results of the biomarker analysis. Tumor response was reported according to the Response Evaluation Criteria in Solid Tumors (RECIST) version 1.1.
Biomarker analysis
The expression of PTEN, PI3K subunit p110a, c-MET, and CAIX was assessed by immunohistochemistry (IHC) on 4 consecutive 2-µm thick tissue sections of formalin-fixed, paraffin-embedded (FFPE) specimens. Sections were immunostained with biomarker-specific antibodies as follows: PTEN (Cell Signalling 9559; dilution 1:100), PI3K subunit p110a (Cell Signalling 4249; dilution 1:200), CAIX (Abcam AB15086; dilution 1:2,000), and c-MET (Assay Designs ADI-905-076; dilution 1:100). Slides were then incubated with the secondary antibody using the MACH1 Universal HRP-Polymer Detection kit (Biocare Medical). Staining was performed with 3,3′ diaminobenzidine (DAB) as a chromogen and sections were then counterstained with hematoxylin. For each batch, positive and negative control slides without the primary antibody were also included.
Biomarker analysis was performed on archived tissue from metastatic sites and, when available, on tissue from the correspondent primary tumors. For patients with archived tissues from 2 or more metastatic sites, all tumor specimens were analyzed. Nevertheless, only the results from the biomarker analysis conducted on tumor tissue obtained before the start of the bevacizumab-based treatment were used for correlation with the clinical data. All the analyses were performed by pathologists who were blinded to baseline patient characteristics and clinical outcomes.
Immunohistochemistry
For each antibody, immunoreactivity was expressed as membrane (for CAIX), membrane/cytoplasmic (for c-MET and p110a), and cytoplasmic (for PTEN) staining, and its intensity was scored according to a four-tier system: 0, no staining; 1, weak; 2, moderate; 3, strong. The percentage of immunoreactive tumor cells was recorded for each tissue specimen. PTEN, PI3K p110a, and c-MET expression was analyzed only in epithelial tumor cells, while the expression of CAIX was assessed in both epithelial tumor cells and tumor stromal cells (myofibroblasts).
Statistical analysis
The objectives of this retrospective, exploratory study were: to assess the value of PTEN, PI3K p110a, c-MET, and CAIX expressed in tumors from metastatic sites, in predicting the outcome of mCRC patients treated with a bevacizumab-containing therapy, as well as to evaluate the concordance of biomarker expression between primary tumors and metastases.
PFS was calculated as the time from treatment start to progressive disease or death from any cause. OS was calculated as the time from treatment start to death from any cause. Patients who were still alive or alive with no evidence of progressive disease when this analysis was conducted were censored respectively at the time of the last contact or last radiological assessment.
Data are summarized as frequencies and proportions or as medians and ranges, as appropriate. Logistic regression was carried out to determine the association between biomarker expression and therapy responsiveness, calculating the adjusted OR with 95% confidence intervals (95% CI). The Cox proportional hazards models was applied to evaluate the hazard ratio (HR) with corresponding 95% CI, controlling for the effect of possible confounders. The McNemar's test was used to compare the expression between primitive and metastatic tumor sites. A p value less than 0.05 was considered to be the limit of statistical significance. All statistical analyses were performed using the R package.
Results
From August 2004 to March 2011, 42 patients who met the study eligibility criteria were identified in the 2 institutions. Baseline patient characteristics are reported in Table I. Patients included 28 males (66.7%) and 14 females (32.3%), and their median age was 64.4 years (range 42.4-80.0 years). In 32 patients (76.2%) the primary tumor was localized in the colon and in 10 patients (23.8%) in the rectum. The majority of patients (64.3%) presented with metastatic disease at diagnosis. In 13 patients (31%) bevacizumab was given with first-line chemotherapy, while 29 patients (69%) received bevacizumab in association with second-line chemotherapy. In the majority of cases (81%), irinotecan was the companion chemotherapy agent. The median number of cycles of bevacizumab administered with first-line and second-line chemotherapy was 12 (range 3-29) and 10 (range 3-20) respectively.
Baseline Patient Ch Aracteristics
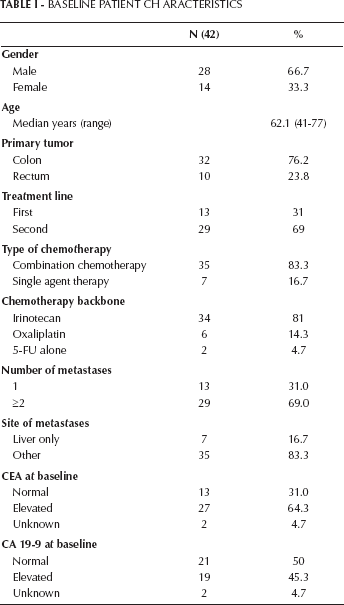
A favorable response to treatment (complete response or partial response) was observed in 76.9% of patients treated in the first-line setting and in 31% of patients treated in the second-line setting. Median PFS was 13.3 months in patients treated in the first-line setting and 8.8 months in patients treated in the second-line setting. The median follow-up was 32 months (range 2.3-49 months). At the time of this analysis, 14 patients (33.3%) were still alive while 28 had died (66.7%). The median OS was 26.4 months for patients receiving bevacizumab with first-line chemotherapy and 17.3 months for patients treated with second-line therapy. Among the baseline clinical factors, number of metastases (1 vs ≥2) and line of treatment (first vs second) were predictive of tumor response, while CA 19-9 level (normal vs high), line of treatment (first vs second), chemotherapy regimen (single agent vs combination therapy), and chemotherapy companion drug (irinotecan vs oxaliplatin) were predictive of PFS and OS.
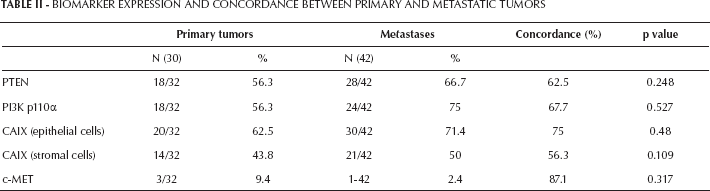
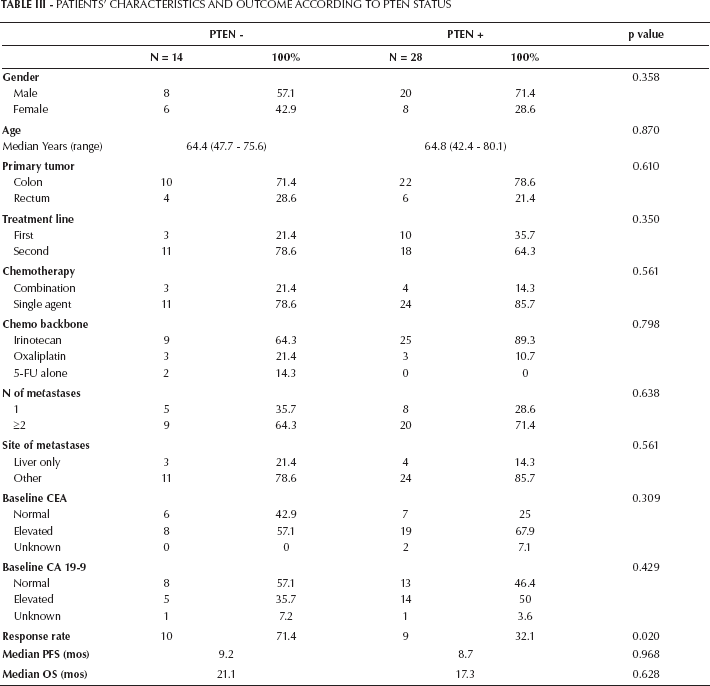
Biomarker expression levels in tumor samples from metastatic sites are reported in Table II: CAIX expression in epithelial tumor or stromal cells, as well as PI3K p110a and c-MET expression in epithelial tumor cells were not found to predict tumor response. By contrast, PTEN loss was associated with increased response to treatment. In patients with tumors exhibiting PTEN loss the response rate was 71% compared to 32% in patients with PTEN-expressing tumors (p=0.02) (Tab. III). In a multivariate analysis, the association between PTEN loss and tumor response remained significant after adjusting for number of tumor metastases and line of treatment (p=0.006) (Tab. IV). When the survival outcomes were analyzed, none of the biomarkers considered was found to predict PFS or OS in a univariate analysis.
Biomarker Expression And Concordance Between Primary And Metastatic Tumors
Patients’ Characteristics And Outcome According To Pten Status
Association Between Biomarker Expression And Tumor Response

Adjusted for treatment line and number of metastatic sites.
Odds ratios and p values for the association between c-MET expression and tumor response are not presented due to the small number of c-MET-positive tumors in the study population.
Thirty-two out of 42 patients (76.2%) had also tissue specimens from the primary tumor available for analysis. The expression of biomarkers in paired specimens is reported in Table III. Concordance between primary tumor and corresponding metastasis was observed in 75% of the cases for CAIX in epithelial tumor cells, 56% for CAIX in stromal cells, 62% for PTEN, and 87% for c-MET. When the expression of biomarkers in the primary tumor was correlated with patients’ response (PFS and OS), the PI3K p110a expression was continuously associated with improved PFS. However, this association did not remain significant after adjusting for other prognostic variables including CA 19-9 level, line of treatment, chemotherapy regimen, and chemotherapy companion drug.
For 3 patients, tissue from metastases taken before and after the administration of bevacizumab was available for analysis. In 2 cases a change in the expression of PTEN was observed (after treatment 1 tumor became PTEN-positive and 1 became PTEN-negative). The expression of CAIX in both epithelial tumor and stromal cells, as well as that of PI3K p110a in epithelial tumor cells changed in 1 case. No changes in the expression of c-MET were observed before and after bevacizumab treatment.
Discussion
In this study we report the results of an exploratory, retrospective biomarker analysis conducted in a small cohort of mCRC patients treated with bevacizumab. The biomarkers here analyzed were selected based on preclinical evidence and preliminary clinical data suggesting their potential involvement in the mechanisms of angiogenesis and hypoxia-induced response.
By restricting our analysis to patients with available tumor tissue from metastatic lesions, we assessed the role of these biomarkers in tissue samples that were likely to be representative of the tumor biology at the time of the bevacizumab-based therapy. Tumor heterogeneity is an established phenomenon that results in significant differences between primary tumors and metastases as well as between different metastases originated from the same tumor (35, 36). Tumor heterogeneity can influence the overall tumor response to treatment and can also negatively affect the ability of a specific biomarker to truly predict the effect of the treatment. Interestingly, when we compared metastases and corresponding primary tumors, the discordance rate for biomarker expression was relatively high, ranging from 13% to 44%. Unfortunately, only for 3 patients metastatic tumor samples before and after the use of bevacizumab were available; this small number of samples did not allow us to investigate potential changes in biomarker expression induced by the use of an anti-angiogenic therapy.
Although the importance of repeated tumor sampling is being recognized in the setting of both clinical trials and routine practice, most of the available information on individual tumor characteristics are still largely based on the assessment of archived tumor tissue taken at the time of the initial diagnosis. We acknowledge that, due to the limited tumor sampling, our study does not take into account the potential contribution of the intralesional heterogeneity to the substantial discordance in biomarker expression between primary tumors and metastases. However, our findings are in line with the recent reports on tumor heterogeneity and clonal evolution, and further support the view that the source of tumor tissue should be an important factor to consider when interpreting the results of tumor biomarker analyses.
We found that lack of PTEN expression in the metastases was an independent predictor of response to treatment with chemotherapy plus bevacizumab. PTEN has already been reported to act as a potential biomarker. In particular, PTEN loss has been suggested to be a negative predictive factor for benefit from other targeted agents such as cetuximab in mCRC and trastuzumab in metastatic breast cancer (36–37–38). To our knowledge, this is the first study to show a potential role for PTEN in predicting response to bevacizumab in mCRC. Given that PTEN loss results in activation of the PI3K/AKT/mTOR pathway, it could be hypothesized that tumors with increased activation of PI3K-dependent angiogenic pathways may be more sensitive to the therapeutic effect of bevacizumab (39). However, it is worth noting that, possibly due to the small sample size, no significant differences in survival outcomes were observed in our population according to the expression of PTEN. Very limited information is currently available on the role of PTEN in predicting the outcome of an anti-angiogenic therapy in CRC. The largest set of data is from a retrospective analysis of the AGITG MAX trial, in which the additional benefit of bevacizumab on PFS and OS was not found to be significantly greater among the patients with tumors exhibiting PTEN loss compared to patients with no PTEN loss in tumors. However, 94% of the tumor tissue analyzed was from the primary site, and all samples were analyzed for PTEN copy number (40).
In contrast with previous studies, CAIX expression, either measured in epithelial tumor cells or stromal cells, was not found to predict tumor response, PFS, or OS (41, 42). Similarly, although preclinical data suggested a role for c-MET in promoting invasiveness and metastasis in hypoxic conditions (25), no association was found between the expression of this biomarker and treatment outcome. However, the association between c-MET and bevacizumab effect could not be properly assessed due to the small number of c-MET-positive tumors in our study.
We also analyzed the expression of PI3K p110a in order to further investigate the potential of the PI3K/AKT/mTOR axis in mediating responsiveness or resistance to an anti-VEGF strategy. PI3K p110a expression in metastatic tumors did not predict treatment outcome; however, its expression in primary tumors was associated with an improvement in PFS in univariate analysis. Although the association was not significant in multivariate analysis, this finding would possibly be in line with the hypothesis of an increased efficacy of bevacizumab in tumors with upregulation of the PI3K/AKT/mTOR pathway.
Studies suggest that high LDH levels are associated with poor prognosis in CRC patients treated with bevacizumab and may represent a potential stratification factor for treatment selection (43, 44). In our study, baseline LDH levels were available for a limited number of patients (n=25). Although a trend towards a better outcome for patients with normal LDH at baseline was observed, this was not statistically significant (data not shown).
We acknowledge that our analysis is negatively affected by several factors. These include the retrospective design of the study, the small sample size and heterogeneity of the study population, the inherent limitations associated with the use of IHC, and the absence of a validated scoring system for any of the biomarkers analyzed. Moreover, our study did not include a control group treated without bevacizumab and, even in the presence of a significant association between PTEN and tumor response, a true predictive (rather than prognostic) value of this biomarker could not be demonstrated.
Conclusions
In this exploratory, retrospective study we showed that significant heterogeneity exists between primary colorectal tumors and metastases with regard to the expression of several biomarkers including CAIX, PTEN, PI3K, and c-MET. Moreover, we found that PTEN loss in the metastases was associated with increased tumor response to a bevacizumab-based therapy. Larger studies are necessary to further investigate whether the PI3K/AKT/mTOR pathway may truly modulate the therapeutic effect of bevacizumab and whether PTEN may represent a valid biomarker for the selection of patients more likely to benefit from an anti-angiogenic therapy.
