Abstract
Measurement of chromogranin-A (CgA) levels is relevant for the diagnosis of neuroendocrine neoplasms. The use of CgA testing for risk stratification of cardiovascular diseases is also increasing. The objective of our study was to determine the performances and reference values of a novel automated assay for CgA testing. The new method was compared with an enzyme-linked immunosorbent assay. Our results showed that the performances of the automated assay were satisfactory and that the agreement between the two methods was excellent. The automation of CgA testing also reduced the turnaround time of analysis and, therefore, might contribute to a faster delivery of the results to physicians.
Introduction
Chromogranin-A (CgA) is a 49-kDa acid protein present in the secretory granules of neuroendocrine cells (1). Intracellular CgA is essential for the formation of secretory granules and sequestration of hormones in neuroendocrine cells. The measurement of CgA levels is included in the work-up procedure of neuroendocrine neoplasms and pheochromocytoma, and reliable CgA testing has improved their diagnostic accuracy (2, 3). CgA has also been detected in both rat and human cardiac secretory granules, where it co-stores with natriuretic peptide hormones; thus, its use for the risk stratification of cardiovascular diseases has been proposed (1).
Several assays are currently available for CgA testing, but they remain mostly manual or semi-automated and need longer turnaround time of analysis (TAT), a feature that might impair a fast transmission of results to physicians. The CgA range measurable with current assays remains limited and requires additional dilutions for high levels of the protein. Furthermore, as the formats of the assays are different, the clinical interpretation of CgA results may be limited by the considerable heterogeneity among the commonly available CgA assays (4).
Recently, an automated assay for CgA measurement has been developed and might contribute to reduce the TAT of CgA testing (5).
The objective of our study was therefore to evaluate the analytical performances of this novel automated immunoassay for CgA measurement, and to determine its reference values in healthy volunteers.
Materials and Methods
We performed the evaluation of the Kryptor® CgA assay (B.R.A.H.M.S GmbH, Thermo Scientific, Germany), a method based on a two-site homogenous immunoassay format and fluorescence detection. The detection limit was determined with a 10 g/L bovine serum albumin solution (BSA 1%), while method imprecision was determined with quality control materials and with 2 pools of serum samples. The linearity of the CgA automated method was verified with 4 dilution tests of 2 samples with high CgA concentrations. The method was compared to an enzyme-linked immunosorbent assay (ELISA; Dako, Glostrup, Denmark) using serum samples of 85 patients monitored at the Cliniques Universitaires St-Luc, an academic hospital in Brussels, Belgium. For each patient, 15 mL of blood were collected from the antecubital vein into dry tubes. Samples were centrifuged within 1 hour of collection and serum was carefully separated and stored at -80ºC until assayed.
The reference values for the Kryptor® CgA assay were determined with serum samples collected from 120 healthy, normotensive volunteers who were not taking any medications. All participants gave their informed consent, and procedures followed the guidelines of the Helsinki Declaration. Statistical analyses were performed using the Medcalc® software. The CgA concentrations in healthy population followed a normal distribution. The distributions of the CgA levels in patients are reported as log-normal and statistics were therefore performed on log-transformed data. The Spearman's coefficient of correlation was calculated. The inter-agreement between methods was also determined through the evaluation of the Cohen's Kappa coefficient.
Results
The detection limit of the Kryptor® CgA assay was 6.2 ng/mL (n=15). The within-run coefficients of variation (CVs) were 3.9% and 3.3% (n=15), and the between-run CVs were 4.3% and 3.7% (n=20) for the mean concentrations of, respectively, 97 ng/mL and 480 ng/mL. The between-run CVs observed in our laboratory with the pools of serum were 3.9% and 2.7% at the mean CgA concentrations of, respectively, 85 ng/mL and 350 ng/mL. The recoveries after a 4-dilution test (1:2, 1:4, 1:8, and 1:16) were 92%, 111%, 113%, and 108% for a sample with a CgA concentration of 1,147 ng/mL, and were 94%, 101%, 107%, and 101% for a sample with a CgA concentration of 1,698 ng/mL, confirming the linearity of the Kryptor® method. The TAT of CgA measurement, from the reception of the whole blood tube at the laboratory to the transmission of the result to the physician, was less than 1 hour (57 minutes).
In the healthy volunteers’ group, the values’ range for circulating CgA levels was 7-115 ng/mL, and the upper limit of the 99% confidence interval was fixed at 103 ng/mL (Fig. 1). On this basis, our institutional cutoff was fixed to 100 ng/mL.
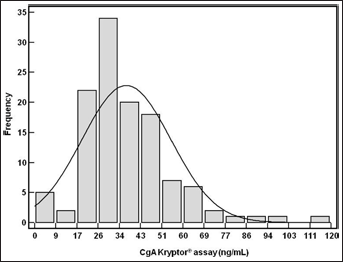
CgA levels, measured with the Kryptor® automated immunoassay, in healthy volunteers.
The median CgA level was 29.3 IU/L (range: 4.7-53,687 IU/L) when determined with the ELISA method, and was 97.7 ng/mL (range: 6.2-165,700 IU/L) when measured with the Kryptor® automated assay. A strong positive correlation was obtained between the 2 methods (r2=0.97; p<0.001) (Fig. 2).
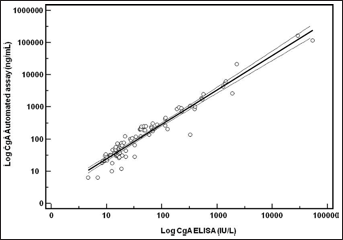
Comparison between the Kryptor® CgA automated immunoassay and an ELISA method.
With our institutional cutoff of 23 IU/L for the ELISA assay, and a cutoff of 100 ng/mL for the Kryptor® automated assay, the overall inter-rate agreement between methods was excellent with a κ coefficient of 0.89. However, in 5 cases we found discrepancies between the results of the 2 methods, and these were not related to patients with neuroendocrine neoplasms or pheochromocytoma.
Discussion
Our study shows very good analytical performances of the Kryptor® automated CgA assay, and an excellent agreement with one of the reference CgA methods; it also provided reference values for this novel automated assay.
Furthermore, our results show excellent repeatability and reproducibility for the Kryptor® CgA assay, as well as the ability to measure automatically very high levels of CgA, and a TAT shorter than that of other CgA assays currently available. Reliable analytical characteristics have also recently been observed for the Kryptor® CgA assay in another recent study (5). The automation of the CgA assay is important as it can contribute to a faster delivery of results to physicians. Without compromising measurement quality and patient safety, the Kryptor® CgA assay might also provide a more advantageous cost base, through a reduction of both the manual time dedicated to the measurement and the need of manual dilutions for high CgA values.
The clinical validity of this novel CgA assay also needed to be investigated. The diagnostic accuracy of several ELISAs or radioimmunoassays for CgA measurement was previously reported (2, 3, 6, 7). The Kryptor® CgA assay has been previously compared to a radioimmunoassay, and both methods agreed well on the basis of the cutoff values provided by the manufacturers (5). The clinical sensitivities of Kryptor® CgA for the diagnosis of pheochromocytoma and paraganglioma, or gastroenteropancreatic neuroendocrine tumors were respectively 100% and 94% (5). Our results showed an excellent concordance between the Kryptor® assay and an ELISA method already validated and recognized for its clinical performances (4). Nevertheless, we observed a limited number of discrepancies between the assays, despite being not statistically significant. As the antibodies used and the analytical properties of the respective kits are different, such discrepancies might be expected (4). It is well established that CgA assays are not commutable, and specialists in laboratory medicine might therefore assume a role of consultants to physicians and inform them about the characteristics and performances of the novel CgA assay.
The importance of a novel automated assay for CgA measurement might be broader than neuroendocrine neoplasms as the potential value of CgA testing for the management of cardiovascular diseases is increasing. Indeed, CgA appears as a novel factor acting at the cross road between the neuroendocrine and the cardiovascular systems (8). The interplay between CgA-derived peptides and cardiac natriuretic peptides in cardioprotection against catecholamine-evoked stress has been reported (9). CgA is also the precursor of several peptides active on the cardiovascular system and heart, and produced by proteolytic cleavage. One of these, vasostatin-1, acts as an inhibitor of cardiac contraction and relaxation, and is presented as a non-competitive counter-regulator of beta-adrenergic stimulation and a protecting agent in ischemic preconditioning (10). Recently, the use of measured CgA circulating levels for the risk stratification of cardiac diseases has been suggested (1, 11). Ceconi et al determined the CgA levels in 160 patients with heart failure (12). Their results showed that CgA levels are increased in chronic heart failure, are related to the clinical severity of the condition, as estimated by the New York Heart Association (NYHA), and were independent predictors of mortality in a multivariate Cox analysis (12). Dieplinger and colleagues have also reported that, in 137 consecutive patients with acute destabilized heart failure attending the emergency department, CgA testing added independent prognostic information to N-Terminal-proB-type natriuretic peptide measurement (13).
In conclusion, reliable and accurate measurements of CgA were obtained with the Kryptor® assay. This novel automated assay also reduced the time of results’ delivery to physicians and might contribute to the clinical assessment of neuroendocrine neoplasms and cardiovascular diseases.
Footnotes
