Abstract
Introduction
Finasteride is a steroid 5-alpha-reductase inhibitor (5-ARI) (commercial name Proscar®; Propecia®) that was approved for the treatment of benign prostatic hyperplasia and androgenetic alopecia (AGA). Finasteride inhibits the 5-alpha-reductase thus blocking the conversion of testosterone to 5-alpha-dihydrotestosterone (DHT), and thereby impairing androgen action through the androgen receptor (AR). Several studies reported that the use of 5-ARI may determine undesirable side effects on overall health and, in particular, sexual function, as well as a predisposition to onset or progression of depression in a subgroup of susceptible patients (1, 2). An association between androgen deficiency and depression has been proposed, although the exact mechanisms remain to be investigated. Low androgen levels have been associated with symptoms of irritability and dysphoria (3-4-5), increased risk of depressive symptoms, and depression (6-7-8-9). In some patients the finasteride-associated side effects were described as long lasting or even persistent with regard to sexual function and with an emotional toll including decreased quality of life (10, 11); this clinical condition has been defined post-finasteride syndrome (PFS).
Previous studies demonstrated the existence of a pharmacogenetic component in the response to finasteride treatment (12); in particular, 2 polymorphisms (rs4045402 and rs3138869), characterizing the AR encoding gene, are related to the AR-mediated signal transduction efficiency, and are hypothesized to play a role in finasteride sensitivity (13, 14). The AR is encoded by the AR gene, located on chromosome Xq11–12. The AR contains 8 exons that encode 3 functional domains of the receptor: the transactivation domain (exon 1), DNA-binding domain (exons 2 and 3), and the ligand-binding domain (exons 4–8) (15). Rare mutations that result in complete or partial androgen insensitivity syndromes have been localized in both the ligand-binding and DNA-binding domains (16). The N-terminal transactivation domain controls the transcription of the AR target genes. Within this domain, there are 3 microsatellite trinucleotide repeats. Two of these repeats are polymorphic and vary in length in the population: a (CAG)nCAA repeat encoding polyglutamine, denoted as CAGn (rs4045402), and a (GGT)3GGG(GGT)2(GGC)n repeat encoding polyglycine, denoted as GGNn (rs3138869). The CAG number of repeats varies in length, with African-Americans having on average fewer repeats than Caucasians, in whom the most common CAG repeats range from 20 to 22 (17) (the length is slightly shorter in African-Americans and longer in Asians) (18, 19). Experimental research suggests that the number of repeats in the CAG and/or GGN tract is correlated with the transcriptional activity of the AR protein (20-21-22). Some preliminary findings gave polyglutamine lengths (CAG)n a clinical value, linking longer (CAG)n stretches with reduced virilization and defective spermatogenesis among men affected by spinal bulbar muscular atrophy, a fatal neuromuscular disease (23). Previous studies in Asian men identified a pharmacogenomic response to finasteride therapy for AGA based on androgen sensitivity and on the effect of CAG repeats in AR (14). Moreover, several studies suggested that 1 or both of the trinucleotide repeat polymorphisms in AR may be associated with prostate cancer (24, 25). In particular, it appears that shorter (CAG)n or (GGN)n alleles predispose to a higher risk of prostate cancer development.
Aim of this study was to assess a possible difference in the distribution of the number of CAG (rs4045402) and/or GGN (rs3138869) repeats in patients experiencing long-term side effects after finasteride treatment for AGA (AGA+PFS); these patients were compared to 2 different groups of male subjects: men with AGA not treated with finasteride (AGA group), and controls without AGA (NO-AGA group).
Material and Methods
Subjects’ Enrolment
We retrospectively enrolled 3 groups of male subjects having Caucasian mothers (because of the AR location on the X chromosome) aged over 18 years. The AGA+PFS group included patients that used finasteride against AGA and referred persistent (i.e. more than 6 months) side effects (included in the definitions of PFS) after drug discontinuation. Patients were recruited both from the Urological Department of Trieste University Hospital (Italy) during routine clinical practice (n=11), and from the Propeciahelp.com Internet forum (http://www.propeciahelp.com/forum/) (n=58). Research experts previously standardized the interview modalities. Eligible subjects were white male patients older than 18 years, who took finasteride once daily to treat AGA and experienced persistent side effects related to sexual dysfunction (evaluated by ASEX score) and/or health-related quality of life and symptoms of aging men (evaluated by AMS score) for at least 6 months after finasteride discontinuation. We excluded all patients with prior diagnosis of sexual dysfunction before finasteride treatment, as well as with prior diagnosis of psychiatric problems or medical co-morbidities, use of psychiatric drugs, drug abuse, or use of drugs with antiandrogenic activity. The second group of subjects, AGA, included unselected healthy male individuals affected by AGA (but never treated with finasteride). The third group of subjects, NO-AGA, was composed of healthy male blood donors not affected by AGA. AGA was diagnosed according to the Hamilton-Norwood criteria. All enrolled subjects, i.e. cases and controls, were evaluated for the presence or absence of AGA by the Hamilton-Norwood scale through a questionnaire reporting pictures showing grades from 1 to 7; the cases had grade 2 or higher (26, 27).
The Institutional Review Board and Ethical Committee of each participating institution approved the study protocol, and all the subjects signed a written informed consent form for the genetic analysis before entering the study.
Evaluation of Persistent Symptoms after Finasteride Discontinuation in Patients
Two different structured questionnaires were self-administered by the patients, supported by an expert clinician, to evaluate the development and severity of long-term side effects development. Patients enrolled at the University Hospital filled the questionnaires in the presence of an expert clinician, while patients enrolled from the Internet forum were called by phone by the same expert clinician after they filled the questionnaires at their home.
The Arizona Sexual Experience Scale (ASEX) questionnaire (28) was selected as a validated instrument to assess sexual function before and after finasteride assumption. The possible total scores ranged from 5 to 30, with the highest scores indicating a higher degree of sexual dysfunction. Sexual dysfunction is present if the total score is ≥19, if any item scores ≥5, or if any of 3 items scores ≥4 points.
The Aging Male Symptoms Scale (AMS) (29) questionnaire was adopted for measuring health-related quality of life and symptoms of aging men. The AMS is composed of 3 subscales measuring, respectively, the psychological, somatic, and sexual symptoms, for a total of 17 items. Each item is scored from 1 (absent) to 5 (very severe), according to the Likert scale. The sum score for each subscale is obtained by adding up the ratings of the specific items. The total score is the sum of the 3 subscale ratings. The androgen deficit is graded accordingly to the AMS total points as absent (17-26 points), slight (27-36 points), moderate (37-49 points), or severe (≥50 points).
Information about finasteride treatment included dosage, name of medication used, and duration.
AR Polymorphisms Genotyping
Genomic DNA was extracted from whole blood or saliva fractions using the High Pure PCR Template preparation Kit (Roche Diagnostics GmbH, Mannheim, Germany).
The AR (CAG)n and (GGN)n polymorphisms were analyzed by automated fragment analysis. For AR (CAG)n, the PCR primers were a forward 5’-HEX-labeled primer, 5’-CCA-AGC-TCA-AGG-ATG-GAA-3’, and a reverse primer 5’-GAA-GGT-TGC-TGT-TCC-TCA-3’. The reaction was carried out in a volume of 30 μL with 2.5 mM MgCl2, dNTPs each 250 μM, 500 nM of each primer and 1 Unit of Taq polymerase (Amplitaq Gold, Life Technologies), for 24 cycles of amplification (1 minute at 95°C, 30 seconds at 64°C, and 40 seconds at 72°C) followed by 11 additional cycles (1 minute at 95°C, 30 seconds at 56°C, and 40 seconds at 72°C), obtaining a fragment of variable length (348 bp for 21 CAG repeats). For AR (GGN)n, the PCR primers were a forward 5’-HEX-labeled primer, 5’-TGG-CAC-ACT-CTC-TTC-ACA-3’, and a reverse primer 5’-GAT-AGG-GCA-CTC-TGC-TCA-3’. The reaction was carried out in a volume of 30 μL with 1.5 mM MgCl2, dNTPs each 250 μM, 500 nM of each primer, 5% DMSO, and 1 Unit of Taq polymerase (Taq Polymed) for 40 cycles of amplification (1 minute at 95°C, 30 seconds at 58°C, and 30 seconds at 72°C) obtaining a fragment of variable length (235 bp for 16 GGN repeats). The diluted amplicons were mixed with the standard molecular weight ROX400 (Life Technologies), run on ABI-3130 automatic sequence analyzer (Life Technologies) to separate the 2 alleles’ peaks, and analyzed with the Gene Scan software (Life Technologies).
Statistics
The (CAG)n allelic variants were grouped as “most frequent” alleles (19 to 26 repeats) and “extremely short or long” alleles (<19 or >26 repeats). The GGN allelic variants were grouped as “most frequent” alleles (23 or 24 repeats) and “extremely short or long” alleles (<23 or >24 repeats). The choice of sorting the 2 polymorphisms into the “most frequent” and “extreme lengths” groups was based on a functional study by Nenonen et al. (22) and Lundin et al. (21), respectively. For both these polymorphisms the authors proposed a curvilinear association between the number of repeats and the AR transduction functionality, with the highest activity among the central, and most common, number of repeats.
The comparison of the “most common” and “extreme lengths” alleles prevalence among different groups of subjects was tested with the Fisher's exact test. In addition, to identify predictive genetic variants, the odds ratios (OR) and 95% confidence intervals (CIs) were computed for categorical variables using a univariate logistic regression model (p<0.025 cutoff, corrected by multiple testing).
Results
Subjects’ Enrolment
The clinicodemographic characteristics of the subjects enrolled in the AGA+PFS group are summarized in Table I. A total of 69 patients retrospectively recruited (AGA+PFS group) and fulfilling the inclusion criteria provided either a saliva or a blood sample and were genotyped for both the (CAG)n and (GGN)n AR polymorphisms. Most of the patients (58/69, 84.1%) were recruited through the Propeciahelp.com Internet forum. The majority of them took finasteride in a dosage of 1 mg per day (68.1%), and the treatment lasted for a mean period of 815 days (range 17-5,280 days). Ninety-two subjects with different grades of AGA were prospectively recruited and genotyped in the AGA group. The NO-AGA group included 79 healthy blood donors not affected by AGA that were retrospectively selected from a preexisting bio-bank and genotyped as an additional control group, after being questioned to assess their Hamilton-Norwood scale grade.
CLINICODEMOGRAPHIC CHARACTERISTICS OF PATIENTS THAT USED FINASTERIDE AGAINST AGA AND REFERRED PERSISTENT SIDE EFFECTS (AGA+PFS)
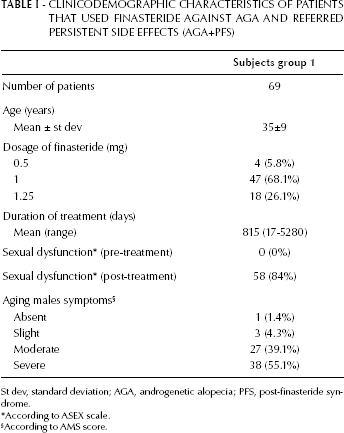
St dev, standard deviation; AGA, androgenetic alopecia; PFS, post-finasteride syndrome.
According to ASEX scale.
According to AMS score.
Post-Finasteride Syndrome
Table I reports the description of the occurrence of sexual dysfunction (measured by ASEX scale) as well as the health-related quality of life and symptoms of aging men (measured by AMS scale) as experienced by the patients after finasteride use. Overall, 84% of the patients self-reported the onset of a sexual dysfunction persistent for at least 6 months after discontinuation of the treatment. The vast majority of AGA+PFS patients (94.2%) described a moderate (39.1%) to severe manifestation (55.1%) of AMS symptoms that were not present before the use of finasteride. Patients spontaneously reported several other symptoms, including anxiety (24.1% of all group), irritability (74.1%), and depression (32.5%) (data not shown).
Androgen Receptor Polymorphisms Genotyping
The genotyping procedure was successful in 236/240 samples (98.3%) for (CAG)n, and in 237/240 samples (98.7%) for the (GGN)n polymorphism. For the (CAG)n variant, the number of repeats found ranged from 9 to 37, whereas for (GGN)n the range was between 16 and 26. The allelic frequency of the most common number of repeats in the 3 groups of subjects is reported in Table II. In the total sample of 236 subjects the most frequent length of CAG was 21 (40/236; 16.9%), the second most frequent lengths were 20 (32/236; 13.6%) and 24 (32/236; 13.6%), followed by 22 (26/236; 11.0%). Regarding the GGN repeats the most frequent length was 23 (151/237; 63.7%), followed by 24 (23.2%).
ANDROGEN RECEPTOR POLYMORPHISMS CAGn (rs4045402) AND GGNn (rs3138869) IN THE 3 GROUPS OF SUBJECTS
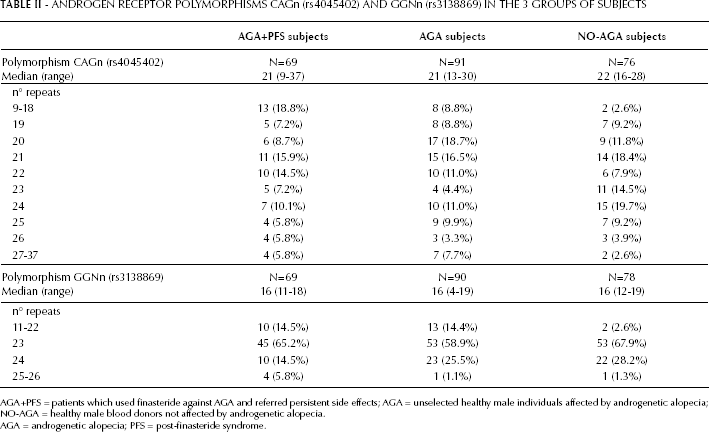
AGA+PFS = patients which used finasteride against AGA and referred persistent side effects; AGA = unselected healthy male individuals affected by androgenetic alopecia; NO-AGA = healthy male blood donors not affected by androgenetic alopecia.
AGA = androgenetic alopecia; PFS = post-finasteride syndrome.
Case-Control Analysis of the Androgen Receptor (CAG)n and (GGN)n Polymorphisms’ Lengths among the 3 Groups of Subjects
Both AR alleles had significantly different lengths in the 3 groups of subjects (Tab. III). We found a significantly higher prevalence of the “extreme lengths” alleles for both polymorphisms in the AGA+PFS group compared to the NO-AGA group (p=0.002 and p=0.003, respectively). Similarly, when comparing the lengths of the repeats between NO-AGA and AGA subjects we found a higher prevalence of the “extreme lengths” alleles in the AGA group (p=0.028 and p=0.019, respectively), despite being a less significant difference and with a smaller effect (Tab. III).
STATISTICAL COMPARISON OF THE DISTRIBUTION OF THE ANDROGEN RECEPTORS CAGn (rs4045402) AND GGNn (rs3138869) ALLELES IN THE 3 GROUPS OF SUBJECTS
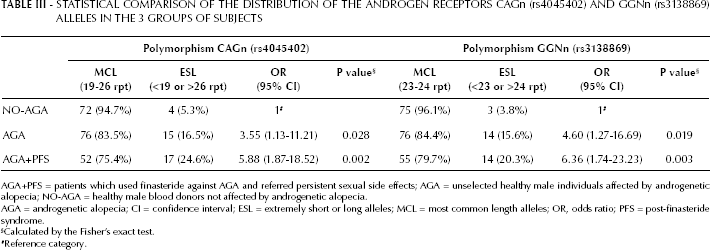
AGA+PFS = patients which used finasteride against AGA and referred persistent sexual side effects; AGA = unselected healthy male individuals affected by androgenetic alopecia; NO-AGA = healthy male blood donors not affected by androgenetic alopecia.
AGA = androgenetic alopecia; CI = confidence interval; ESL = extremely short or long alleles; MCL = most common length alleles; OR, odds ratio; PFS = post-finasteride syndrome.
Calculated by the Fisher's exact test.
Reference category.
Discussion
Finasteride administration for the treatment of AGA has been recently associated with the occurrence of severe sexual and psychiatric side effects in a subset of patients; these effects persist despite drug withdrawal and are sometimes associated with depression and suicidal thoughts (1, 10, 11). It has been previously reported that pharmacogenetics may play a role in the identification of good and poor responders to finasteride (12-13-14). We are the first to report a different distribution of 2 AR genetic polymorphisms (CAG-rs4045402 and GGN-rs3138869) in patients experiencing persistent side effects after finasteride treatment for AGA, as compared to 2 different groups of male control subjects.
The main finding of this work is that the allelic variants of both polymorphisms show a different prevalence in the selected group of patients treated with finasteride and affected by persistent adverse side effects (AGA+PFS group) as compared to a group of healthy men without AGA (NO-AGA group). Considering that the AR gene has been recognized as an important determinant for cellular androgen sensitivity and has been associated with hair loss in AGA, we compared our data to a group of healthy men with AGA (AGA group). Also in this case we found that the allelic variants of both polymorphisms were differently distributed in the AGA and NO-AGA groups.
The study allowed us to conclude that “extreme lengths” repeats in CAG-rs4045402 and GGN-rs3138869 are both risk factors for AGA. Similar findings were previously reported by others (30, 31), although a recent meta-analysis failed to confirm a link between the 2 AR genetic polymorphisms and the occurrence of AGA (32). However, in all these previous studies the authors assumed that the longer repeats were associated to a higher androgenic functionality for both the polymorphisms, thus considering longer CAG or GGN stretches as a possible risk factor for AGA. In our study, we grouped the allelic lengths according to the results of 2 recent, functional, in vitro studies (21, 22) reporting that the highest AR functionality corresponds to the central, and most common, value of CAG or GGN repeats, whereas extreme values of both polymorphisms are associated to a reduced AR functionality. Specifically, Lundin and colleagues (18) showed that the in vitro (GGN)23 activity was significantly higher than that of other lengths. ARs with glutamine repeats other than 23 had lower transactivating capacity in response to both testosterone and DHT. In 2011, Nenonen and colleagues (19) demonstrated that men with (CAG)<22 and (CAG)>23 had a 20% increased OR of infertility compared with carriers of the median lengths of 23 glutamine residues. A similar curvilinear association of CAG and GGN repeats length with some androgen-related reproductive parameters has been recently described in Inuit and Caucasian men (33). The rationale of such phenomena still needs to be fully elucidated; nevertheless, a reasonable “spatial” hypothesis is that the most frequent (CAG) and (GGN) repeats correspond, respectively, to a polyglutamine and polyglycinine lengths, which allow optimal folding of the AR protein to achieve full transcriptional activity. The adoption of this kind of stratification of the alleles, suggested by the hypothesized functional effect, has here been used for the first time in this kind of study, and allowed us to highlight a stronger predictive effect of the polymorphisms on the risk of AGA. A different stratification of the alleles could be among the reasons of the controversial results previously reported on this topic.
PFS is a disabling condition developed by an unspecified percentage of patients assuming finasteride for the treatment of AGA. The genetic background of the patients and, specifically, the genetic polymorphism in AR, could be at the basis of the syndrome. However, the molecular mechanism of the development of a persistent toxic syndrome in patients treated with finasteride for AGA is still unclear. Some reports have been published in the last years describing this phenomenon (1, 10, 11) but, to our knowledge, no formal randomized clinical studies have been recently published to describe its incidence. In addition, the transcription of AR in response to environmental stimuli, such as androgen levels variation, as those occurring during finasteride treatment, has been reported to be also epigenetically controlled by gene promoter CpG islands and post-translational histones methylation. In particular, a role of the LSD1 and JMJD2C demethylases in the epigenetic regulation of AR expression was described (34). Therefore, the proteins involved in the epigenetic regulation of genes’ transcription through CpG islands and histone methylation/demethylation (LSD1, JMJD2C, MGMT, DNMT1, DNMT3A, DNMT3B, DNMT3L and DNMT2 [TRDMT1]) could play a role in the AR expression regulation after finasteride discontinuation and development of chronic side effects.
To address such an important issue, prospective trials with control arms of patients assuming finasteride but not experiencing any persistent drug-related effect are highly needed. A limitation of the present study is actually the lack of a proper control group of patients undergoing finasteride treatment but not experiencing PFS. This implementation would be fundamental to verify the possible predictive effect of the 2 polymorphisms on PFS development.
Another limitation of the present study is the lack of estimation of the ASEX or AMS scores for the individuals retrospectively included in the control groups. Unfortunately, this prevents us to draw any final conclusion from our data. Our explorative study tried to shed a light on such phenomenon as well as to highlight a possible genetic predisposition to the development of such disabling chronic side effects that can deeply impact the quality of life of young men assuming finasteride to treat alopecia.
With our study we confirmed the predictive value of CAG-rs4045402 and GGN-rs3138869 on AGA development, and the validity to functionally group the extreme number of repeats as suggested by the most recent in vitro functional studies. Further prospective trials are warranted to better investigate this phenomenon and to highlight possible pharmacogenetic predictive markers of toxicity development.
