Abstract
Objective
The aim of this study was to look for predictive and prognostic factors in anthracycline-based neoadjuvant chemotherapy.
Methods
Three hundred and nine patients with early-stage or locally advanced breast cancer were enrolled in this study and preoperatively treated with neoadjuvant chemotherapies in intense dose-dense (cyclophosphamide + epirubicin) or conventional (paclitaxel + epirubicin) regimens. Outcome parameters included overall objective response rate, as well as factors for determining pathological features influencing the efficacy of chemotherapy, such as estrogen receptor, progesterone receptor, and HER2 status, Eastern Cooperative Oncology Group (ECOG) score, tumor size, tumor inflammation, and lymph node metastasis.
Results
The overall pathological complete response (pCR) rate was 14.3%, and the main factor affecting the efficacy of chemotherapy was HER2 expression. The pCR rate was significantly higher for patients with HER2 overexpression, compared with patients with low HER2 expression (27.5% vs. 9.6%, p<0.001). The recurrence risk for patients with pCR decreased 1.12-fold compared with patients without pCR (5-year disease-free survival [DFS]: 81.8% vs. 65.7%, p=0.038). Patients in the HER2 overexpression subgroup benefited more from pCR than those in the HER2 low expression group (5-year DFS: 86.4% vs. 62.1%, p=0.049).
Conclusion
HER2 overexpression in primary tumors might be a predictive marker for good efficacy of anthracycline-based neoadjuvant chemotherapy. The pCR is a suitable prognostic factor, even for patients with HER2 overexpression.
Introduction
Over the past 20 years, great progress has been made in the treatment of breast cancer, and the overall survival of breast cancer patients has been significantly improved with the development of comprehensive treatments, but the relationship between clinical features and prognostic factors for the efficacy of specific treatments has not been elucidated (1, 2). Human epidermal growth factor receptor 2 (HER2) gene amplification or its protein overexpression (HER2-positivity) occurs in 20%-30% of breast cancers, and for these patients, the higher pathological complete response (pCR) rate for a neoadjuvant chemotherapy with trastuzumab predicts improved survival (3). In contrast, for HER2-negative patients unresponsive to an initial neoadjuvant (cytotoxic and antiangiogenic cyclophosphamide plus epirubicin (CE) treatment with or without bevacizumab), a neoadjuvant therapy with everolimus and paclitaxel did not improve the pCR rate (4). Studies of predictive factors of response to chemotherapies in advanced breast cancer are commonly affected by previous treatments, and evaluating the efficacy of adjuvant chemotherapies is influenced by surgery and needs a long observation period. To study the effects of various clinical and pathological factors on different chemotherapies for primary breast cancer, comparing combined preoperative drug application and surgery facilitates a direct comparison of tumor repression and predictive factors between different treatments. Anthracycline-based regimens are known to be highly effective for the treatment of breast cancer and are most commonly used in neoadjuvant chemotherapy settings. A recent study showed that patients with HER2-negative tumors derived a larger benefit from adjuvant anthracyclines than did patients with HER2-positive tumors (5). Another report suggested that HER2-positive tumors had a lower rate of pCR than HER2-negative tumors, and negative HER2 status remained an independent favorable predictor of pCR, after controlling for age, estrogen and progesterone receptor status, tumor size, and chemotherapy cycles, as well as tumor grade, in a multivariate analysis of anthracycline-based neoadjuvant chemotherapies (6). Up to now, few studies have been done with large sample sizes, which describe the association between HER2 status and the response to neoadjuvant chemotherapy treatments. In this prospective study, we compared the efficacy of different preoperative neoadjuvant chemotherapy regimens in our hospital and analyzed correlations between the efficacy of different treatments and clinical features.
Materials and Methods
Patients
The clinical data of 309 patients with stage IIa-IIIc breast cancer who were treated with neoadjuvant chemotherapies in our hospital from January 2003 to December 2008 were included in this study. All patients received neoadjuvant chemotherapies with CE (intense dose-dense [IDD]) or paclitaxel + epirubicin (TE) regimens after core needle biopsies of primary tumors and their immunohistochemical examination for estrogen receptor (ER), progesterone receptor (PgR), and HER2 status were compared. The last follow-up date was March 2014, with a median follow-up of 60 months. All participants signed written informed consent forms, and the study was approved by the ethics committee of the Cancer Hospital, Chinese Academy of Medical Sciences, Peking Union Medical College.
Medications
CE-T regimen: cyclophosphamide 600 mg/m2 i.v., d1, and epirubicin 80 mg/m2 i.v., d1, q14d×4 cycles (CE) in neoadjvant chemotherapy, plus postoperative sequential paclitaxel (T) 175 mg/m2 i.v., d1, q14d×4 cycles.
TE regimen: epirubicin 75 mg/m2 i.v., d1, and paclitaxel 175 mg/m2 i.v., d2, q21d×4 cycles as neoadjuvant chemotherapy; 2 cycles of TE regimen were repeated after surgery.
Efficacy evaluation criteria
Efficacy was estimated every 2 cycles, according to response evaluation criteria in solid tumors (RECIST) 1.0 criteria, and the patients were evaluated by clinical, mammographic, and B-ultrasound examinations. Efficacy evaluation indicators included pCR (i.e., no invasive components in pathological specimens obtained through modified radical mastectomy after neoadjuvant therapy), disease-free survival (DFS; time from radical surgery to recurrence or metastasis), complete response (CR), and partial response (PR), as well as overall objective response rate (ORR = CR + PR), progressive disease (PD), and stable disease (SD). Patients with PR received surgical treatment after 4 cycles of chemotherapy and sequential paclitaxel or repeated TE regimen after surgery. Patients who met the criteria for SD or PD at the initial efficacy evaluation received surgery as soon as possible and were treated with alternative postoperative regimens.
Statistical Analysis
All data were analyzed using SPSS medical statistical software (version 15.0). The correlations between efficacy and various factors, such as patient menstrual status, age, tumor size, tumor inflammation, lymph node status, ER/PgR status, HER2 expression, general condition (Eastern Cooperative Oncology Group [ECOG] score), and neoadjuvant chemotherapy regimens were assessed using the chi-square test. The risk factors affecting DFS were identified and analyzed using the Cox multivariate regression model. Survival curves were drawn using the Kaplan-Meier function. Statistical significance was defined as a p value <0.05.
Results
Patient characteristics and treatment
From January 2003 to December 2008, 309 patients were enrolled in this study. The median number of neoadjuvant chemotherapy cycles was 4 (range 2-4 cycles). One hundred and nineteen patients received CE (most of patients received postoperative T×4) (IDD) chemotherapy, and 190 patients received TE chemotherapy (most of patients received postoperative 2 cycles). Patient characteristics, such as menstrual status, ECOG score, hormone receptor status, triple-negative breast cancer (TNBC) and HER2 status, tumor size, tumor inflammation, and clinical lymph node status were well balanced between the 2 groups (Tab. I). A total of 285 patients (92.2%) underwent modified radical mastectomy after neoadjuvant chemotherapies, and only 21 patients (6.8%) underwent breast-conserving surgery, while 3 patients (1.0%) did not receive any surgery, for various reasons. One hundred and eighty-six patients (60.2%) received radiotherapy, and 250 patients (80.9%) received adjuvant chemotherapy after surgery. One hundred and thirty patients continuously received 2 cycles of TE regimens in the TE group, accounting for 68.4% (130/190), while 76 patients received postoperative IDD chemotherapy with paclitaxel (T) alone in the CE (IDD) group, accounting for 63.9% (76/119). The chemotherapy regimens were changed for a total of 44 patients (14.2%) who had a poor response to preoperative chemotherapies. Fifty-nine patients (19.1%) did not receive postoperative adjuvant chemotherapies, including some patients with pCR. All 231 patients with preoperative or postoperative pathological ER-positive or PgR-positive breast cancer (74.8%, 231/309) received different postoperative adjuvant endocrine therapies.
PATIENTS’ BASELINE CHARACTERISTICS
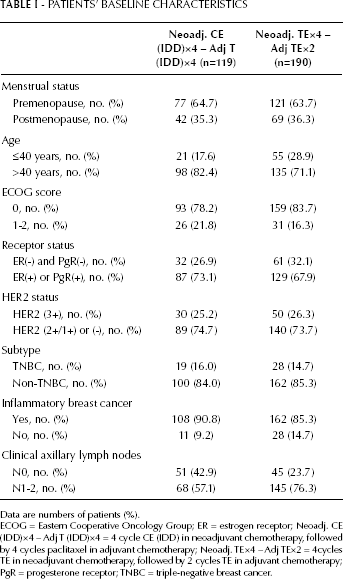
Data are numbers of patients (%).
ECOG = Eastern Cooperative Oncology Group; ER = estrogen receptor; Neoadj. CE (IDD)×4 – Adj T (IDD)×4 = 4 cycle CE (IDD) in neoadjuvant chemotherapy, followed by 4 cycles paclitaxel in adjuvant chemotherapy; Neoadj. TE×4 – Adj TE×2 = 4cycles TE in neoadjuvant chemotherapy, followed by 2 cycles TE in adjuvant chemotherapy; PgR = progesterone receptor; TNBC = triple-negative breast cancer.
Analysis of predictive factors
The evaluation of treatment efficacy showed that 44 patients (14.3%) achieved pCR, and 192 patients (62.1%) achieved PR, with 76.4% pathological ORRs. The pathological ORR outcome of the neoadjuvant chemotherapy was associated with the patients’ performance status at the beginning of the therapy, HER2 expression in primary tumors, and primary tumor sizes. Efficacy was better in patients with good performance status (ECOG score 0), compared with patients with poor performance status (ECOG score 1-2). Compared with patients with HER2 low expression (2+/1+) or no HER2 expression (-), efficacy was better in patients with HER2 overexpressing primary tumors (3+). Furthermore, efficacy was better in patients with a large tumor mass (primary tumor >5 cm in diameter) than in patients with a small tumor mass (primary tumor ≤5 cm in diameter). Pathological efficacy was not associated with age, axillary lymph node metastasis from primary tumors, or menstrual status (Tab. II). The pCR rate of neoadjuvant chemotherapies was only associated with the HER2 expression status of primary breast cancer tumors, which was significantly higher in patients with HER2-overexpressing primary tumors. The efficacy advantages among patients with HER2 overexpression were observed in both the CE (IDD) and the TE group, with pCR rates for patients with and without HER2 overexpression of 20.0% vs. 5.62% in the CE (p=0.029) and 29.1% vs. 12.6% in the TE group (p=0.007), respectively (Tab. III). Additionally, the pCR rate for patients with hormone receptor–negative breast cancer was higher than in hormone receptor–positive patients, but without statistical significance (p=0.068).
CLINICAL AND PATHOLOGICAL FACTORS AFFECTING ORR AND pCR IN ALL PATIENTS
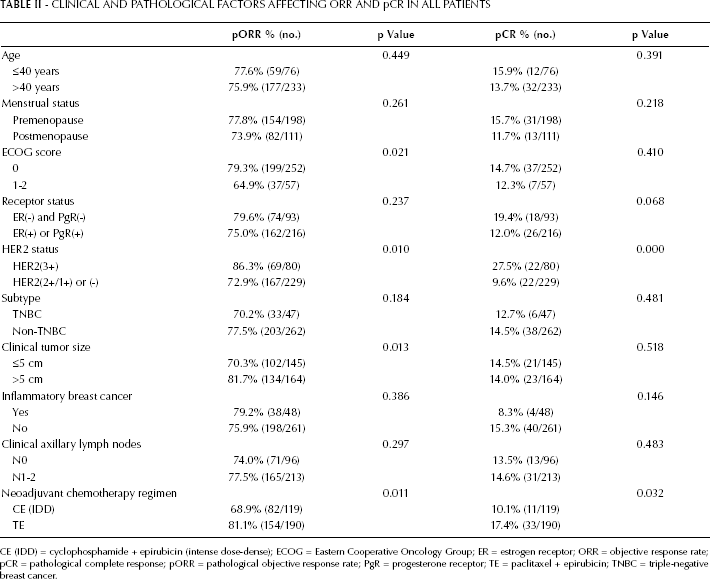
CE (IDD) = cyclophosphamide + epirubicin (intense dose-dense); ECOG = Eastern Cooperative Oncology Group; ER = estrogen receptor; ORR = objective response rate; pCR = pathological complete response; pORR = pathological objective response rate; PgR = progesterone receptor; TE = paclitaxel + epirubicin; TNBC = triple-negative breast cancer.
HER2 OVEREXPRESSION AND NEOADJUVANT CHEMOTHERAPY TREATMENTS
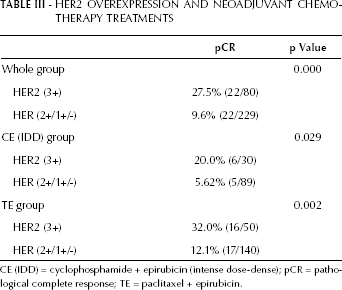
CE (IDD) = cyclophosphamide + epirubicin (intense dose-dense); pCR = pathological complete response; TE = paclitaxel + epirubicin.
Cox regression multivariate analysis showed that pCR in neoadjuvant chemotherapy (NCT), primary tumor size, and axillary lymph node metastasis before neoadjuvant chemotherapy were the important risk factors affecting DFS (Tab. IV). The recurrence risk for patients with pCR decreased 1.12-fold, compared with patients without pCR (5-year DFS: 81.8% vs. 65.7%, p=0.038) (Fig. 1). Stratum analysis indicated that patients with pCR showed more benefit than those without pCR in the HER2 overexpression subgroup (5-year DFS: 86.4% vs. 62.1%, p=0.049) (Fig. 2), but there was no significant benefit in the HER2 lower expression subgroup relative to pCR.
CLINICAL AND PATHOLOGICAL FACTORS AFFECTING DFS (COX MULTIVARIATE ANALYSIS)
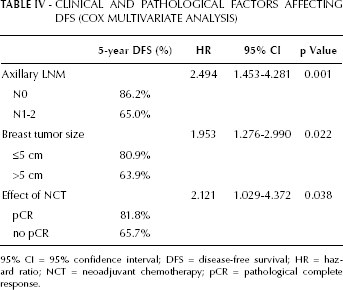
95% CI = 95% confidence interval; DFS = disease-free survival; HR = hazard ratio; NCT = neoadjuvant chemotherapy; pCR = pathological complete response.
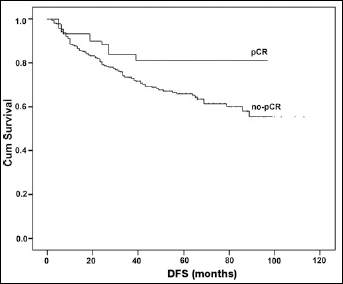
Comparison of disease-free survival (DFS) between patients with different pathological outcomes (pathological complete response [pCR] vs. no pCR).
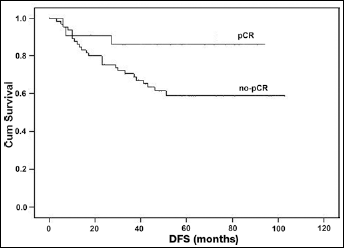
Comparison of disease-free survival (DFS) between HER2 overexpressing patients with different pathological outcomes (pathological complete response [pCR] vs. no pCR).
Discussion
Over the past 10 years, a large number of adjuvant therapy studies have confirmed that patients with HER2-positive breast cancer are more sensitive to anthracycline-based chemotherapies, compared with non-anthracycline medications (7-8-9-10-11), and two thirds of surveyed oncologists prefer an anthracycline-based adjuvant therapy, particularly for node-positive cancers (12). A meta-analysis based on these studies (including 6 clinical trials comparing anthracycline with non-anthracycline as adjuvant therapy) showed that anthracycline-based regimens significantly improved DFS and overall survival (OS) in HER2-positive patients, compared with non-anthracycline regimens, with a 29% decrease in recurrence risk (hazard ratio [HR] = 0.71; 95% confidence interval [95% CI], 0.60-0.83) and a 27% decrease in death risk (HR=0.73; 95% CI, 0.62-0.86). However, for HER2-negative patients, there was no significant difference in DFS between the anthracycline-based and the non-anthracycline chemotherapy groups (13). The response of primary tumors to chemotherapy agents can be observed more directly in neoadjuvant chemotherapies, compared with adjuvant chemotherapies, thereby providing better clinical information for determining the role of biomarkers in predicting treatment efficacy. In general, HER2 overexpression is considered to be an indicator of poor prognosis because of decreased disease-free survival and overall survival (14, 15). On the other hand, the effectiveness of anthracycline-based neoadjuvant therapies has previously been reported to be higher for HER2-overexpressing tumors (16). Our study revealed that patients with HER2 overexpression were more sensitive to neoadjuvant chemotherapies, compared with low HER2 expressing patients (27.5 vs. 9.6% pCR rates, respectively; p<0.001), which we observed in both the CE (IDD) and TE groups. This also underlines recent findings, in which HER2 overexpression is described as an advantage for neoadjuvant chemotherapy efficacy (17, 18). Taken together, our data show that HER2 overexpression in primary breast cancer tumors is a positive correlating factor for the efficacy of neoadjuvant anthracycline-based chemotherapy regimes, and even for HER2-overexpressing patients, pCR is a suitable prognostic factor.
