Abstract
The two most important factors in tumor-stromal interactions are tumor-infiltrating lymphocytes (TIL) and neoangiogenesis (NAng). While changes of these parameters in responders of neoadjuvant chemotherapy (NCTx) have been reported, their correlation with pathological response in breast cancer (BC) patients treated with NCTx have not been described. We therefore evaluated alterations of the TIL subtypes ratio and alterations of NAng using the vasohibin-1-positive ratio (VPR) in BC patients during the course of NCTx. To this aim we used: (i) double immunohistochemistry of CD8 cytotoxic T cells and T regulatory cells (Treg) with Foxp3, determining the CD8+/Foxp3 ratio; (ii) immunostaining of CD31 and vasohibin-1, yielding the VPR, which reflects the NAng status. Changes between the CD8+/Foxp3 ratio and VPR before and after therapy were then correlated with the pathological response of the patients. A concomitant significant decrement of Foxp3 and NAng, represented by VPR, were detected only in NCTx pathological responders (p<0.001 and p=0.044, respectively). The CD8+/Foxp3 ratio increased in both responders and non-responders, but to greater extent in responders (p=0.02). The changes of VPR in the NCTx-treated group differed from those recorded for the patients treated with aromatase inhibitors and shown in our earlier study; this indicates that the reactions of the tumor-stromal interaction to therapy were different among different treatments in BC patients. Changes in Foxp3 and VPR in responders may reflect the dynamic activity of tumor stroma and host immune response to tumor antigens in the tumor microenvironment in response to the NCTx. VPR can be a potential surrogate marker in BC specimens for predicting the response to NCTx, incorporating both features of carcinoma and stromal cells.
Introduction
Tumor-stromal interactions in solid tumors involve a complex of continuous cross-talks, interactions, and equilibriums among different cellular pathways in the tumor microenvironment. These tumor-stromal interactions play pivotal roles in tumorigenesis, development, disease progression, subsequent metastasis, and also tumor response to therapeutic agents (1-2-3-4-5-6-7-8-9-10-11-12-13-14). Two of the main components of these tumor-stromal interactions are neoangiogenesis (NAng) (3-4, 15-16-17-18-19-20-21) and tumor-infiltrating lymphocytes (TIL) (3-4-5-6-7-8-9-10-11-12-13-14), which are frequently co-regulated in both normal, physiological processes, such as inflammation and wound healing, and pathological processes like in tumor-stromal interactions, which are entirely context-dependent processes.
Breast cancer (BC) is one of the well-known angiogenesis-dependent neoplasms, and neo-vascularization and NAng are pivotal in the tumor's development, subsequent invasion, and metastasis (4, 9, 19, 13, 22-23-24). Cross-talks between tumor and endothelial cells occur as a result of the in situ production and secretion of numerous cytokines and growth factors including angiogenic factors (such as vascular endothelial growth factor) that are elevated in their microenvironment (16, 13, 21, 25-26-27-28-29-30-31). These cytokines and growth factors act upon their corresponding receptors present in the endothelial cells and, subsequently, induce angiogenesis. Any disturbances of NAng could result in the progression or cessation of the processes mentioned above (17, 22, 30-31-32-33). The status of in situ angiogenesis in BC tissues has been reported to be correlated with the overall and relapse-free interval of the patients (34-35-36-37-38). We have shown the increased NAng in responders to neoadjuvant aromatase inhibitor (AI) therapy in a group of East Asian postmenopausal estrogen receptor-positive BC patients (39), as reflected by the increase in the vasohibin-1 positive ratio (VPR), derived from the CD31 to vashohibin-1 (VASH-1) levels, in paired pre and post-treatment specimens.
CD31, the most widely used immunohistochemical marker of endothelium, was expressed in both actively proliferating and quiescent endothelial cells (40-41-42). VASH-1 is an endothelium-derived negative feedback regulator of angiogenesis (25, 40, 41, 43-44-45-46-47-48-49) that has been demonstrated as the first physiologically specific suppressor of angiogenesis in humans (25, 43-44-45-46); it acts by inhibiting several endothelial functions relevant to neovascularization in vitro (25, 40, 41, 43-44-45-46-47-48-49). VASH-1 is induced by angiogenic growth factors such as FGF-2, bFGR, and VEGF (via the type-2 VEGF receptor activation and its downstream receptor, PKC-δ) (25, 40, 41, 43-44-45-46-47-48-49). In contrast to CD31, VASH-1 is selectively expressed in proliferating endothelial cells (40-41-42, 45) and its status in particular tissues is directly related to the degree of NAng (40-41-42, 45). Tamaki et al (42, 45) suggested the use of the VPR as an indicator of neovascularization status in human BC.
Of equal importance to NAng is the second component, TIL. Several human neoplasms are infiltrated by immune cells (50-51-52) that have dual roles in tumorigenesis (53, 54). TIL play their roles through the dynamic process of “immunoediting”, which is best summarized by the three “E's” —“elimination”, “equilibrium”, and “escape” (54) —between tumor cells and inflammatory infiltrates. The dynamic balance between these 2 dual actions of immunosurveillance and immunotolerance can determine the outcome (50-51-52-53-54-55). Among the TIL, T cells are considered the main component of the adaptive immune infiltrate/leukocyte pool that can be detected in the tumor tissue (24, 56), with the minor component being that of B lymphocytes. Different immunophenotypes of T lymphocytes have different biologic functions (1) that are known to be involved in the clinical course of individual patients with various solid neoplasms. The most studied components of these T cells are the effector T cells, including CD3+/CD4+, CD3+/CD8+ (CD8+), CD4+ CD25+ T regulatory (Treg) cells, and other cell types like the CD4+ helper lymphocytes (1). The CD8+ subset constitutes the cytotoxic T lymphocytes that lead to the direct destruction of the tumor cells. A high T-cell infiltration with the CD8+ subset in the primary tumor tissue was reported to be associated with better clinical outcome of the patients with ovarian cancer and other malignancies (57-58-59). On the other hand, the Treg subset was reported as a regulator or suppressor of the effector T lymphocytes (28-29-30-31-32-33, 60-61-62-63-64-65) suppressing the proliferation of other T cells subtypes, including CD8+ in the tumor milieu, through contact-dependent mechanisms or cytokines such as IFN-γ (61-62-63-64, 66), IL-10, and TGF-β (29-32, 35, 36, 61-62-63-64, 67, 68).
Foxp3 is considered a master regulatory gene for lineage commitment and/or development of Tregs (69) and is related to their function. Foxp3 was also demonstrated to be specific for the regulatory T-cell lineage with suppressor activity (69) and was considered the most important marker identifying Tregs (70). Among these T lymphocytes in the tumor tissue, the CD8+/Foxp3+ ratio, but not the absolute number of CD8+ T cells or Tregs, was recently shown to display a more impressive hazard ratio (HR) (71-72-73) in predicting the clinical outcome of the patients (71, 72). Previous studies in BC reported the changes of TIL before and after chemotherapy (59, 74, 75). We previously reported the parallel increase in CD8+/Foxp3 ratio and NAng, as reflected by the VPR, in responders undergoing neoadjuvant endocrine therapy with AI in East Asian postmenopausal ER-positive BC patients (76). To the best of our knowledge, alterations in the subtypes' composition of in situ TIL and their correlation with concomitant changes of NAng and treatment response in BC patients during the course of neoadjuvant chemotherapy have not been reported. Therefore, in this study, we evaluated the changes of TIL subtypes using double immunohistochemistry for CD8 and Foxp3 in a cohort of operable BC patients treated with neoadjuvant chemotherapy. Results were then correlated with changes of NAng, evaluated by the changes in the VPR, as well as with the pathological response of individual patients based on the alterations of the histological transformations in the stroma and carcinoma cells.
STUDY DESIGN
Patients and methods
A total of 100 Japanese patients with primary breast invasive ductal carcinoma referred to the Department of Surgery, at the Tohoku University between 2003 and 2012 were included in the analysis. Approval for performing this translational research was obtained from the institutional review board at the Tohoku University.
Breast tissue specimens
The specimens available for this study were core needle biopsies of the treatment-naïve tumor and surgical pathology specimens obtained after the treatment at the time of surgery. All biopsy and surgical specimens were fixed in 10% formalin for, respectively, 12-24 hours and 24-48 hours. Pre and post-treatment specimens of all these 100 patients were available for the study of the pathological response, immunohistochemical evaluation, ER, PgR, and HER2 status.
Neoadjuvant chemotherapy treatment regimen
All the subjects were given either (i) Canadian CEF: cyclophosphamide 75 mg/m2 orally from day 1 to 14, epirubicin 60 mg/m2 intravenously on days 1 and 8, and fluorouracil 500 mg/m2 intravenously on days 1 and 8, every 28 day for 6 cycles; or (ii) FEC 100: fluorouracil 500 mg/m2, epirubicin 100 mg/m2, and cyclophosphamide 500 mg/m2 every 21 days for 4 cycles; with or without subsequent docetaxel 75 mg/m2 intravenously on day 1 every 21 days for 4 cycles.
Immunohistochemistry
1. Immunohistochemical staining
a. Tumor infiltrative lymphocytes — CD8+ and Foxp3+
The distribution of the subtypes of tumor infiltrating T lymphocytes was evaluated by the immunoreactivities to CD8 and Foxp3, using double immunostaining for the 2 markers in paired pre and post-treatment specimens.
Double immunostaining for Foxp3 and CD8 was performed with the immunohistochemical staining of Foxp3 followed by that of CD8. The methods and the antibodies used for the double immunohistochemical staining of the individual biomarkers for CD8 and Foxp3 were those previously reported in details by Chan et al (76), Liu et al (77), and Fu et al (78). In brief, serial tissue sections (3 μm) were prepared for hematoxylin-eosin staining and immunohistochemistry of CD8 and Foxp3. The specimens were deparaffinized in xylene, and sequentially hydrated with graded solutions of alcohol and distilled water. Endogenous peroxidase activity was inhibited by 3% hydrogen peroxidase for 10 minutes at room temperature. Immunohistochemical staining was performed with the streptavidin-biotin amplification method using a Histofine Kit (Nichirei Bioscience, Tokyo, Japan).
b. Neoangiogenesis — VASH-1 and CD31
We evaluated the immunoreactivities of CD31 and VASH-1 (Fig. 2). The degree of NAng was assessed by the pre and post-treatment VPR; VPR was defined as the ratio between VASH-1 and CD31-positive counts obtained at the same hot spots in the same fields of the slides, as described in previous studies (39, 42). The details of the immunohistochemistry technique for CD31 and VASH-1 were previous described by Tamaki et al and Chan et al (39, 42). Also, the methodology and the list of antibodies used for immunohistochemical staining of different biomarkers were previously reported in details by Tamaki et al and Chan et al (39, 42). In brief, serial tissue sections (4 μm) were prepared from selected pre and post-treatment blocks from the chemotherapy-treated cohort for hematoxylin-eosin staining and immunohistochemistry for CD31 and VASH-1. The specimens were deparaffinized in xylene, and sequentially hydrated with graded solutions of alcohol and distilled water. Endogenous peroxidase activity was blocked by 3% hydrogen peroxidase for 10 minutes at room temperature. Immunohistochemical staining was performed with the streptavidin-biotin amplification method using a Histofine Kit (Nichirei Bioscience, Tokyo, Japan).
2. Evaluation of immunoreactivity
The scoring of the ER, PgR, and HER2 immunoreactivity was evaluated by one of the authors (NN) who was blinded to the treatment regimens and clinical course of the patients. The ER and PgR scorings were determined by assigning both proportion and intensity scores, referring to the Allred's procedure. The membrane's staining pattern was estimated for HER2 immunohistochemistry and scored on a scale of 0-3.
The immunoreactivities of CD8+ and Foxp3+ T lymphocytes, CD31, and VASH-1 were independently evaluated by 4 of the authors (MC, LW, SC, and SF), all blinded to the treatment response of the patients. In brief, for each biomarker 3 different hot spots in each of the specimens in the paired pre and post-treatment groups were selected for the assessment. The immunoreactivity of Foxp3+ and CD8+ T lymphocytes, CD31, and VASH-1 were assessed by counting the number of cells within the 3 designated hot spots where the most abundant positively stained cells were identified. This was done using an eyepiece graticule under light microscopy at 400 high-power field, with an Olympus BX50 (Olympus, Tokyo, Japan), using the 20X and 40X objectives. The average numbers counted by each investigator were used for subsequent data analysis.
3. Pathological response
The pathological response was assessed by using the hematoxylin-eosin staining according to the histopathological criteria for the assessment of therapeutic response in BC (The Japan Breast Cancer Society, 2007 version) (79). Three of the authors (MC, SF, and NN), all blinded to the treatment response of the patients, assessed the pathological response using the paired pre and post-treatment specimens.
The pathological response criteria for classification were:
Grade 0 = no response
Grade 1a and 1b = slight response (respectively mild and moderate)
Grade 2 = marked response
Grade 3 = complete response (pCR)
According to the grading of the pathological response, the subjects in the whole group were further classified into 2 groups: responders (including those of grades 2 and 3), and non-responders (including those of grades 0, 1a, and 1b).
Statistical Analysis
Statistical analyses were performed using the SPSS 13.0 statistical analysis software package (IBM Corporation, Route 100, Somers, NY 10589, USA). The non-parametric Mann—Whitney U test was used to determine the homogeneity of the whole group and the different pre-treatment biomarkers in the responders and non-responders group. The Wilcoxon paired non-parametric test was used to determine the statistically significant difference of the mean changes of IHC scores of the individual biological markers including the CD8+, Foxp3+, CD31, and VASH-1, as well as the CD8+/Foxp3+ ratio, and the VPR between pre and post-treatment specimens. For comparing the changes in VPR or CD8+/Foxp3 ratio between the pathological responder and non-responder group, the Mann-Whitney non-parametric test was used. A P value <0.05 was considered statistically significant.
Results
Pre-treatment status of the biological markers between responders and non-responders
Except for PgR, there were no other statistically significant differences in the immunopositivity of all pre-treatment biomarkers between the 2 subgroups in the pre-treatment cohort. No statistically significant differences in the pre-treatment CD8+/Foxp3 ratio or VPR were detected between responders and non-responders. The details are summarized in Table I.
COMPARISON OF THE SINGLE PRE-TREATMENT BIOMARKERS' LEVELS BETWEEN PATHOLOGICAL RESPONDERS AND NON-RESPONDERS
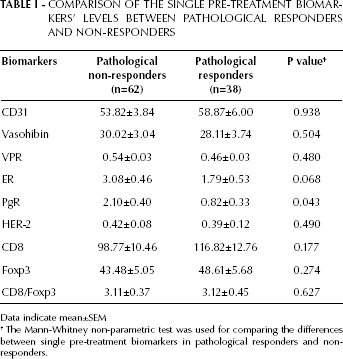
Data indicate mean±SEM
The Mann-Whitney non-parametric test was used for comparing the differences between single pre-treatment biomarkers in pathological responders and non-responders.
Using the Mann-Whitney non-parametric test we found no significant effects of positive or negative pre-treatment status of ER/PgR/HER-2 on the changes of VPR or CD8+/Foxp3 ratio; for this analysis we first compared the patients positive for the 3 biomarkers (ER/PgR/HER-2) to those biomarker-negative, and then we compared the pathological responders to the non-responders (data not shown).
Analysis of alterations of different biomarkers with respect to treatment response
After NCTx treatment, a significant decrease in Foxp3 was only detected in the group of pathological responders (p<0.001) (Tab. II, Figs. 1A and 1B).
COMPARISON OF THE SINGLE PRE AND POST-TREATMENT BIOMARKERS' LEVELS BETWEEN RESPONDERS AND NON-RESPONDERS
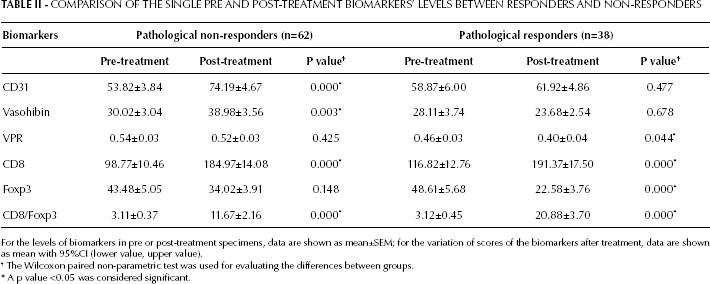
For the levels of biomarkers in pre or post-treatment specimens, data are shown as mean±SEM; for the variation of scores of the biomarkers after treatment, data are shown as mean with 95%CI (lower value, upper value).
The Wilcoxon paired non-parametric test was used for evaluating the differences between groups.
A p value <0.05 was considered significant.
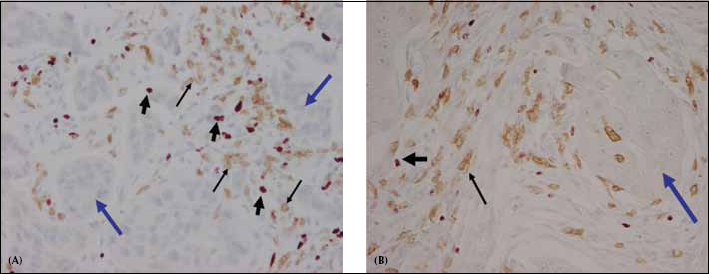
Double immunostaining for CD8 (thin arrows) and Foxp3 (thick black short arrows) in both the pre and post-treatment specimens showing infiltration of both types of T cells at the peri-tumoral area around breast cancer cells (blue arrow). Of note is the relative abundance of Foxp3+ cells compared with CD8+ cells in the pre-treatment specimens (A), which is in contrast with the relative abundance of CD8+ cells to Foxp3+ cells in the post-chemotherapy specimens (B).
A significant decrement of VPR was only detected in pathological responders (p=0.044) (Tab. II, Figs. 2A and 2B).
Representative illustrations of the immunohistochemical findings of CD31 and VASH-1 in 2 consecutive sections of one of the post-chemotherapy breast carcinoma specimens examined. (A) Post-treatment specimen at one of the hot spots, showing positive staining of the vessels for CD31 (black, yellow, and red arrows); note the moderate-to-intense staining of the small-sized vessels (red arrow) in the background of the medium and large-sized vessels (yellow and black arrows, respectively). (B) Immunohistochemical VASH-1 staining in consecutive slides at the same site of the hot spot as that of CD31: the staining shows that not all vessels that were positive for CD31 were also positive for VASH-1. The colors of the arrows correspond to those indicated for Figure 2A: the VASH-1 positively-stained vessels were all small-sized vessels (red arrow). The medium and large size vessels were not stained by VASH-1 but were positive for CD31 (yellow and black arrows).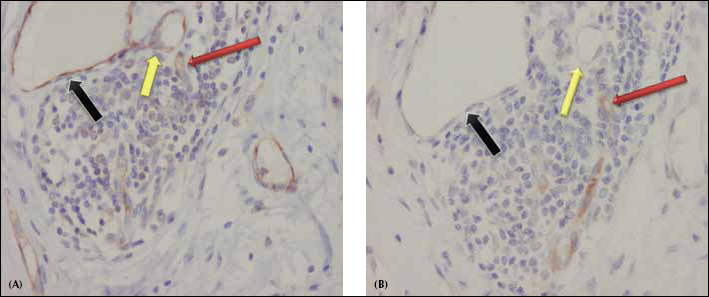
A difference in terms of change of VPR between responders and non-responders was detected but did not reach statistical significance (p=0.068) (Tab. III).
COMPARISON OF THE CHANGES IN VPR AND CD8+/FOXP3 RATIO BETWEEN PATHOLOGICAL RESPONDERS AND NON-RESPONDERS
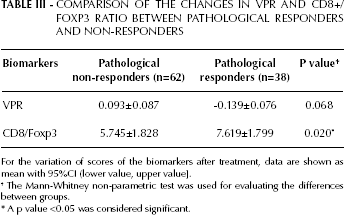
For the variation of scores of the biomarkers after treatment, data are shown as mean with 95%CI (lower value, upper value].
The Mann-Whitney non-parametric test was used for evaluating the differences between groups.
A p value <0.05 was considered significant.
Both the CD8+ cells and the CD8+/Foxp3 ratio increased in both pathological responders and non-responders, although the change was significantly higher in the responders' group (p=0.020) (Tab. III).
The changes of VPR and CD8+/Foxp3 were inversely correlated (r=-0.244, p=0.015, calculated with the Spearman correlation test, data not shown) (Fig. 3A).
(A) The variations of VPR and of CD8/Foxp3 ratio were significantly negatively correlated (r=-0.244, p=0.015). (B) The low pre-treatment CD31 was correlated with a better OS (p=0.042). (C) A high pre-treatment CD8+/Foxp3 ratio was correlated with a better DFS (p=0.027).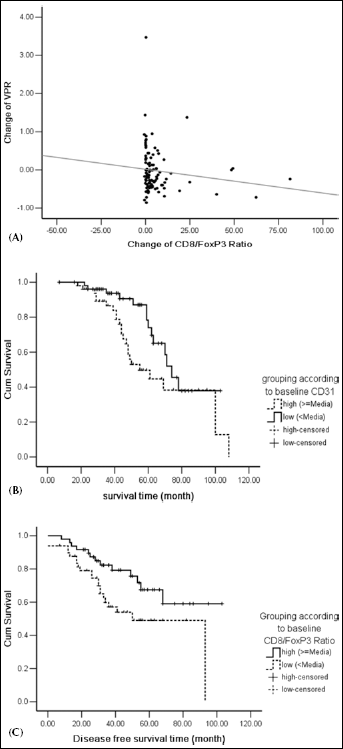
Low pre-treatment CD31 levels were significantly correlated with a better overall survival (OS) (p=0.042) (Fig. 3B). A high pre-treatment CD8+/Foxp3 ratio was significantly correlated with a better disease-free survival (DFS) (p=0.027) (Fig. 3C). However, the VPR and the changes in VPR were not correlated with the OS or DFS of the patients examined in this study.
Discussion
In BC, the tumorigenesis and disease progression/metastasis processes, as well as the response of the tumor to therapeutic agents are all closely correlated. These correlations are mediated by complex cross-talks and equilibriums involving different cellular pathways within the tumor micro-milieu (3, 4, 6, 7, 9, 11-12-13, 15-16-17-18-19-20) and include the interactions among different cell types including fibroblasts, lymphatics, endothelial cells, inflammatory cells, and different types of mediators such as cytokines, chemokines, and products of cellular metabolism (1-2-3-4-5-6-7-8-9-10-11-12-13). Two of the most important carcinoma-stromal cell interactions involved in this cascade are NAng and TIL.
BC is one of the well-known angiogenesis-dependent diseases. Any off-balance of NAng can determine the progression or arrest of the processes mentioned above in the tumor (7, 9, 11-12-13, 17-18-19-20, 22-23-24, 30-31-32-33). The status of the in situ angiogenesis in BC tissues has been reported to be correlated with the patients' overall and relapse-free interval (34-35-36-37-38). The results reported for the analysis of angiogenesis actually considered the static vascularity in terms of microvascular density (MVD) using immunohistochemistry for CD34 or other endothelial markers (34-35-36-37-38, 80), which can by no means demonstrate the actual degree of NAng. Tamaki et al (42) clearly demonstrated that tumor NAng could be assessed by the ratio between VASH-1 and CD31, and the VPR assessed in surgical pathology materials. An increased VPR and CD8+/Foxp3 ratio during neoadjuvant endocrine treatment was also reported to predict a better response to AI (76).
An involvement of specific immunophenotypes in the clinical course of individual patients has been reported. Elevated T-cell infiltration with the CD8+ subset (cytotoxic T lymphocytes) in the primary tumor tissue was reported to be associated with better clinical outcome in some malignancies such as ovarian cancer (27, 45, 69, 75). Demaria et al also reported that response to neoadjuvant chemotherapy was also associated with a significant increment of the CD8+/Foxp3 ratio (74), while Ladorie et al reported the disappearance of the Foxp3+ Tregs following chemotherapy (27, 45, 59). We also reported a parallel increase in the CD8+/Foxp3 ratio with increased VPR on estrogen depletion with steroidal AI therapy (76). However, whether VPR and the CD8/Foxp3 ratio together could be used as outcome predictors of neoadjuvant chemotherapy in BC patients still requires further investigations.
In the present study, both the pathological responders and non-responders had an increase in their CD8 and CD8+/Foxp3 ratio, a result that is different from that reported in other studies, such as that of Demaria et al, in which the increased ratio was detected only in the pathological complete responders' group (74). However, in previous studies by Ladorie et al the decreased Foxp3 expression was only detected in the pathological responders' group, while Foxp3 completely disappeared in the pathological complete responders (59). Therefore, the results of our study indicate that only the changes in Foxp3 could be involved in the achievement of a pathological response in chemotherapy, and thus Foxp3 can be considered more important than CD8 in the spectrum of tumor immunology. In addition, we demonstrated that the baseline CD8+/Foxp3 ratio and the patients' OS were positively correlated. We also noted a significant correlation between higher pre-treatment CD8+/Foxp3 ratio and better DFS, as well as the lack of a correlation between baseline status of Foxp3, CD8, and VPR. We showed that the changes of VPR and CD8+/Foxp3 ratio were inversely correlated, and that a high CD8+/Foxp3 ratio was correlated with a better patients' DFS. However, no significant correlation between the baseline VPR or VPR changes and DFS was detected in our study. This discrepancy is probably due to possible different effects of the TIL subtypes and VPR on tumor cells and the tumor microenvironment in an independent fashion, possibly involving specific pathways over a different time course including long-term prognosis of the patients. Hence, even the VPR and CD8+/Foxp3 ratio were significantly and inversely correlated during the initial treatment phase; their effects over time could possibly be different and, in turn, might have different consequences on the long-term course of the disease. When considering the correlation between TIL and the treatment response and survival in previous studies, the ratio between the different subtypes of tumor infiltrative lymphocytes was reported more important and determinant than the actual quantity of the individual TIL subtypes. The results of our study are interesting in this sense since Foxp3, CD8, and VPR were by no means statistically correlated with the DFS or OS of our patients. However, when considering the overall baseline ratio of CD8+/Foxp3, representing the overall/combination of the effects of the 2 factors, we detected a statistically significant correlation. The ratio between the TIL subtypes is therefore considered to reflect more the effector-to-suppressor ratio, which is supposed to be dynamic during the disease course; besides, any change towards one of the 2 factors could subsequently result in different patients' response to treatment in terms of tumor killing. Also, these findings further suggest that the ratio of the TIL subtypes is more determinant in the spectrum of tumor immune reactions. The status of the parameters reflecting the baseline tumor immune reaction could therefore determine the overall prognosis, regardless of the response or non-response to chemotherapy.
The findings in our study also indicated that the baseline status of ER/PgR/HER-2 did not affect the alteration of TIL and NAng in the setting of neoadjuvant chemotherapy.
In our study, a decrease in NAng was correlated to treatment outcome in response to chemotherapy, a finding that is in contrast to that shown in our previous study in the endocrine therapy group for which we observed an increase in VPR in the responders' group. The decrease in VPR and the suppressed or lowered NAng both play important roles in the biological behavior of the tumor in patients treated with chemotherapy. The difference in the variations of NAng within the chemotherapy group in contrast to the endocrine therapy group reported in our previous study suggest that the tumor-stromal reaction is different in these 2 groups. This disparity could be possibly due to the potentially different tumor-stromal interactions involved in the mechanisms of tumor killing typically activated by the 2 groups, that is, direct cytotoxic in the chemotherapy group versus estrogen depletion resulting in apoptosis and/or tumor cell death in the endocrine therapy group. However, further investigations are required for clarification. Chemotherapeutic agents exert direct cytotoxic effects not only on tumor cells but also on other cells including endothelial cells and tumor infiltrative lymphocytes. The response to chemotherapy treatment may also include direct cytotoxic effects on the tumor infiltrative lymphocytes and also on endothelial cells, thus resulting in their death and decreased proliferation, as reflected by the decreased VPR. This is a physiological process/response of the body in an attempt to dispose dying/necrotic carcinoma cells from the body in order to achieve a homeostasis of the stromal microenvironment and hence the immunological tolerance of the host (81) upon carcinoma cell death. Therefore, the dying tumor cells may possibly be more “immunogenic” and less “immuno-tolerant” with the decrease of Tregs in the tumor milieu. Whether the response to chemotherapy results in immunomodulation of the tumor-infiltrating lymphocytes (as suggested by the decrease in Foxp3 in the responders' group) remains to be validated. Considering the small number of cases in our cohort, further large-scale studies are required to provide more insight into the role of tumor immunobiology in chemotherapy.
On the other hand, endocrine therapy with AI resulted in estrogen depletion, which suppresses ER-positive tumor cell proliferation, resulting in an induction of apoptosis (82), and subsequent death of these carcinoma cells (50, 66, 69-73, 83-89). These changes subsequently induced an antinflammatory environment around the dying/dead tumor cells through the secretion of antinflammatory cytokines and the subsequent expression of certain growth and survival factors such as VEGF and TGF-ß (90-91-92). In the areas adjacent to the necrotic foci was demonstrated an increment in both VEGF and angiogenesis stimulators (90, 91), as well as a downregulation of the angiostatic genes and an upregulation of pro-angiogenic genes, which could promote proliferation of endothelial cells resulting in NAng around the necrotic areas upon tumor cell death (90-91-92). In contrast with pathological angiogenesis sustaining tumor growth and progression (8, 10, 11, 93-94-95-96-97-98), the increase in NAng following AI treatment was suggested to possibly represent part of the more physiological process/response of the body in an attempt to dispose dying/necrotic carcinoma cells from the body in order to achieve a hemostasis of the stromal microenvironment and hence immunological tolerance of the host (81, 92). However, our study suggests the lack of a direct correlation between the status of NAng and the most important factor in tumor immunology. The VPR, reflecting the process of NAng, and Foxp3, the most important regulatory factor in tumor immunology, were not correlated to each other in their basal status, neither were their changes before and after chemotherapy. These 2 factors probably exert their effects upon the clinical course or biological behavior of the patients in an independent fashion, which might involve different cellular pathways that need to be investigated by further studies. Subjects having tumors with less suppressed killer T-cell activity by Tregs/Foxp3, as well as with low NAng properties, were postulated to have the better prognosis.
In summary, the response to chemotherapy resulted in a significant decrement of both NAng and Foxp3 accompanied by a significant increment in the CD8+/Foxp3 ratio through possible complex interaction of the tumor-stromal microenvironment in response to the apoptosis/death of the tumor cells. These results also indicate that not only the features of carcinoma cells but also those of adjacent stromal cells in the tumor microenvironment should be considered when assessing the response to the chemotherapeutic agent.
Footnotes
Acknowledgements
The authors appreciated the technical assistance of K. Ono, Department of Pathology, Tohoku University School of Medicine, Sendai, Japan.
