Abstract
Introduction
Breast cancer is a leading cause of cancer-related death in women worldwide, and its metastasis is a major cause of disease mortality. Therefore, identification of the mechanisms underlying breast cancer metastasis is crucial for the development of therapeutic and diagnostic strategies. Our recent study of immunodeficient female mice transplanted with MDA-MB231 breast cancer cells demonstrated that tumor cell-derived angiopoietin-like protein 2 (ANGPTL2) accelerates metastasis through both increasing tumor cell migration in an autocrine/paracrine manner, and enhancing tumor angiogenesis. To determine whether ANGPTL2 contributes to its clinical pathogenesis, we asked whether serum ANGPTL2 levels reflect the clinical features of breast cancer progression.
Methods
We monitored the levels of secreted ANGPTL2 in supernatants of cultured proliferating MDA-MB231 cells. We also determined whether the circulating ANGPTL2 levels were positively correlated with cancer progression in an in vivo breast cancer xenograft model using MDA-MB231 cells. Finally, we investigated whether serum ANGPTL2 levels were associated with clinical features in breast cancer patients.
Results
Both in vitro and in vivo experiments showed that the levels of ANGPTL2 secreted from breast cancer cells increased with cell proliferation and cancer progression. Serum ANGPTL2 levels in patients with metastatic breast cancer were significantly higher than those in healthy subjects or in patients with ductal carcinoma in situ or non-metastatic invasive ductal carcinoma. Serum ANGPTL2 levels in patients negative for estrogen receptors and progesterone receptors, particularly triple-negative cases, reflected histological grades.
Conclusions
These findings suggest that serum ANGPTL2 levels in breast cancer patients could represent a potential marker of breast cancer metastasis.
Introduction
Breast cancer is the most frequently diagnosed malignant neoplasm in women and is a leading cause of cancer-related death worldwide (1). Breast cancer mortality after therapeutic intervention is primarily due to metastasis of tumor cells from the primary lesion (2, 3). Therefore, identification of the molecular mechanisms driving breast cancer metastasis is important in order to develop effective therapeutic or early diagnostic strategies to treat this condition.
Recently, chronic inflammation has received much attention as playing a key role at various stages of tumor development, including initiation, growth, invasion, and metastasis (4). In this regard, we recently reported that angiopoietin-like protein 2 (ANGPTL2) increases inflammatory carcinogenesis (5). Furthermore, our xenograft studies in female mice transplanted with MDA-MB231 breast cancer cells demonstrated that tumor cell-derived ANGPTL2 accelerates tumor metastasis by increasing tumor cell migration and enhancing tumor angiogenesis (6). However, the role of ANGPTL2 in the clinical pathogenesis of breast cancer remains uncharacterized.
To investigate this question, we asked whether serum ANGPTL2 levels paralleled clinical features of breast cancer progression. We found that circulating ANGPTL2 levels in xenograft mouse models increased according to cancer progression, particularly in the presence of metastasis. Serum ANGPTL2 levels in patients with advanced-stage breast cancer were significantly higher than those observed in healthy subjects or patients with ductal carcinoma in situ (DCIS) or non-metastatic invasive ductal carcinoma (IDC). In patients negative for estrogen receptors and progesterone receptors, serum ANGPTL2 levels were positively correlated with nuclear grade status. Our findings show that serum ANGPTL2 levels in hormone receptor (HR)-negative patients, and especially triple-negative (TN) ones, might reflect the clinical features of breast cancer.
Materials and Methods
Quantitation of the ANGPTL2 protein by ELISA
The ANGPTL2 concentrations in culture medium from tumor cells or in serum from humans or mice were estimated using the ANGPTL2 assay kit (IBL, Gunma, Japan) as previously described (7, 8).
Human studies
We collected blood samples from breast cancer patients (n=625) at Kumamoto University between May 2003 and July 2011, as well as from healthy female volunteers (n=19) aged 20-54 years working at Kumamoto University. All patients were examined to evaluate potential tumor spread in the whole body using mammography, ultrasound sonography, and computed tomography (CT) at the time of initial diagnosis. Stage IV patients and subjects with relapse after initial treatment (such as surgery and adjuvant treatment) also received bone scintigraphy or positron emission tomography to search for additional lesions. Diagnosis of solitary lesions resulting from potential relapse and/or metastasis was undertaken using fine needle aspiration biopsy and core needle biopsy to confirm metastasis.
Blood samples from breast cancer patients without distant metastasis were obtained immediately before surgery or at the time of initial systemic treatment with chemotherapy, anti-HER2 therapy, or endocrine therapy. Serum samples of patients with distant metastasis were obtained before initial treatment. Blood samples were centrifuged at 3,000 rpm for 10 minutes, and supernatants were stored at -80°C prior to assay. This study was approved by the ethics committees of the Kumamoto University. Written informed consent was obtained from each subject.
Cell lines and culture
Human breast adenocarcinoma cell lines MCF-7, T47D, SK-BR-3, MDA-MB453, and MDA-MB231 were purchased from the American Type Culture Collection (ATCC) and cultured in conditions recommended by the supplier. Luciferase-expressing MDA-MB231 cells (MB231/luc) were generated and maintained as described (6). To detect ANGPTL2 concentrations in culture medium, 5,000 cells were plated in each well of a 96-well plate (Iwaki, Tokyo, Japan) with 100 μL medium; after the first plating, the medium was then assayed at specific time points using an ANGPTL2 assay kit (IBL, Gunma, Japan).
Real-time quantitative RT-PCR
Total RNA from cultured cells was isolated using TRIzol reagent (Invitrogen, Carlsbad, CA). DNase-treated RNA was reverse transcribed using a PrimeScript RT Reagent Kit (Takara Bio, Otsu, Japan). PCR products were amplified using a Thermal Cycler Dice Real Time system (Takara Bio, Otsu, Japan), and the relative transcript abundance was normalized to that of the 18S mRNA. PCR oligonucleotides were as follows: human ANGPTL2, forward, 5′- GCCACCAAGTGTCAGCCTCA-3′, reverse, 5′- TGGACAGTACCAAACATCCAACATC-3′: human 18S, forward, 5′- TTTGCGAGTACTCAACACCAACATC-3′, reverse, 5′- GAGCATATCTTCGGCCCACAC-3′. The data was analyzed with the 2-tailed Student's t-test using Excel software (Microsoft, Redmond, WA). A p value of less than 0.05 was considered statistically significant.
Proliferation assay
A total of 5,000 cells per well were plated in 96-well plates (Iwaki, Tokyo, Japan), and cell number was determined at specific time points using a Cell Counting Kit-8 (Dojindo, Kumamoto, Japan) according to the manufacturer's protocol. The growth rate was expressed as the number of cells at a specific time point relative to that determined at 0 hours.
Mouse xenograft model
For the breast cancer xenograft model, we used MB231/luc cells implanted in the mammary fat pad of an 8-week-old female JAK3-deficient NOD-SCID (NOJ) mice (6), and we monitored bioluminescence images as previously described (6). All experiments were performed according to the guidelines of the Institutional Animal Committee of Kumamoto University.
Statistics
Cell proliferation and ANGPTL2 levels in the culture medium were analyzed using the Kruskal-Wallis test. Serum ANGPTL2 levels were analyzed using the Steel-Dwass test. All calculations were performed using the JMP10 software (SAS Institute, Cary, NC, USA). A p value of less than 0.05 was considered statistically significant.
Results
Proliferating ANGPTL2-expressing cells secrete ANGPTL2 protein in vitro
To examine whether tumor cell-secreted ANGPTL2 concentrations increase with tumor cell proliferation, we employed an enzyme-linked immunosorbent assay (ELISA) to measure ANGPTL2 protein levels in the medium of cultured human breast cancer MDA-MB231 cells, which abundantly express ANGPTL2 (6). As expected, MDA-MB231 cells proliferated over time (Fig. 1A) and ANGPTL2 concentrations in the medium increased with proliferation (Fig. 1B). Next, we calculated the ratio between ANGPTL2 concentration and proliferation rate and found that ANGPTL2 levels increased from day 1 to day 2 but remained unchanged from day 2 to day 3 (Fig. 1C). We also examined ANGPTL2 mRNA induction and observed a trend similar to that of the secreted ANGPTL2 protein (Fig. 1D). Thus, cultured breast cancer cells secrete ANGPTL2 as they proliferate, and induction is more apparent at early proliferative stages.
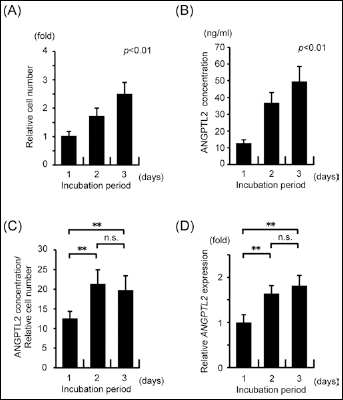
MDA-MB231 breast cancer cells secrete ANGPTL2 in vitro. (A) Relative number of proliferating MDA-MB231 cells at the indicated time points of the in vitro culture (4 independent experiments, each containing 4 replicates). On day 1 of cell culture the number of proliferating cells was set at 1. (B) ANGPTL2 protein concentration in the culture medium at the indicated days (4 independent experiments, each containing 4 replicates). (C) The ratio between ANGPTL2 concentration and relative cell number. (D) Comparative ANGPTL2 expression levels of MDA-MB231 cells at the indicated time points (4 independent experiments, each containing 2 replicates). mRNA levels on day 1 of cell culture were set at 1. Error bars show SD. **p<0.01; n.s., not statistically significant.
Serum ANGPTL2 levels increase in parallel with metastasis in a xenograft mouse model
Next, we analyzed whether circulating tumor-cell secreted ANGPTL2 levels reflect cancer progression in vivo in a breast cancer mouse model. To do so, we first examined the ANGPTL2 mRNA levels in various breast cancer cell lines, including MCF-7 (ER+, HER2-), T47D (ER+, HER2-), SK-BR-3 (ER-, HER2+), MDA-MB453 (ER-, HER2+), and MDA-MB231 (ER-, HER2-) (Fig. 2A). As we previously reported (6), ANGPTL2 induction is more robust in MDA-MB231 cells than in other breast cancer cell lines. Therefore, we implanted MDA-MB231 cells harboring a luciferase expression vector (MB231/luc) into the mouse mammary fat pad of JAK3-deficient NOD-SCID (NOJ) mice (7) and then monitored bioluminescence images and serum ANGPTL2 concentrations. Tumors in the mammary fat pad grew gradually, based on bioluminescence analysis, and lung metastasis was observed at 9 weeks after the xenograft (Fig. 2B). Interestingly, serum ANGPTL2 levels in these mice increased significantly once lung metastasis was detected (Fig. 2C). These results suggest that serum ANGPTL2 levels in vivo parallel tumor progression, particularly metastasis.
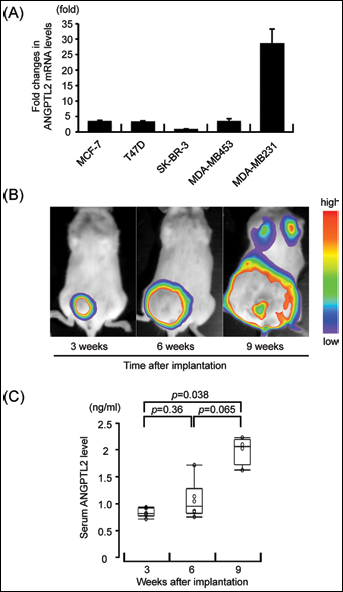
ANGPTL2 levels are elevated with metastasis in a xenograft mouse model of human breast cancer. (A) Comparative ANGPTL2 expression levels in various breast cancer cell lines. SK-BR-3 mRNA levels were set at 1. Bars indicate means ± ranges (of 2 independent experiments). (B) Representative bioluminescence images of mice bearing MB231/luc cells. Images were taken at 3, 6, and 9 weeks after implantation (n=6). (C) Serum ANGPTL2 concentrations at the same time points after subcutaneous injection with MB231/luc cells (n=6). Serum ANGPTL2 concentrations were determined by ELISA.
Serum ANGPTL2 levels in breast cancer patients correlate with tumor development
Based on our findings that circulating tumor cell-secreted ANGPTL2 levels are associated with metastasis in mice, we asked whether serum ANGPTL2 levels reflect tumor progression in breast cancer patients. To do so, we compared serum ANGPTL2 levels from breast cancer patients with those from healthy volunteers (Tab. I, Fig. 3). ANGPTL2 concentrations in patients diagnosed at an advanced breast cancer stage were significantly higher than those seen in healthy subjects or in patients with DCIS or IDC (Fig. 3). These results suggest that serum ANGPTL2 levels correlate with breast tumor progression and metastasis in patients.
PATIENT CHARACTERISTICS
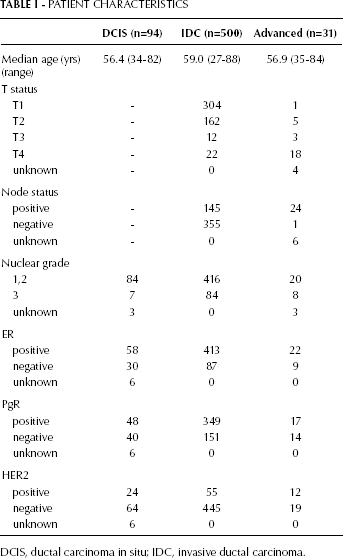
DCIS, ductal carcinoma in situ; IDC, invasive ductal carcinoma.
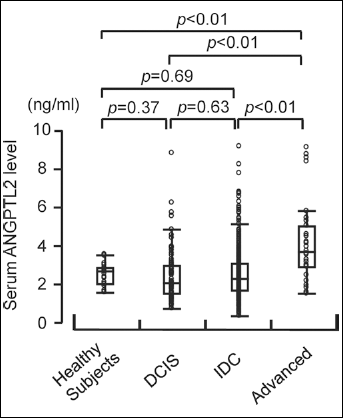
Serum ANGPTL2 levels in cancer patients are positively correlated with tumor progression. Serum ANGPTL2 concentrations in healthy volunteers (n=19) and breast cancer patients with ductal carcinoma in situ (DCIS, n=94), invasive ductal carcinoma (IDC) without metastasis (n=500), and advanced breast cancer (n=31).
Serum ANGPTL2 levels reflect histological grades in HR-negative patients
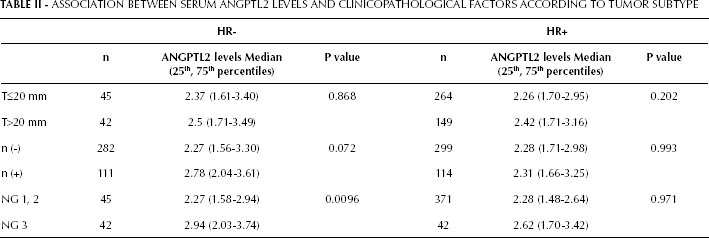
MDA-MB231 cells are a triple-negative (TN) line, and we showed that serum ANGPTL2 levels reflect tumor progression in an MDA-MB231-xenografted mouse model. Therefore, we asked whether serum ANGPTL2 levels would differ in terms of metastasis in patients who were either HR-positive or negative. We primarily focused on IDC patients, and we first examined the potential association between serum ANGPTL2 levels and clinicopathological features, such as tumor size (T), nodal status (n), and nuclear grade (NG), according to HR status (Tab. II). In HR-positive (ER+ and/or PgR+) patients, serum ANGPTL2 levels showed no association with clinicopathological factors. In HR-negative (ER- and/or PgR-) patients, serum ANGPTL2 levels did not differ with tumor size. However, serum ANGPTL2 levels in HR-negative patients with lymph node metastasis tended to be higher than in patients without lymph nodes metastasis. In addition, serum ANGPTL2 levels in HR-negative patients diagnosed as NG 3 were significantly higher than those classified as NG 1 or 2, indicating that serum ANGPTL2 levels in HR-negative patients reflect NG status. Next, we examined the potential association between serum ANGPTL2 levels and clinicopathological factors based on tumor subtype as determined by immunohistochemistry (luminal: HR+ and HER2-; HER2+: HER2+, irrespective of HR status; TN: HR- and HER2-) (Tab. III). In HR+ and HER2+ patients, serum ANGPTL2 levels showed no correlation with clinicopathological factors. However, serum ANGPTL2 levels in TN patients diagnosed as NG 3 were significantly higher than those classified as NG 1 or 2. Overall, we can conclude that serum ANGPTL2 levels reflect NG status in HR-negative patients, especially TN patients.
ASSOCIATION BETWEEN SERUM ANGPTL2 LEVELS AND CLINICOPATHOLOGICAL FACTORS ACCORDING TO TUMOR SUBTYPE
ASSOCIATION BETWEEN SERUM ANGPTL2 LEVELS AND CLINICOPATHOLOGICAL FACTORS ACCORDING TO HORMONE RECEPTOR STATUS
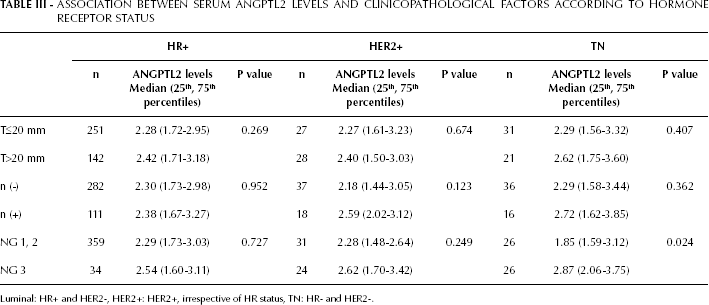
Luminal: HR+ and HER2-, HER2+: HER2+, irrespective of HR status, TN: HR- and HER2-.
Discussion
The present study demonstrates that serum ANGPTL2 levels increase in patients with advanced breast cancer, and that serum ANGPTL2 levels in HR-negative patients, and especially TN patients, reflect nuclear grade status. It is well accepted that significant genetic alterations and genomic instability promote changes in the morphology of tumor cells and their nuclei. A high nuclear grade is also correlated with poor prognosis (3). ANGPTL2 activity in TN tumor cells may promote such changes, which are related to aggressive phenotypes, through mechanisms such as genetic alterations. This is the first report showing that ANGPTL2 levels are positively correlated with the clinical pathogenesis of breast cancer progression.
A family of proteins structurally similar to angiopoietins (Ang) has been designated “angiopoietin-like proteins” (ANGPTLs). ANGPTLs exhibit an N-terminal coiled-coil domain and a C-terminal fibrinogen-like domain, both also characteristic of Ang proteins. However, ANGPTLs do not bind to either the Ang receptor Tie2 or the related Tie1 receptor, strongly suggesting that these ligands function differently from Angs (9). Many studies report that Ang and ANGPTL family proteins promote cancer metastasis (10-11-12-13-14-15). Minami et al. showed that the calcineurin-NFAT pathway promotes pulmonary tumor metastasis by inducing Ang2 (10, 11). Ang2 enhances tumor progression by activating macrophages in the tumor environment, decreases endothelial integrity, and promotes lymph node and lung metastasis (12). Ang2 and ANGPTL2 are induced by the hypoxic microenvironment of cancer cells (6, 13). ANGPTL4 is also induced by hypoxia and inhibits endothelial cells interactions to promote tumor metastasis (14). A common theme of these studies is that ANGPTL2 and 4 and Ang2 are induced by hypoxia, a state created by many tumor cells. Recently, we showed that ANGPTL2 induction is regulated by DNA methylation, which is seen in hypoxic cancer microenvironments (15). In this study, we did not examine whether ANGPTL2 and other Ang cooperate with each other in the cancer cell microenvironment. That relationship should be investigated in future studies.
We recently reported studies of immunodeficient female mice transplanted with MDA-MB231 breast cancer cells showing that tumor cell-secreted ANGPTL2 shortened survival time and promoted cancer metastasis by accelerating motility, invasion, and tumor angiogenesis in an autocrine/paracrine manner (6). Taken together with the clinical findings reported here, we suggest that inhibiting ANGPTL2 activity or secretion from breast cancer cells represents a novel potential therapy to antagonize advanced breast cancer.
Recently, Zheng et al reported that the immune inhibitory receptor human leukocyte immunoglobulin-like receptor B2 (LILRB2), and its mouse orthologue paired immunoglobulin-like receptor B2 (PIRB2), are ANGPTL2 receptors (16). The authors also reported that LILRB2 and PIRB2 are respectively expressed on human and mouse hematopoietic stem cells (HSCs), and that ANGPTL2 binding to these receptors supports ex vivo HSC expansion. Furthermore, in mouse models of human acute myeloid leukemia, impaired PIRB signaling promotes leukemia cell differentiation (16). Since we reported that ANGPTL2 contributes to tumor invasion, metastasis, and recurrence (6), it will be of interest to investigate whether breast cancer stem cells express ANGPTL2 and whether ANGPTL2 signaling contributes to the invasive phenotypes of those cells.
Currently, tumor markers such as CEA and CA15-3 are used to detect breast cancer recurrence and are thought, in some cases, to be potentially more sensitive than radiographic imaging to detect metastatic disease. However, the use of these markers alone is not recommended to detect relapse or to monitor the therapeutic effect (17), partly because these markers are not always sensitive enough to detect cancer recurrence (18, 19), and partly because their serum levels can vary with liver function or lifestyle (20). Also, successful systemic therapy often increases serum concentrations of these factors (an occurrence known as marker “flare”) due to the release of antigens by cytolysis (20, 21). Thus, additional tools enabling earlier diagnosis are needed to decrease the number of deaths from tumor metastasis.
Here we demonstrated that serum ANGPTL2 levels in patients with metastatic breast cancer were significantly higher than those in healthy subjects or in patients with DCIS or non-metastatic IDC. In addition, serum ANGPTL2 levels in HR-negative patients, especially TN patients, reflected the nuclear grade status. These findings suggest that serum ANGPTL2 levels in breast cancer patients could serve as a potential diagnostic tool of histological grades.
Footnotes
Acknowledgements
We thank Mss. S. Iwaki, O. Takahashi, K. Tabu, and M. Nakata for technical assistance.
