Abstract
Purposes
Obesity is associated with an increased risk of colorectal cancer (CRC), and ghrelin (GHRL) and resistin (RETN) are thought to be related to obesity. Our aim was to investigate whether GHRL and RETN gene variants are associated with CRC risk.
Materials and Methods
All 414 subjects, including 197 cases with CRC and 217 controls, were genotyped for the GHRL (rs26802) and RETN (rs1862513) or -420C>G gene variants using the PCR-RFLP method.
Results
Our findings indicated that the RETN -420C>G “CC” genotype, compared with the “GG” and “GC” genotypes, was a marker of decreased CRC susceptibility; the difference remained significant after adjustment for age, BMI, gender, smoking status, NSAID use, and family history of CRC (p=0.020; OR=0.52, 95% CI=0.30-0.90). Furthermore, after adjustment for confounding factors, the -420C>G “CC” genotype, compared with the “GG” genotype, was associated with a decreased risk for CRC (p=0.044; OR=0.53, 95% CI=0.29-0.98). In addition, no significant difference was observed for the GHRL (rs26802) gene variant.
Conclusions
To our knowledge, this is the first study suggesting that the RETN -420C>G “CC” genotype is a marker of decreased CRC susceptibility. This observation is relevant from a scientific perspective and deserves further investigations.
Introduction
Colorectal cancer (CRC) is a common type of cancer in the world and is the second leading cause of cancer-related mortality (1). Previous studies demonstrated that obesity is a risk factor for CRC (2, 3) and could promote cancer through both inflammation and insulin resistance (4). Previous studies have also demonstrated that ghrelin (product of the GHRL gene) and resistin (product of the RETN gene) participate in body weight regulation. Low ghrelin levels result in an increased risk of obesity (5); in contrast, resistin is positively related to obesity (6).
The main source of ghrelin, a 28-aminoacid peptide, is the stomach (7). Ghrelin stimulates food intake (8) and might have a role in cell proliferation and apoptosis (9). Furthermore, the expression of GHRL is enhanced in malignant colorectal cells (10), while its serum level is significantly lower in patients with colon cancer than in controls (11). In addition, GHRL gene polymorphisms were found to be significantly associated with body mass index (BMI) (12).
On the other hand, resistin, an adipocyte-derived peptide, plays an important role in energy homeostasis and carcinogenesis (13), and its serum levels were higher in CRC patients than controls (14). Previous studies also demonstrated that the RETN gene variants were associated with the regulation of RETN gene expression (15), serum resistin levels (16), and BMI (17).
Accordingly, these observations led us to look for the possible associations of the GHRL (rs26802) and RETN (rs1862513) gene variants with CRC risk. Selection criteria for these polymorphisms were based on: (i) their use in previous genetic epidemiology studies, (ii) degree of heterozygosity, (iii) position in the gene, and (iv) functional importance.
Materials and Methods
Participants
A total of 197 patients with CRC (age range, 21-83 years) and 217 controls (age range, 19-81 years) who referred to the Taleghani Hospital (Tehran, Iran) between January 2010 and May 2012 were enrolled into this case-control study. All the 414 subjects were Iranian and genetically unrelated. CRC patients and control subjects were all recruited among individuals undergoing colonoscopy for various gastrointestinal (GI) complaints, among which long-term unexplained abdominal pain, unexplained weight loss, unexplained changes in bowel habit, rectal bleeding, chronic diarrhea, and constipation, or among individuals participating in a screening program. The cases' group consisted of all patients eligible for colonoscopy and with positive pathologic report for CRC; eligibility criteria for the control group included no individual history of malignant colorectal tumors, adenomatous polyps, or other polyps. Before undergoing colonoscopy, patients were interviewed using a self-administered questionnaire, and information regarding the subjects' demographic, anthropometric, and clinical characteristics was recorded. Prior to entering the study, informed consent was obtained from each subject; the study was approved by the ethical committee of the Gastroenterology and Liver Diseases Research Center, Shahid Beheshti University of Medical Sciences, Tehran, Iran. The study protocol was in accordance with the principles of the Helsinki Declaration. The BMI of each subject was calculated as body weight divided by the height squared (kg/m2). Consequently, subjects were divided in subgroups based on the diagnosis of CRC and BMI values as follows: normal weight (BMI<25 kg/m2) controls (n=117); overweight/obese (BMI≥25 kg/m2) controls (n=100); normal weight CRC cases (n=79); and overweight/obese CRC cases (n=118).
Genotype analysis
Five milliliters of peripheral blood were collected from all 414 subjects in tubes containing EDTA as anticoagulant, and stored at 4°C. Genomic DNA was isolated from peripheral blood leucocytes using standard methods, and genotyping was performed by the PCR-RFLP method. Characteristics of the studied gene variants, PCR primers, PCR and RFLP conditions, and the length of the PCR and RFLP products are summarized in Table I. Furthermore, Figure 1 shows a schematic representation of the GHRL and RETN genes with the variants genotyped in this study. The PCR products were digested overnight with the appropriate restriction enzymes (Fermentas, Leon-Rot, Germany), and the digested products were run on 2%-3% agarose gels (Fig. 2). Bands in gels were stained with ethidium bromide for visualization under ultraviolet light. The GHRL and RETN genotypes of each subject were identified according to the digestion pattern and the presence or absence of, respectively, the Mwo I and Ear I sites. The concordance of genotyping was confirmed by duplicate analysis of approximately 23% of the randomly selected samples.
INFORMATION FOR THE STUDIED MARKERS IN GHRELIN (GHRL) AND RESISTIN (RETN) GENES

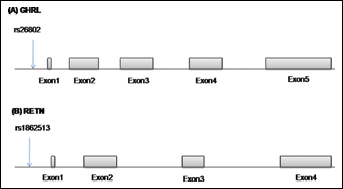
Overview of the ghrelin (A) and resistin (B) genes with the single nucleotide polymorphisms (SNPs) genotyped in this study.
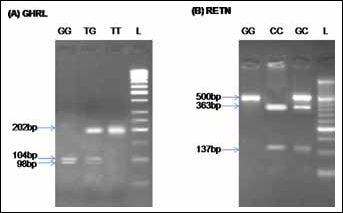
Representative results of the PCR-RFLP analysis for GHRL (A) and RETN (B) gene variants detected by agarose gel electrophoresis. Lane L illustrates the 50-bp DNA ladder.
Statistical Methods
Differences in demographic or anthropometric factors were calculated using the T-test or the chi-square test, as appropriate. We used the chi-square test for assessing the Hardy-Weinberg equilibrium (HWE) for the GHRL (rs26802) and RETN (rs1862513) gene variants among cases and controls, separately, and for comparing the distribution of the allele frequencies between the groups. Logistic regression was used to examine genotype frequencies between the different groups. We also used logistic regression analysis to adjust for confounding factors. All statistical analyses were performed with the SPSS software (version 15.0; SPSS Inc. Chicago, IL, USA) and p values less than 0.05 were considered statistically significant.
Results
The selected characteristics of the study population and their statistical significance are summarized in Table II. In general, CRC patients were older (p<0.001), more likely to be overweight or obese (p=0.011), and less likely to use NSAIDs (p<0.001) than their control counterparts. However, there were no significant differences between cases and controls in terms of gender, smoking status, and family history of CRC.
SELECTED CHARACTERISTICS OF THE STUDY SUBJECTSa
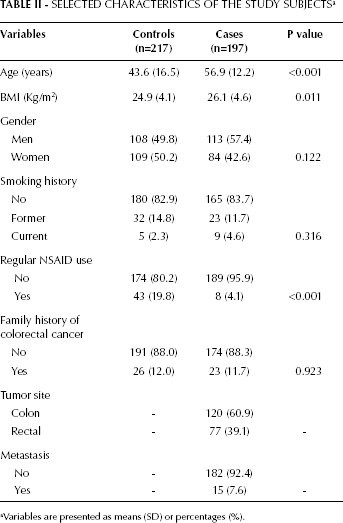
aVariables are presented as means (SD) or percentages (%).
The distribution of genotypes and alleles of the GHRL (rs26802) and RETN (rs1862513) gene variants in CRC cases and controls are shown in Table III. The GHRL and RETN genotype frequencies were in HWE among the cases, but were out of HWE in controls (p<0.05), with decreased heterozygosity. As shown in Table III, no significant difference was observed in genotype and allele frequencies between the cases and the controls for the GHRL (rs26802) gene variant. Furthermore, after adjustment for age, BMI, gender, smoking status, NSAID use, and family history of CRC, no significant association between the rs26802 variant and CRC risk was found. However, analysis of the RETN -420C>G (rs1862513) variant showed a significant difference between CRC patients and controls (Tab. III). These data indicate that the
GENOTYPE AND ALLELE FREQUENCIES OF GHRELIN (GHRL) AND RESISTIN (RETN) GENE VARIANTS IN CASES OF COLORECTAL CANCER AND IN CONTROLS a
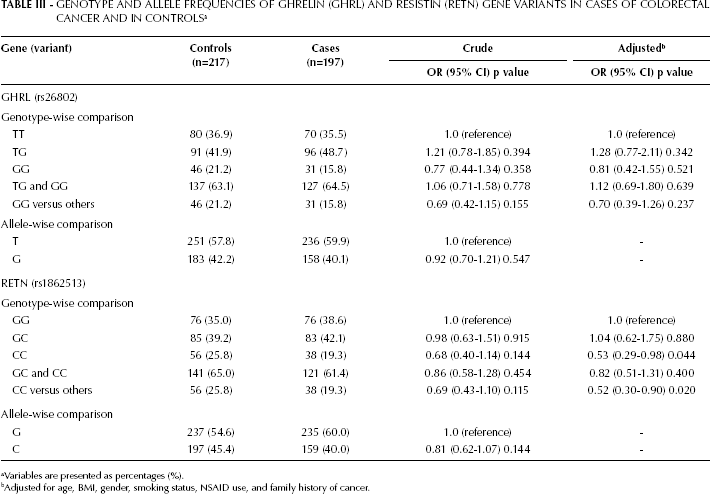
Variables are presented as percentages (%).
Adjusted for age, BMI, gender, smoking status, NSAID use, and family history of cancer.
RETN -420C>G “CC” genotype, compared with the “GG” and “GC” genotypes, is a marker of decreased CRC susceptibility; this difference remained significant after adjustment for age, BMI, gender, smoking status, NSAID use, and family history of CRC (p=0.020; OR=0.52, 95% CI=0.30-0.90). In addition, compared with the “GG” genotype, the -420C>G “CC” genotype occurred less frequently in cases than controls, and the difference remained significant after adjustment for confounding factors (p=0.044; OR=0.53, 95% CI=0.29-0.98) (Tab. III).
Finally, we also assessed the risk of obesity in relation to the GHRL (rs26802) and RETN (rs1862513) gene variants (data not shown). For both gene variants, we did not find any significant difference in genotype and allele frequencies between the normal-weight and overweight/obese patients, nor between normal-weight and overweight/obese controls.
Discussion
We conducted a case-control study to explore the possible association between the GHRL (rs26802) and RETN (rs1862513) gene variants and CRC risk among Iranians. For GHRL, we found no significant difference in terms of genotype or allele frequencies between patients with CRC and controls; this was true also after adjusting for age, BMI, gender, smoking status, NSAID use, and family history of CRC. Also, our findings indicated for the first time that the RETN -420C>G “CC” genotype, compared with the “GG” and “GC” genotypes is a marker of decreased CRC susceptibility; this difference remained significant after adjustment for the confounders. However, the distribution of the RETN -420C>G genotypes deviated significantly from the HWE in the controls. Deviations from the HWE may be due to different reasons, including population stratification, genotyping error, small sample size, inbreeding, or a true association. Since all the individuals included in this study were Iranian (as their parents and grandparents were) and our genotype data was of high quality, it seems unlikely that population stratification or genotyping error can results in departure from the HWE. However, it is possible that this deviation is a function of the small sample size, because the chance of a deviation from the HWE decreases with increasing sample size. Another important point that should be kept in mind is that no deviation from the HWE would be observed in the controls if they were recruited from the general population without any exclusion criteria. In other words, when individuals are excluded from the control group because not fulfilling the inclusion criteria, a violation of the laws of the HWE will occur and this may result in a deviation from the HWE. Accordingly, the deviation from the HWE can affect the interpretation of the associations observed in this study; nonetheless, the possibility of a true association between the RETN -420C>G variant and CRC risk should not be excluded.
At present, CRC is considered as a complex disease that might result from the interaction between genetic and environmental factors. One method for identifying novel susceptibility genes for complex diseases, such as CRC, is to study the SNPs in candidate genes. Interestingly, in the present study we demonstrated, for the first time, a negative association between the RETN -420C>G “CC” genotype and CRC risk,. This finding is in line with 2 recent studies (18, 19) showing a higher frequency of the “GG” genotype among CRC patients than controls. However, while we observed that the RETN -420C>G “CC” genotype was associated with a decreased risk of CRC, the 2 previous studies did not find this same association. In addition, the allelic frequencies and genotype distributions in this study were different from those reported for the populations in the other 2 studies. In our study, the frequencies of the CC, CG, and GG genotypes were 25.8, 39.2, and 35.0, while those for the C and G alleles were 45.4 and 54.6 in controls; in CRC patients, the corresponding frequencies were respectively 19.3, 42.1, 38.6, 40.0, and 60.0. Conversely, in the study by Wagsater et al (18) the frequencies of the CC, CG, and GG genotypes were 53.5, 40.2, 6.3 and for the C and G alleles were 73.6 and 26.4 in controls; in CRC patients, the corresponding frequencies were, respectively, 51.2, 38.3, 10.5, 70.4, and 29.6. Similarly, in the study by Pechlivanis et al (19) the frequencies of the CC, CG, and GG genotypes were 55.0, 37.1, and 7.9 and for the C and G alleles were 73.6 and 26.4 in controls; in CRC patients, the corresponding frequencies were, respectively, 49.4, 40.8, 9.8, 69.8, and 30.2. It is possible that the different allelic and genotype frequencies observed are due to ethnic differences among the selected populations. A recent study showed that resistin levels were higher in CRC cases than controls (14). Previous studies have also shown that the RETN -420C>G variant was associated with the concentration of resistin in serum (16) and with the regulation of RETN gene expression (15). The -420G allele was associated with higher serum resistin concentrations (16) and the RETN -420C>G “GG” genotype increases RETN promoter activity (15). Accordingly, our finding that the RETN -420C>G “CC” genotype appeared to be a marker of decreased CRC susceptibility is consistent with the notions above. The -420C>G variant is located in the promoter of the RETN gene, and alterations in the promoter sequence may influence protein expression. A possible hypothesis is that, since the “C” allele of the -420C>G variant is less stable and translated less efficiently into RETN protein, reduced RETN abundance may impede RETN actions and may contribute to the decreased CRC risk. Such mechanism is only speculative at present but biologically plausible. Alternatively, the -420C>G variant may be in linkage disequilibrium with another functional variant of the RETN gene, which may explain the associations observed. Previous studies have shown that obesity is a risk factor for CRC (2) and significant associations have been found between obesity risk and both high levels of resistin (6) and the RETN 420G allele (17). An alternative mechanism linking resistin with CRC is inflammation. The relation between resistin and inflammatory markers appears to be independent of BMI (20), and resistin may present a molecular link between CRC and inflammation. Furthermore, Danese et al (14) demonstrated that, in CRC cases, the serum levels of resistin were positively associated with tumor stage and C-reactive protein (CRP) levels, and negatively associated with high-density lipoprotein (HDL) and cholesterol levels. However, they also showed that there was no association between resistin, tumor location, and vascular invasion. Clearly, despite these facts, the exact molecular mechanism behind the association observed in this study for the -420C>G variant and CRC risk is largely undetermined.
The other SNP studied here, the rs26802 polymorphism, is located in intron 1 at the 5′-untranslated region (5′-UTR) of the GHRL gene; alterations in intronic sequences may influence protein expression and RNA splicing. Our findings are in concordance with a recent study (21), in which no association was found between the rs26802 variant of the GHRL gene and CRC risk; nevertheless, Campa et al (21) found significant associations between other variants of this gene (rs27647 and rs35683) and CRC risk. Unfortunately, inconsistent results, such as these, are common in genetic association studies (22, 23) and discrepancies in these studies may be due to false positive results, differences in the genetic and/or environmental factors triggering the development of CRC, small sample size, and statistical methods. Alternatively, the rs26802 variant may be in linkage disequilibrium with another unknown functional variant of the GHRL gene that explains the discrepancy observed. However, larger studies in other populations should be conducted as to be able to conclude that there is no relationship between the GHRL gene and CRC risk.
Although well designed, our study has several limitations. One limitation is the modest sample size that precludes drawing strong conclusions. Another limitation is that only 1 variant for each of the 2 genes was genotyped, and thus coverage of the gene was incomplete. The other potential limitation is the lack of information on serum levels of ghrelin and resistin, which could modify the effects observed here. Accordingly, we could not completely rule out the possibility of changes in our findings. Nonetheless, the possibility of a true finding should not be excluded.
In conclusion, our findings suggest that the RETN -420C>G “CC” genotype is a marker of decreased CRC susceptibility. To our knowledge, this is the first study reporting this result. This observation is relevant from a scientific perspective and deserves further investigations. Furthermore, in this case-control study, the rs26802 variant of the GHRL gene does not appear to affect the development of CRC in this Iranian population.
Footnotes
Acknowledgments
The authors thank all patients and healthy blood donors for providing blood samples.
