Abstract
Background
Disparities of biomarkers’ expression in breast cancer across different races and ethnicities have been well documented. Proline, glutamic acid, and leucine-rich protein 1 (PELP1), a novel ER coregulator, has been considered as a promising biomarker of breast cancer prognosis; however, the pattern of PELP1 expression in Chinese women with breast cancer has never been investigated. This study aims to provide useful reference on possible racial or ethnic differences of PELP1 expression in breast cancer by exploring the pattern of PELP1 expression in Chinese women with primary breast cancer.
Methods
The expression of PELP1 in primary breast cancer samples from 130 Chinese female patients was detected by immunohistochemistry and correlated to other clinicopathological parameters; for comparison, the expression of PELP1 in 26 benign breast fibroadenomas was also examined.
Results
The overall value of the PELP1 H-score in breast cancer was significantly higher than that in breast fibroadenoma (p<0.001). In our breast cancer patients, the ER/HER-2-positive group had significantly higher PELP1 H-scores than their negative counterparts (p=0.003 for ER and p=0.022 for HER-2); the Ki-67-high group also showed significantly higher PELP1 H-scores than the Ki-67-low group (p=0.008). No significant association between PELP1 H-scores and other clinicopathological parameters was found. Finally, the PELP1 H-score in breast cancers of the luminal B subtype was significantly higher than that in the triple negative subtype (p=0.002).
Conclusion
Overexpression of PELP1 in Chinese women with primary breast cancer appears to be associated with biomarkers of poor outcome; these results are similar to other reports based on Western populations.
Introduction
Mammary tumorigenesis is closely associated with ovarian hormones, and approximately 70% of breast cancers are estrogen receptor (ER)-positive at the time of tumor presentation (1). The biological action of estrogen mediated by ER requires coregulatory proteins, including coactivators and corepressors, which usually form complexes with the activated ER and determine the magnitude or specificity of ER signaling. The coregulators of estrogen's function are multitasking molecules and are involved in various estrogenic activities, including the modulation of genes’ transcription and the activation of cytoplasmic kinase cascades (2). Accumulating evidence suggests that, more than ER itself, the deregulation of the ER coregulators may provide breast cancer cells an advantage in tumor initiation, progression, metastasis, and therapeutic resistance (3).
Proline, glutamic acid, and leucine-rich protein 1 (PELP1), also know as modulator of non-genomic actions of estrogen receptor (MNAR), is a novel ER coregulator that has shown distinctive characteristics compared to other ER coregulators. PELP1 contains several motifs and domains that can be uniquely recognized by steroid receptors including ER, androgen receptors, glucocorticoid receptors, and progesterone receptors. In the ER signaling pathway, PELP1 serves as a scaffolding protein that couples various signaling complexes with ER and participates in both genomic and non-genomic functions (4, 5).
Accumulating evidence suggested that PELP1 acts as a proto-oncogene. Overexpression of PELP1 induces malignant transformation of normal cells, accelerates cell cycle progression, promotes tumor cells proliferation, and enhances migration and invasion of tumor cells (6–10). The expression of PELP1 has been found to be deregulated in a panel of cancers including carcinoma of the breast (6, 11), endometrium (12), and ovary (13, 14). Recent studies based on large clinical samples also suggest that PELP1 may be used as a biomarker for predicting breast cancer prognosis (11).
Disparities in terms of incidence, outcome, as well as expression levels of biomarkers in breast cancer have been well documented across different races and ethnicities (15–17). Despite representing a promising biomarker of breast cancer prognosis, to date little is known about the patterns of PELP1 expression in patients with breast cancer besides the few studies in the American population. In the present study, we aim at providing useful reference on the possible racial or ethnic differences of PELP1 expression in breast cancer. For this aim we investigated the expression of PELP1 in a series of 130 samples of breast cancer and 26 benign breast fibroadenomas from Chinese women of Han nationality; we also correlated the PELP1 expression levels in breast cancer to clinicopathological parameters. To the best of our knowledge, this is the first study reporting the pattern of PELP1 expression in women with breast cancer from the Chinese population.
Methods
Patients’ samples
The protocol of this study was approved by the institutional review board of the Harbin Medical University. Formalin-fixed, paraffin-embedded surgical excisional tissue specimens from 130 primary breast cancers and 26 benign breast fibroadenomas, from patients diagnosed between 2010 and 2012, were obtained from the archives of the Department of Pathology at the Fifth Affiliated Hospital of Harbin Medical University. Clinical information was extracted from the case files: all patients were of Han nationality, females, and with an age ranging between 26 and 76 years; none of the patients underwent chemotherapy, radiotherapy, or endocrine therapy before surgical excision.
Immunohistochemistry (IHC)
All samples were sliced into 5-μm thick serial sections, then deparaffinized with xylene and rehydrated on alcohol gradients. Endogenetic peroxidase was blocked with 3% hydrogen peroxide-methanol for 30 minutes before antigen retrieval. For HER-2 immunostaining, the HercepTest™ kit (DAKO, Denmark) was used following the kit's manual recommendations. For PELP1, ER, progestogen receptor (PR), and Ki-67/MIB-1 immunohistochemical staining, antigen retrieval and primary antibody incubation were performed as reported in Table I. The Real EnVision Detection system (DAKO, Denmark) was used instead of the traditional secondary antibody, and sections were visualized with the chromogen DAB and counter-stained with hematoxylin. Negative controls were performed by omitting the primary antibodies, which were substituted by dilution buffer (DAKO, Denmark).
Antigen Retrieval And Primary Antibody Working Conditions For Ihc
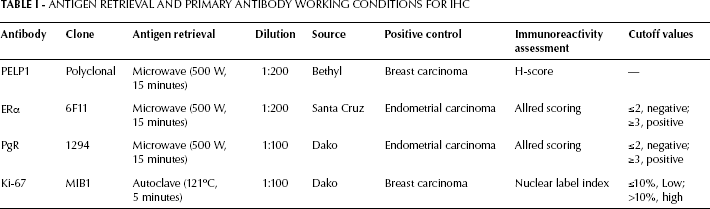
Evaluation
The evaluation was independently performed by 2 of the investigators (PW and LSG). The H-score was used to assess immunoreactivity of PELP1 as previously described (11): briefly, PELP1 staining intensity was scored as 0, 1, 2, or 3, and the percentage of positive cells was determined for each score as to generate a final score in the range of 0–300. For assessing ER and PR immunoreactivity the Allred scoring system was applied; a score ≥3 was defined as positive. HER-2 assessment followed the guidelines of the HercepTest scoring: a score ≥3 was defined as positive (18, 19). The Ki-67 labeling index (LI) was defined as the percentage of cells that showed nuclear immunoreactivity to MIB-1 after counting 1,000 tumor cells, and a Ki-67 Li>10% was defined as high.
Statistical analysis
To date, a commonly accepted criterion for determining the cutoff of the PELP1 immunostaining score is still not available. To increase the comparability of our results to those of other similar studies, we here chose to use the H-score directly for statistical analysis. All statistical analyses were performed using the SPSS 17.0 statistical software (Chicago, USA). Associations between the PELP1 H-score and different clinicopathological parameters were determined with a non-parametric test; for multiple groups’ analysis, we used the Kruskal-Wallis H test, while for the comparison between 2 groups, we used the Mann-Whitney U test. P values <0.05 were considered statistically significant.
Results
PELP1 expression in breast cancer and benign breast fibroadenoma
In breast cancer tissues, PELP1 staining was mainly distributed in the nuclei of tumor cells, while in benign breast fibroadenomas PELP1 staining was mainly detected in the nuclei of ductal epithelial cells; no cytoplasmic staining of PELP1 was found in either tumor cells or normal ductal epithelial cells (Fig. 1). The overall value of the PELP1 H-score in our breast cancer patients was significantly higher (169.1±6.9) than that in benign breast fibroadenoma patients (76.4±15.3, p<0.001).
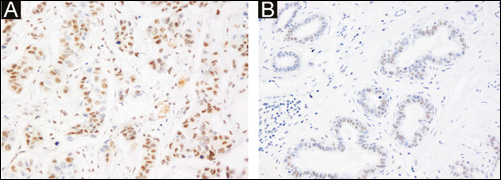
IHC detecting PELP1 expression in (A) breast cancer and (B) benign breast fibroadenoma from Chinese women. The positive immunostaining of PELP1 is mainly distributed in the nuclei of tumor cells and normal ductal epithelial cells (Magnification: 200x).
Correlating PELP1 expression to clinicopathological parameters
In our breast cancer patients, PELP1 expression was correlated to both clinical and pathological parameters including patient's age, histologic type, tumor size, lymph node stage, NPI, mitosis, clinical stage, and status of ER, PR, HER-2, and Ki-67 LI. Both the ER-positive and HER-2-positive groups showed significantly higher PELP1 H-scores than their negative counterparts (178.7±7.1 vs 119.2±17.7 for ER, p=0.003; 197.9±14.0 vs 160.4±7.7 for HER-2, p=0.022); the Ki-67-high group also showed significantly higher PELP1 H-scores than the Ki-67-low group (196.9±10.7 vs 156.7±8.4, p=0.008). Additionally, we did no find any significant association between PELP1 H-scores and other clinicopathological parameters (Tab. II).
Correlation Between Pelp1 Expression And Other Clinicopathological Parameters In Patients With Breast Cancer
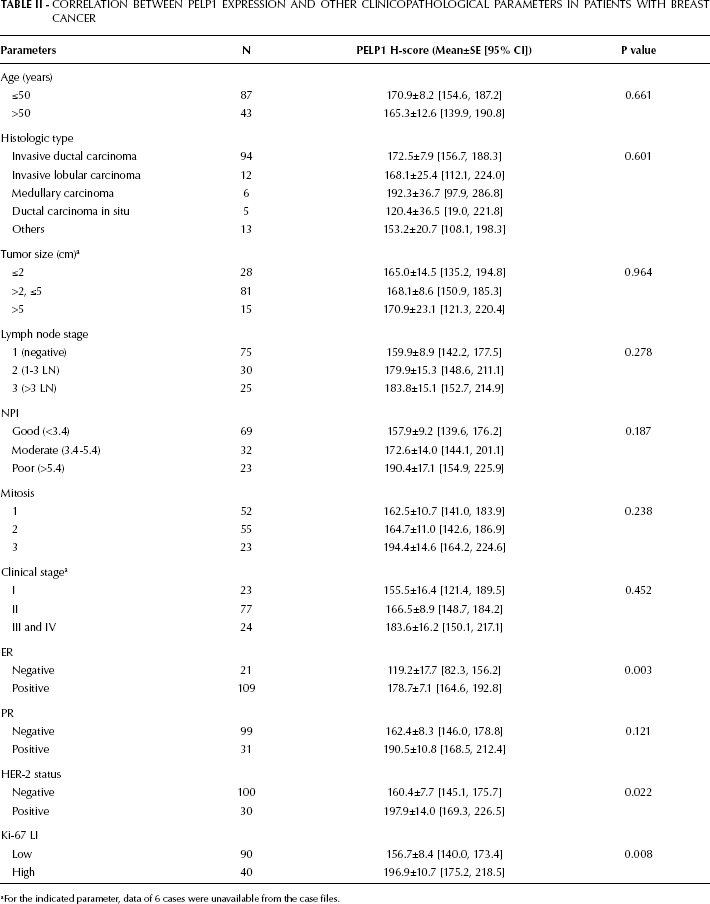
For the indicated parameter, data of 6 cases were unavailable from the case files.
Correlation of PELP1 expression and breast cancer molecular subtypes
We also considered the correlation between the PELP1 expression in breast cancer and the molecular subtypes defined by ER, PR, and HER-2 status. The average PELP1 H-score was 169.4±8.0 in the luminal A subtype (ER+ or PR+, HER-2-, 84 cases), 205.8±14.4 in the luminal B subtype (ER+ or PR+, HER-2+, 27 cases), 127.0±36.9 in the HER-2 subtype (ER-, PR-, HER-2+, 3 cases), and 113.5±19.9 in the triple negative subtype (TN, ER-, PR-, HER-2-, 16 cases). The Kruskal-Wallis non-parametric H test showed a significant difference of the PELP1 H-score among the 4 groups (p=0.003), and subsequent post-hoc comparison showed that the difference mainly occurred between the luminal B and TN subtypes (p=0.002, Fig. 2).
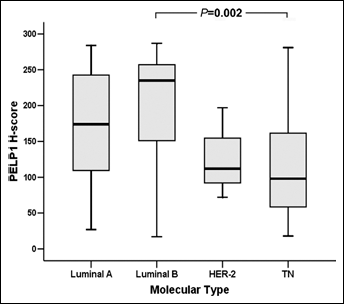
Comparison of the PELP1 H-scores among different molecular subtypes of breast cancer in Chinese women. The H-score of PELP1 in the luminal B subtype of breast cancer was significantly higher than that in the TN subtype.
Discussion
In this study, we detected the PELP1 expression in a series of 130 primary breast cancers and 26 benign breast fibroadenomas from Chinese women of Han nationality. In line with other studies using the commercially available antibodies against PELP1 for IHC (11, 14), we only found a positive staining of PELP1 in nuclei of tumor cells and normal ductal epithelial cells, while no cytoplasmic staining of PELP1 was found. Evidence from in vitro studies suggests that PELP1 is involved in the membrane-initiated effects of estrogen; however, most of the data were derived from a special cell model in which an exogenous PELP1 mutant, lacking the nuclear localization domain, was transfected and forced to localize into the cytoplasm (9, 20). It was also reported that the intracellular localization of PELP1 in wild-type breast cancer cells was regulated by its interaction with a trafficking protein, the hepatocyte growth factor-regulated tyrosine kinase substrate (HRS) (21); however, the detailed mechanisms regulating PELP1 subcellular distribution in tumor cells, especially in circumstances of primary breast cancer tissues, still need further investigation considering the complicated interactions between tumor cells and stroma.
While it is already clear that PELP1 plays an important role in tumorigenesis and progression of breast cancer, whether PELP1 expression is also deregulated in breast cancer in Chinese women was not systematically investigated until now. By comparing the overall H-score of PELP1 between the breast cancer cohort and the breast fibroadenoma cohort in our series, we found that PELP1 expression in breast cancers was significantly higher than that in breast fibroadenomas. This result not only supports the previous reports showing that PELP1 expression was deregulated during development of breast cancers but also suggests that deregulation of PELP1 expression may be a common event in breast cancers across different races and ethnicities.
PELP1 has important functional roles in the crosstalk between estrogen and growth factors’ signaling. Besides acting as a coregulator of ER, PELP1 was also reported to interact with several growth factor receptors, including EGFR and HER-2 (22). In our breast cancer cohort, we found that PELP1 expression was positively correlated with ER and HER-2. It has been demonstrated that PELP1 contains 2 estrogen-responsive elements’ (ERE) half-sites in its promoter region, and its expression can be upregulated by estrogen-ER signaling at the transcriptional level (23). Moreover, it is well known that the transcriptional function of ER could be enhanced by excessively activated epidermal growth factor signaling (24). Therefore, the positive correlation between PELP1 and ER, as well as the positive correlation between PELP1 and HER-2, is in accordance with the biological function of PELP1 in the crosstalk between estrogen and growth factor signaling.
Ki-67, an indicator of cells’ proliferation, can only be detected during the active phases of the cell cycle, and is not detectable in resting cells. Numerous studies associated the percentage of Ki-67-positive cells (Ki-67 LI) to the overall and disease-free survival of patients with breast cancer. Usually, higher Ki-67 LI in breast cancer suggests a worse outcome (25, 26). In this study, we found that PELP1 expression in breast cancer cells was positively correlated with Ki-67 LI. In a previous study considering a large cohort (1,162 patients), the authors also reported the positive correlation between PELP1 expression and Ki-67 LI in breast cancer, and proposed PELP1 as a prognostic predictor of breast cancer (11). Despite the limited sample size and the lack of information on survival, our results still indirectly suggested the prognostic implication of PELP1 in Chinese women with breast cancer.
The identification of the molecular subtypes of breast cancer has enhanced our understanding of tumor biology, providing specific therapeutic targets, and aiding the clinical trials’ design (27). Specifically, in this study we tried to correlate the PELP1 expression levels in breast cancer and the molecular subtypes of this cancer: we found that PELP1 expression in the luminal B breast cancer subtype was significantly higher than in the TN subtype. Luminal B breast cancer is characterized by a higher proliferation, and is a type of cancer relatively insensitive to endocrine therapy, when compared with the luminal A type, and to chemotherapy, when compared with the TN type (28, 29). PELP1 has been reported to have great potentials in promoting cell proliferation and inducing tamoxifen/aromatase inhibitors resistance of breast cancer (7, 30); additionally, our ongoing study suggests that PELP1 may be also involved in chemotherapy resistance of breast cancer (unpublished data). The remarkably higher expression of PELP1 in luminal B breast cancer is in agreement with the functions of PELP1 and the characteristics of this subtype of breast cancer, and suggests that PELP1 may play a more important role in luminal B breast cancer. Several signaling pathways, such as the insulin-like growth factor pathway, the fibroblast growth factor pathway, and the phosphoinositide 3-kinase pathway have been proposed as potential therapeutic targets in luminal B breast cancer (31). Given the proto-oncogenic property of PELP1 and its dramatically higher expression in this subtype of breast cancer, it is important to further investigate whether PELP1 could also be listed as a candidate therapeutic target of luminal B breast cancer.
Despite its limitations, our study is the first to demonstrate the pattern of PELP1 expression in Chinese women with breast cancer. Further, our goal is to obtain the follow-up data of the Chinese patients here included, as to correlate the PELP1 expression with patient's outcome and compare the prognostic role of PELP1 in Chinese women with that in other racial groups. Moreover, subsequent work will also include the comparison of the predictive significance of PELP1 in different endocrine therapies (tamoxifen treatment vs aromatase-inhibitor treatment) in Chinese women with breast cancer. These additional analyses will provide deeper insight into the role of PELP1 in breast cancers across different races and ethnicities, and will contribute to the translational application of PELP1 research for the personalized treatment of breast cancer.
