Abstract
Introduction
It has been reported that elderly subjects have a compromised ability to produce melatonin nightly, and that reduced melatonin levels may be a risk factor for cancer. The purpose of this study was to evaluate the relationship between melatonin levels and chronic diseases in a cohort of elderly subjects using the Charlson comorbidity index (CCI).
Design
We performed a secondary data analysis of a longitudinal study of a representative, age-stratified, sample population.
Setting
The Treviso Longeva (Trelong) study, in Treviso, Italy.
Participants
A total of 114 men and 146 women, aged 77 years and older, still alive after 7 years of follow-up.
Measurements
As an estimation of serum melatonin secretion levels, urinary 6-sulfatoxymelatonin (aMT6s) was assayed in the urine of 260 elderly subjects using an enzyme-linked immunosorbent assay (ELISA) kit (product 01-EK-M6S, ALPCO Immunoassays, Windham, NH). All aMT6s levels were creatinine standardized ([aMT6s]/[creatinine]), and the CCI was calculated.
Results
The melatonin levels decreased with aging despite not reaching statistical significance, and the decrease was more evident in males than in females (40.5 ng vs 47.0 ng aMT6s/mg creatinine, ns). Melatonin levels were significantly lower in patients reporting insomnia (p=0.05). The CCI score was inversely correlated with the levels of melatonin (p=0.03). Melatonin levels of subjects affected by CCI pathologies were significantly lower than those of healthy subjects (p=0.03) and of subjects suffering from diseases not included in the CCI and, therefore, less severe (p=0.03).
Conclusion
Melatonin appears to be a marker of disease state and severity, as well as of sleep disorders, in the elderly. These early findings would confirm the protective role of melatonin against several chronic diseases. The benefits of this agent as a possible medication should be more thoroughly clinically tested.
Introduction
Melatonin was discovered in 1958 by Aaron Lerner and colleagues who described a year later its chemical structure as N-acetyl-5-methoxytryptamine (1, 2). Although in mammals melatonin is mostly secreted in the pineal gland, it can also be produced in other organs, such as the gastrointestinal tract, bone marrow, blood platelets, retina, and the extraorbital lacrimal glands (3). Melatonin is synthesized from serotonin through 2 steps: N-acetylation to N-acetylserotonin (NAS), followed by O-methylation. The major pathway of melatonin catabolism consists of hepatic 6-hydroxylation, followed by conjugation with sulfate to produce the easily excretable metabolite 6-sulfatoxymelatonin (3, 4). The levels of melatonin and their variation can be indirectly followed by measuring this urinary product.
In mammals, 2 melatonin receptor subtypes are currently known: MT1 (alias Mel1a, MTNR1A) (5) and MT2 (alias Mel1b, MTNR1B) (6). These receptors have been shown to be present in numerous human tissues, such as the gastrointestinal tract, both exocrine and endocrine pancreas, parotid gland, skin, breast epithelium, myometrium, cardiac ventricular wall, aorta, coronary and cerebral arteries, adipose tissue, platelets, and immune cells, with the highest density in the central nervous system (CNS). This remarkable variety of targets inevitably leads to highly pleiotropic effects exerted in numerous parts of the body (7). Melatonin has a well-defined circadian rhythm, which is synchronized during the 24 hours by the light-dark cycle. During the day serum melatonin concentrations are low (10-20 pg/mL) and increase at night (80-120 pg/mL) reaching a peak between midnight and 3 am (8).
In a young person, this daytime/nighttime difference in melatonin concentration is evident, but in a 60-year old person the difference is 80% lower than during puberty and it is almost null at older ages (9). Whether the disappearing of the circadian rhythm is a cause or a consequence of the aging process in humans is not known yet (10). Reduced concentrations of melatonin may be related to the deterioration of the circadian rhythm and to a lower sleep efficacy, both conditions associated with advanced age (11). On the other hand, melatonin has been suggested to play a role in aging because it is a potent free radical scavenger, and oxidative stress is one of the hypothesis used to explain aging (12).
It has been reported that low melatonin levels could be associated with an increased risk for breast, endometrial, colorectal, and prostate cancer as well as non-Hodgkin lymphoma (13-14-15-16-17-18). In several neurodegenerative disorders, especially Alzheimer's disease (AD), the levels of melatonin are frequently found to be lower than in age-matched controls (19-20-21). In many of these patients, the melatonin rhythm is practically abolished. There are other diseases and disorders in which melatonin results to be decreased, such as pain, cardiovascular diseases, endocrine and metabolic disorders, and in particular type 2 diabetes (22-23-24-25).
We examined the relationship between urinary 6-sulfatoxymelatonin (aMT6s), a useful tool for the estimation of serum-melatonin secretion (26, 27), aging, and chronic degenerative diseases in the population of the “Treviso Longeva” (Trelong) study (28), which is a prospective cohort study of women and men from Northern Italy aged 70 years and older. The Trelong study was conducted in the area of Treviso, which is characterized by the highest longevity in Italy, especially among women (29).
Materials and Methods
The Trelong study started in 2003. Eligible participants were resident in the municipality of Treviso, in Northeast Italy, an area with a resident population of 81,700 subjects, of whom 13,861 (17%) were over the age of 70 years. Participants were systematically sampled from the list of residents of the Registry Office of Treviso, based on an initial plan to include at least 125 women and 125 men aged 70-79 years; a total of 100 participants for each gender and per 10-year group, between the age of 80 and 100 years, were included; also, all subjects aged 100 and over were scheduled for inclusion. Considering these a priori requirements, 670 adults were eligible for enrollment. Of these, 668 participated (99.7% response rate): 311 men and 357 women, aged 70 years and older (mean age 84±8 years, range 70-105.5 years). The study protocol was approved by the ethical committee of the National Institute on Research and Care of the Elderly (INRCA, Italy). All participants and/or their caregivers provided written informed consent. An interviewer-administered questionnaire and a blood sample were collected at each participant's home (28). Information on cognitive performances was also obtained from caregivers. The baseline characteristics of this study population and the methodological details have been previously published (28). A follow-up questionnaire administered by the interviewer and a blood sample were collected at home from the 309 participants still alive in March 2010. Among these, 260 participants provided an early morning urine sample; the remaining 49 survivors had urinary incontinence, a condition that prevented the collection of urine.
Measurement of aMT6s in urine and other biochemical measures. A fasting morning urine sample and a blood sample were collected at home from each participant and transferred to the Clinical Chemistry Laboratory of the Treviso Hospital within 30 minutes.
Urinary aMT6s was assayed in each sample of urine previously collected and stored at -80°C by using an enzyme-linked immunosorbent assay (ELISA) kit (product 01-EK-M6S, ALPCO Immunoassays, Windham, NH) following the manufacturer's recommendations. This commercially available aMT6s ELISA is a competitive immunoassay using an antibody-capture technique with a lower detection limit of 0.8 ng/mL for aMT6s. The assay endpoint was read using a Bio-tek plate reader (Bio-Tek Instruments Winoski, VT) and the data were analyzed using the Kineticalc Application software (Bio-Tek). Creatinine levels were also measured for each sample using Sigma Diagnostics creatinine reagents (St Louis, MO, USA), and all aMT6s levels were standardized to creatinine ([aMT6s]/[creatinine]) to account for differences arising from variations in urine concentrations. To show equivalence between different runs, reference samples at 2 levels (4.0 and 22.6 ng/ml aMT6s) were analyzed together with the samples.
Routine hematological and clinical chemistry tests were performed immediately using standard laboratory methods. Serum biochemical markers were determined by commercially available methods (Roche Diagnostics GMBH, D-68298 Mannheim Germany) on a Modular Analyzer (28).
Assessment of chronic disease: incidence and prevalence. In-person interviews and examination of medical records provided information on history of chronic diseases, such as cerebrovascular diseases, chronic renal insufficiency, and cancer (28, 30). Using this information, a Charlson comorbidity index (CCI) was calculated (31-32-33-34-35). The CCI contains 19 categories of pathologies, which are defined using the International Classification of Diseases, Ninth Revision, Clinical Modification (ICD-9-CM) (36): acute myocardial infarction, congestive heart failure, peripheral vascular disease, cerebrovascular disease, dementia, chronic obstructive pulmonary disease, connective tissue disease, gastric ulcer, mild liver insufficiency, diabetes, hemiplegia, moderate-to-severe renal insufficiency, diabetes with end organ damage, solid tumors, leukemia, lymphomas, medium-to-severe liver insufficiency, tumor metastases, acquired immune deficiency syndrome (AIDS), and age. Each category is weighed according to the adjusted risk of 1-year mortality. The overall comorbidity score is related to the likelihood of mortality within 1 year; the higher the score (ranging from 0 to 37), the more severe the comorbidity burden (31).
Statistical Analysis
Means and standard deviations, along with percentages, were calculated for descriptive variables. The association between melatonin levels and health status has been assessed by the Pearson correlation, considering the associations between gender, age, insomnia, CCI score, C-reactive protein (CRP) levels, erythrocyte sedimentation rate (ESR), and melatonin levels. Kurtosis and skewness indices revealed a skewed distribution for the melatonin levels; therefore, the natural log of the melatonin levels were used when comparing patients with diseases included in the calculation of CCI with those not included in the calculation of CCI, through a t-test and a Mann-Whitney test.
Statistical analyses were performed using the R software (37). Results were considered significant at a level of p<0.05.
Results
The main characteristics of the 260 participants that provided a urine sample are presented in Table I. The sample is composed of 114 men and 146 women (mean age 85.4±6.4 years, range 77-105.5 years). The average melatonin levels were 44.2±42.7 ng aMT6s/mg creatinine (median 29.150 ng aMT6s/mg creatinine, interquartile range 14.05-61.83 ng aMT6s/mg creatinine); melatonin's levels decrease with aging, and were lower in males than in females (40.5 vs 47.0 ng aMT6s/mg creatinine), but in both cases the difference was not statistically significant. Melatonin levels were lower in patients reporting insomnia than in those without sleep disorders (34.8 vs 46.8 ng aMT6s/mg creatinine, p=0.05).
GENERAL CHARACTERISTICS OF THE 260 SUBJECTS STILL ALIVE AT THE TIME OF THE FOLLOW-UP VISIT, AND DISEASES ARISING AFTER THE STUDY INITIATION (IN 2003)
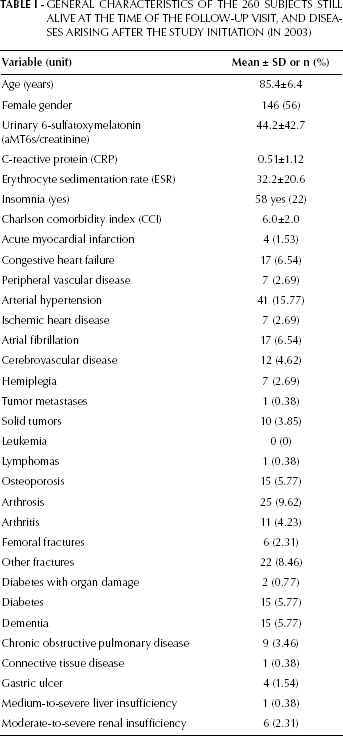
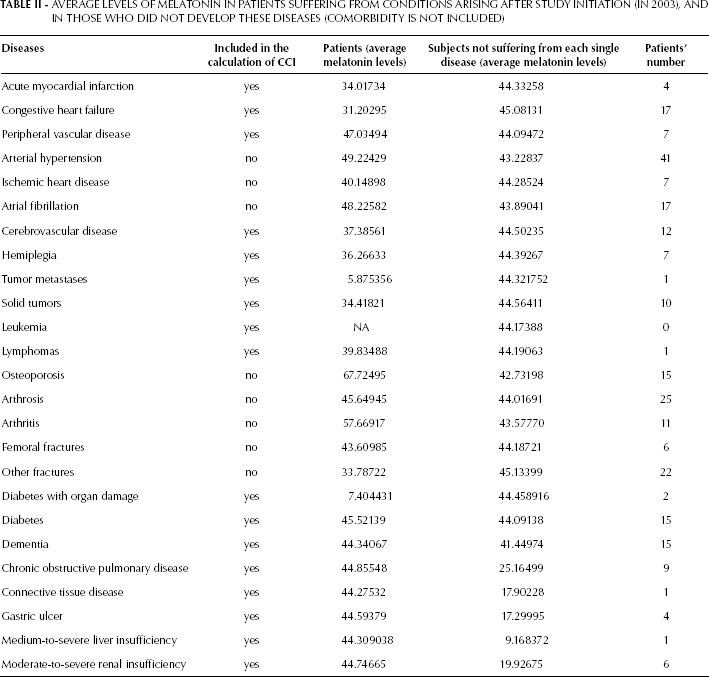
Table II shows the average value of melatonin both in patients who did and did not develop the abovementioned diseases after the initiation of the study (in 2003).
AVERAGE LEVELS OF MELATONIN IN PATIENTS SUFFERING FROM CONDITIONS ARISING AFTER STUDY INITIATION (IN 2003), AND IN THOSE WHO DID NOT DEVELOP THESE DISEASES (COMORBIDITY IS NOT INCLUDED)
CCI score and melatonin levels. For the purpose of this analysis, 4 groups of subjects were considered: 42 subjects who, during the follow-up, developed the abovementioned chronic diseases (cancer, diabetes, cerebrovascular disease, dementia, etc.) considered for the calculation of the CCI score (hereon called group P, for patients); 95 subjects who did not develop such pathologies before or after baseline (hereon called H, for healthy); 13 subjects free from CCI diseases and from any other disease and, therefore, completely healthy (heron called CH); and 76 patients that include the 42 patients with onset of CCI diseases after baseline and the 34 patients already suffering from CCI diseases before baseline (hereon called AP, for all patients).
The CCI score was inversely associated with the levels of melatonin (p=0.03): low levels of melatonin were associated with the presence of a higher number of diseases in a given subject and with a greater disease severity (Fig. 1). No significant association was observed between the levels of melatonin and those of circulating biomarkers of unhealthy aging, such as CRP and ESR. The comparison of the melatonin levels between the P and the H groups (Tab. III) showed that these were significantly lower in the P group (38.6 vs 48.4 ng aMT6s/mg creatinine, p value=0.04). Similarly, melatonin levels were significantly lower in the P group than in the CH group (38.6 vs 49.7 ng aMT6s/mg creatinine, p value=0.03) (Tab. III).
COMPARISON OF THE MELATONIN LEVELS BETWEEN PATIENTS AND HEALTHY SUBJECTS
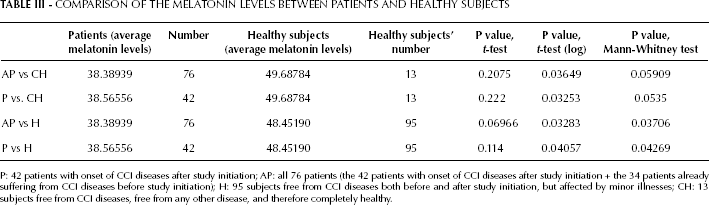
P: 42 patients with onset of CCI diseases after study initiation; AP: all 76 patients (the 42 patients with onset of CCI diseases after study initiation + the 34 patients already suffering from CCI diseases before study initiation); H: 95 subjects free from CCI diseases both before and after study initiation, but affected by minor illnesses; CH: 13 subjects free from CCI diseases, free from any other disease, and therefore completely healthy.
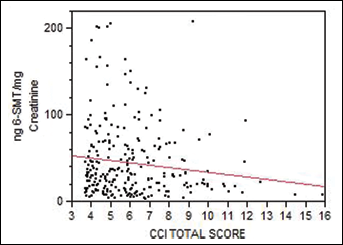
Urinary 6-sulfatoxymelatonin (aMT6) and CCI (p=0.03).
Melatonin levels were significantly lower in the AP group than in the H group (38.4 vs 48.4 ng aMT6s/mg creatinine, p value=0.03). The same results were observed when the AP group was compared to the CH subjects (38.4 vs 49.7 ng aMT6s/mg creatinine, p value = 0.03) (Tab. III).
Discussion
Great variations in the range of melatonin levels are found among individuals, suggesting that during their lifetime some individuals produce significantly less melatonin than others. The clinical significance of this observation is still unknown.
Melatonin production decreases with age, therefore the plasma concentrations and urinary amounts of the main metabolite, aMT6s, are reduced (25). In the current study, melatonin levels decrease with aging, but did not reach statistical significance. The fact that the sampled population is aged over 77 years, may be one of the reasons why we did not find a significant reduction in melatonin levels with increasing age. Other relevant reasons may be the roles played by unknown confounding factors.
Studies of gender-related differences in the timing of human circadian rhythms have reported conflicting results (38). A gender-related difference describing significantly higher levels of plasma melatonin in elderly women was previously observed (39). Our results are in line with this evidence: in our elderly population, melatonin levels were lower in males than in females, although this difference was not statistically significant.
Insomnia, defined as difficulties initiating or maintaining sleep, or non-restorative sleep associated with significant daytime distress (from the Diagnostic and Statistical Manual of Mental Disorders, 4th ed [DSM-IV]), occurs in about 30% of subjects aged 55 years and older (40). Insomnia impairs cognitive and physical functioning across a number of emotional, social, and physical domains (41). Since melatonin receptors are present in the CNS, and melatonin production in mammals exhibits a circadian rhythm, it was speculated that the day/night rhythm of melatonin production may be associated with the circadian rhythm of sleep and wakefulness. In our study, melatonin levels were lower in patients reporting insomnia than in those without sleep disorders, confirming the close association between this molecule and the rhythm of sleep even in older subjects.
In the current study, low levels of melatonin were associated with a higher CCI score, and, therefore, with the presence of a larger number of diseases and a greater severity of such diseases. Melatonin levels of subjects affected by CCI pathologies were significantly lower than those of subjects suffering from less severe conditions (not included in the CCI), and significantly lower than those of healthy subjects. Thus, melatonin appears to be a marker of disease state in the elderly, in whom low melatonin levels are associated with conditions of more numerous and more serious diseases. The powerful antioxidant action of melatonin would suggest that oxidation may mediate the increased susceptibility to diseases' development in situations in which melatonin levels are reduced.
When compared with classic antioxidants, melatonin is better in terms of limiting the destruction of intracellular macromolecules in those cases in which the damage is a consequence of excessive oxygen or nitrogen-based toxic reactants (42). The large amount of experimental data documenting melatonin's high efficacy and lack of, or minimal, toxicity over a very wide dosage range, would suggest that the usefulness of this agent should be more thoroughly tested at the clinical level.
This study has some limitations. First, the sample size is limited by the study design, thus making the statistical tests less powerful. The second limitation is the fact that the study relies on a single measurement of melatonin in urine: this may not reflect the true melatonin levels of the subject, given the circadian variability of the hormone. Another possible limitation is the self-reported health status of the subjects: in fact, the identification of chronic and acute conditions was largely the result of personal interviews. Finally, no significant association has been found with circulating biomarkers of unhealthy aging, such as CRP and ESR; the reason for this result could be that these biomarkers are also sensitive to acute inflammatory states and, thus, their values could reflect not only the chronic diseases considered in this work but also any temporary acute inflammatory state present at the time of blood sampling.
Nevertheless, this work has several strengths. Firstly, this is a systematic sample of adults aged 77 years and older from one of the longer-living communities in Italy; second, the sampled population is highly representative of the community, is residing at home, and was randomly drawn from the list of residents of the municipality of Treviso. Further studies are being conducted in selected samples (e.g., patients, only men or only women, etc.). Additional years of observation may further confirm our current findings.
Conclusion
In the current study, melatonin appears to be a marker of disease state and severity, with lower melatonin levels associated with higher CCI scores and with the presence of CCI pathologies. Similarly, melatonin levels were lower in patients reporting insomnia than in those without sleep disorders. Melatonin levels decreased with aging and were lower in males than in females, despite not reaching statistical significance.
Footnotes
Acknowledgements
We are grateful to all people who kindly agreed to participate in this study.
Experiments on Human Subjects: The study protocol was approved by the ethical committee of the National Institute on Research and Care of the Elderly (INRCA, Italy).
