Abstract
Objectives
The aim of this study was to analyze the roles of miR-143 and miR-145, as well as the gene and protein expression of their targets (
Methods
We analyzed the specimens of 44 patients diagnosed with BPH who underwent surgical treatment. The control group consisted of prostate samples from 2 young patients who were organ donors. miRNAs and their target genes were assessed using real-time polymerase chain reaction (qRT-PCR), and protein levels were assessed by Western blotting.
Results
miR-143 and miR-145 were overexpressed in, respectively, 62.5% and 73.8% of the cases. The
Conclusions
The overexpression of miR-143 and miR-145 in BPH samples suggests an association with the pathogenesis of the disease; additionally, the latter miRNA may act through the inhibition of MAP4K4.
Introduction
Benign prostatic hyperplasia (BPH) is one of the most common urologic diseases among elderly men (1) and is characterized by a marked stromal and acinar proliferation, which results in the occurrence of lower urinary tract symptoms (LUTS) (2) that may affect the quality of life of the patients (3). BPH pathogenesis has not been fully elucidated. Despite the well-established role of aging, hormonal factors, and genetic predisposition, the molecular and cellular mechanisms involving the stromal and epithelial components of the prostate that lead to BPH remain unclear (4).
MicroRNAs (miRNA) are a group of small RNA molecules of between 19 and 25 nucleotides that have a fundamental action in regulating the expression of genes in plants and animals (5). miRNAs are involved in a number of cellular processes, including apoptosis, differentiation, cell proliferation, and development (6-7-8). Additionally, the aberrant expression of these small RNAs has been linked to human diseases.
miR-143 and miR-145 play a fundamental role in the differentiation and proliferation of smooth muscle cells, with the ability to control the quiescent versus proliferative phenotype (9). These miRNAs appear also to have a key role in the development and differentiation of cardiac muscle and skeletal remodeling in response to tissue injury (10). miR-143 targets the genes KRAS and ERK5, and miR-145 targets the MAP3K3 and MAP4K4 genes (11).
The actions of miR-143, miR-145, and their target genes in prostate smooth muscle tissue have not been described. Therefore, the objectives of this study were to use tissue samples from patients with BPH to: (i) analyze the expression of miR-143 and its target genes (
Materials and Methods
Patients
We analyzed the specimens from 44 patients diagnosed with BPH who were surgically treated by transurethral resection of the prostate or open prostatectomy at the Hospital das Clinicas of University of Sao Paulo Medical School. The exclusion criteria were previous prostatic surgery, pelvic radiotherapy, prostate cancer, and the use of 5-alpha reductase inhibitors. The control group included prostate fragments (of the transition zone) from 2 young patients who were organ donors (mean age, 30 years). All subjects provided informed consent to participate in the study and to allow the genetical analysis of their biological samples. Approval for the study was given by the institutional board of ethics (no. 0258/10).
Surgical specimens were collected immediately after surgery. A fragment of 1 cm2 was sectioned from the transition zone of the prostate and stored at -80°C. The samples were used for the analysis of miRNA expression, as well as target genes' and proteins' expression. We analyzed miR-143 and miR-145, as well as their target genes
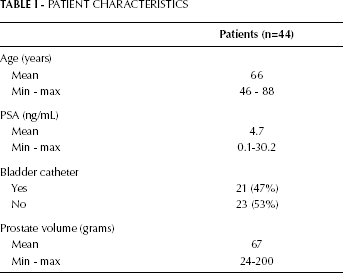
The expression levels were compared between the study group and the 2 controls. The patients were also categorized into groups according to selected clinical parameters (Tab. I): we considered the serum levels of preoperative PSA (≤4 ng/mL or >4 ng/mL), the use of a urinary catheter, the prostate volume (≤60 g or >60 g), and age (≤65 years and >65 years).
PATIENT CHARACTERISTICS
Isolation of RNA and miRNA
We used the mirVana kit (Ambion, Austin, TX) for RNA and miRNA extraction; cDNA was obtained using the TaqMan miRNA Reverse Transcription kit (Applied Biosystems, Foster City, CA) for miRNA and the High-Capacity cDNA Reverse Transcription kit for RNA according to the manufacturer's recommendations. An amount of 10 ng of miRNA was subjected to miR-143 and miR-145 sequence-specific primer stem-loop reverse transcription. The PCR reaction for obtaining cDNA from miRNA was performed using Veriti equipment (Applied Biosystem, Foster City, CA) according to the following parameters: 30 minutes at 16°C, 30 minutes at 42°C, and 5 minutes at 85°C. The same equipment was used to obtain cDNA from RNA with the following parameters: 10 minutes at 25°C, 120 minutes at 37°C, and 5 minutes at 85°C.
Analysis of gene expression
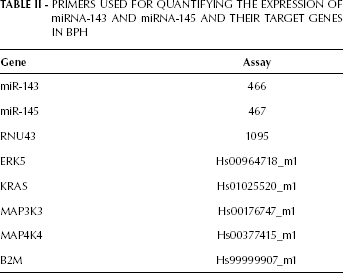
The miRNA and RNA expression levels were analyzed by qRT-PCR using an ABI 7500 Fast Real-Time PCR System (Applied Biosystems). The target sequences were amplified in a 10-μL reaction containing 5 μL TaqMan Universal PCR Master Mix, 0.5 μL TaqMan Gene Expression Assays (primers and probes; Tab. II), 1 μL cDNA, and 3.5 μL DNase-free water. The PCR cycling conditions were: 2 minutes at 50°C, 10 minutes at 95°C, and 40 cycles of 15 seconds at 95°C and 1 min at 60°C. All reactions were performed in duplicate and TaqMan B2M and RNU 43 were utilized, respectively, as the endogenous controls for gene and miRNA expression.
PRIMERS USED FOR QUANTIFYING THE EXPRESSION OF miRNA-143 AND miRNA-145 AND THEIR TARGET GENES IN BPH
We used the ΔΔCT method to calculate the relative expression of the miRNA and target genes using the formula ΔΔCT = (CT target gene/BPH sample - CT endogenous control/BPH sample) — (CT target gene/control sample - CT endogenous control/control sample). The fold change in gene expression was calculated as 2−ΔΔCT.
Western blotting
For this analysis we used specimens from 38 of the 44 patients. The control group was represented by the same samples used for gene expression. The samples were macerated using TissueLyser (Qiagen®) equipment with RIPA lysis buffer (Lysis Buffer, Millipore) containing protease and phosphatase inhibitors. The samples were centrifuged for 20 minutes at 15,000 rpm and 4°C. The supernatant was collected and mixed with sample buffer in a 1:1 ratio. The protein samples at a concentration of 100 ng/mL were loaded onto 12% acrylamide gels (Bio-Rad; Mini-Protean TGX) and subjected to electrophoresis. The proteins were transferred to polyvinylidene fluoride (PVDF) membranes at 300 mA for 2 hours. Antibody incubation was performed using the SNAP apparatus (SNAP id system — Millipore). Each membrane was blocked with 1% bovine serum albumin (BSA, Sigma) diluted in Tris-buffered saline-Tween 20 (TBS-T) for 15 seconds. The following primary antibodies were diluted in BSA, as suggested by the manufacturer: anti-ERK5 (1:1000, Upstate), anti-KRAS (1:500; Abnova), anti-MAP3K3 (1:1000, Upstate), and anti-MAP4K4 (1:500; Abnova). The membranes were incubated for 10 minutes at room temperature, followed by incubation with the secondary antibody. We used an antibody against β-actin (1:500, Millipore) to normalize the protein loading. Lastly, the blots were subjected to detection using the Alliance 4.7 device (Uvitec Cambridge). A quantitative analysis was performed using Alliance 16.06 software.
Statistical Analysis
A statistical analysis was performed using the SPSS 19.0 software. For the comparison of gene, miRNA, and protein expression with clinical variables, we used the Student's t-test for homogeneous variables and the Mann-Whitney U test for non-homogeneous variables. For all statistical analyses we considered a level of significance of 5% (p<0.05).
Results
Analysis of miR-143 and the
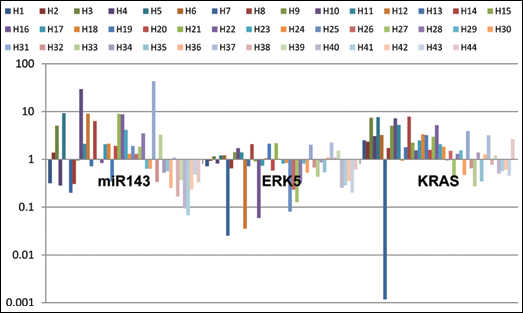
Expression profile of miR-143 and KRAS and ERK5 genes in 44 patients with BPH using qRT-PCR.
In the same analysis, miR-145 was found to be overexpressed in 73.8% of the cases (mean of 3.9-fold), and
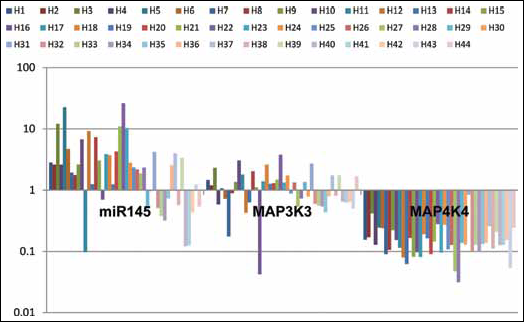
Expression profile of miR-145 and MAP3K3 and MAP4K4 genes in 44 patients with BPH using qRT-PCR.
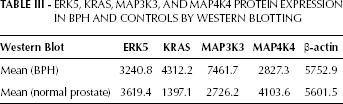
The analysis of the proteins encoded by the ERK5, KRAS, MAP3K3, and MAP4K4 genes is shown in Figure 3. We found a strong expression of the MAP3K3 and KRAS proteins after normalization to the β-actin levels (Tab. III).
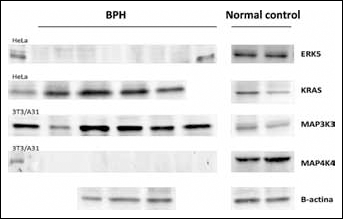
KRAS, MAP3K3, ERK5, MAP4K4, and β-actin proteins in BPH and normal tissue samples. The HeLa cell line was used as a positive control for KRAS and ERK5, while the 3T3/A31 cell line was used for MAP3K3 and MAP4K4.
ERK5, KRAS, MAP3K3, AND MAP4K4 PROTEIN EXPRESSION IN BPH AND CONTROLS BY WESTERN BLOTTING
The only statistically significant correlation between ERK5, KRAS, MAP3K3, and MAP4K4 protein expression with clinical parameters was found for ERK5, which was more highly expressed in patients with a prostate greater than 60 grams (p=0.019).
Discussion
Discovered almost 20 years ago, miRNAs have been recognized as molecules that function specifically in post-transcriptional control in most eukaryotic genomes (6). However, their action in BPH has been poorly explored to date. In the present study we found an overexpression of miR-143 and miR-145 respectively in 62.5% and 73.8% of the cases compared to normal prostatic tissue. As these miRNAs target a number of transcriptional factors involved in the differentiation and proliferation of smooth muscle cells (10), the major reason for investigating these specific miRNAs was their considerable increase in smooth muscle fibers, which are known to play an important role in BPH physiopathology (12). BPH involves high cell proliferation; thus, the overexpression of these miRNAs may be related to the cellular proliferation found in BPH.
In cardiac muscle, miR-143 and miR-145 can regulate the quiescent versus proliferative phenotype in smooth muscle cells. Moreover, miR-145 is instrumental in defining the fate of smooth muscle and is associated with a contractile phenotype (13). Our hypothesis is that these miRNAs may function in both increased stromal proliferation in prostatic tissue and in the induction of a contractile phenotype, which has been shown in BPH. There are several reports in the literature that show the involvement of miR-143 and miR-145 in modulating cellular activities that are important in cardiac morphogenesis (13), in maintaining aortic smooth muscle cells (14), and in modulating the response of cytoskeletal smooth muscle cells to injury (15). The strong expression of miR-143 and miR-145 in human corneal epithelium has been linked to the regulation and formation of epithelium and to maintaining its integrity (16).
Growth factors are recognized as potent stimulators of cell proliferation in the normal prostate and in BPH, and this important group of peptides and their cellular receptors are found at higher amounts in BPH specimens when compared to normal prostates. The growth factor TGF-β uses distinct pathways for the transcriptional activation of miR-143 and miR-145 in smooth muscle cells in the human coronary artery (17), potentially explaining the overexpression of these miRNAs found in our work.
The expression of miR-143 and miR-145 is reduced in cells of colorectal tumor origin when compared to normal colon epithelia (11). The present study also identified KRAS as a protein target of these miRNAs and revealed that the main mediators of the oncosuppression by miR-143 and miR-145 are genes belonging to the growth factor receptor-mitogen-activated protein kinase network and to the p53 signaling pathway. We can infer that overexpression of these miRNAs appears to be only observed in normal tissues, suggesting that these miRNAs are associated with processes of neoplastic transformation.
The overexpression of miR-143 and the decreased expression of
Despite the decreases in ERK5 gene and protein expression in most cases of BPH, we found an increased protein expression in prostates greater than 60 grams when compared to smaller prostates (p=0.019). The presence of the ERK5 protein has been associated with the stimulation of proliferation in prostate cancer, which may also be reflected in our results, since the presence of this protein allows the activation of factors related to cell growth (22). Our results demonstrate that this signaling pathway may be important in the pathophysiology of BPH.
The
The
MAPKs play important roles in regulating immune responses and immune cell development in addition to activation, differentiation, and survival. The MAP3K3 protein is constitutively expressed in T lymphocytes (28), and T effector lymphocytes are found near the prostatic ducts approximately 6 times more frequently in men over 55 years old. This finding may indicate that an autoimmune phenomenon generated by some specific antigens appears in the prostate over time (24). As a result, the microenvironment surrounding the prostate epithelial cells would be flooded by lymphokines that can influence the local production of growth factors and angiogenesis in prostate tissue (29). Thus, MAP3K3 overexpression in T lymphocytes could contribute to this process and to the induction of angiogenesis. In accordance with these findings, we detected the MAP3K3 protein in all the BPH samples examined.
Conversely,
Although the pathogenesis of BPH is not yet fully understood, the present study allows us to confirm that miRNAs may be involved in disease development. Further studies with other miRNAs are likely to provide important information for the understanding of this disease and the development of new potential therapies.
In conclusion, miR-143 and miR-145 are overexpressed in patients with BPH, and the latter of these miRNAs may act through the inhibition of MAP4K4.
