Abstract
Non-small cell lung cancer (NSCLC) is a leading cause of death worldwide. The upregulation of the epidermal growth factor receptor (EGFR) due to mutations has been observed in a number of cancers, and tyrosine kinase inhibitors (TKIs), such as gefitinib and erlotinib, which specifically target EGFR signaling, have been used to treat NSCLC patients. The presence of EGFR mutations was previously shown to confer sensitivity to TKIs. In this study, we evaluated the correlation between EGFR mutations and response to erlotinib in Chinese NSCLC patients. We recruited 36 patients with stage IIIB/IV NSCLC who had failed first-line chemotherapy, and treated them with erlotinib. We used immunohistochemistry to determine EGFR expression, and we screened for mutations using PCR analysis. We used Cox regression analysis and Kaplan-Meier curves for survival analysis. We found that 8 patients had exon 19 mutations, while 3 patients had exon 21 mutations. An Eastern Cooperative Oncology Group (ECOG) grade of 2 was a significant negative predictor of overall survival (OS). Patients with EGFR mutations showed a significantly better OS compared to those without EGFR mutations. Additionally, multivariate analysis showed that erlotinib-treated stage IV patients had a significantly longer progression-free survival (PFS) compared to stage IIIB patients. Patients with EGFR mutations also had a significantly better PFS compared to those without EGFR mutations. The overall remission rate (22.2%) and disease control rate (75%) were significantly higher compared to the rates after second-line chemotherapy (<10%). In conclusion, the presence of EGFR mutations could be a marker to predict the therapeutic efficacy of erlotinib and the prognosis in Chinese NSCLC patients.
Keywords
Introduction
Lung cancer is a leading cause of death and is estimated to be responsible for 1.3 million deaths annually (1). Based on biological characteristics, therapy, and prognosis, lung cancer has been classified as small cell lung cancer (SCLC) or non-small cell lung cancer (NSCLC), and NSCLC has been shown to account for 80%-85% of all lung cancers (2). The poor prognosis of NSCLC has been attributed to the fact that most patients are diagnosed at an advanced stage when surgery is not possible (3). The current first-line treatment for advanced NSCLC consists of a platinum-based two-drug combination (4), while docetaxel, pemetrexed, erlotinib, and gefitinib are the commonly used agents for second-line chemotherapy (4–7).
Understanding the molecular mechanisms underlying the occurrence and progression of various cancers had led to the development of therapeutic strategies that target biochemical or signaling pathways that are differentially activated in malignant cells. Activation of protein kinases, such as the epidermal growth factor receptor (EGFR), has been observed in a number of cancers including lung carcinomas (8), and this activation is sometimes due to somatic mutations (9). Researchers have found a correlation between EGFR activity and disease progression, poor prognosis, and resistance to chemotherapeutic agents in NSCLC patients (10–11). Strategies to treat NSCLC include the use of tyrosine kinase inhibitors, such as gefitinib and erlotinib, that specifically target EGFR signaling (12). The tyrosine kinase inhibitors of EGFR have been shown to suppress signaling by competing with ATP for binding to the tyrosine kinase pocket of the receptor (7, 13, 14). Although the exact mechanism of action is not understood, NSCLC cells treated with tyrosine kinase inhibitors exhibit suppression of the EGFR-mediated AKT and MEK-ERK signaling, and induction of apoptosis (15).
The response to gefitinib and erlotinib depends on a number of demographic and clinical factors such as ethnicity, gender, history of smoking, and tumor pathology as well as genetic factors such the presence of activating mutations in the EGFR gene (14, 16–18). A number of clinical studies showed the benefit of using tyrosine kinase inhibitors as first-line therapy in EGFR-mutant NSCLC adenocarcinoma patients (19). High blood concentrations of gefitinib and erlotinib correlated with significantly longer progression-free survival (PFS) periods in NSCLC patients (20–24). In addition to the improved therapeutic efficacy, tyrosine kinase inhibitors also have an improved tolerability and result in improved quality of life compared to standard chemotherapeutic regimens (7, 16).
Although erlotinib was shown to prolong survival in NSCLC patients when used as second-line treatment (7, 22), there is limited information on long-term survival in these patients. In the present study, we investigated the relationship between EGFR mutations, clinical characteristics, and therapeutic efficacy of erlotinib in Chinese patients with advanced NSCLC. We also aimed to identify predictors of therapeutic efficacy of erlotinib and to evaluate the effect of erlotinib on long-term survival (6 years of follow-up) in Chinese NSCLC patients.
Patients and Methods
We recruited a total of 36 patients who presented with stage IIIB/IV NSCLC at the Tongji Hospital, Tongji Medical College of Huazhong University of Science and Technology between June 2006 and December 2006. All study patients had failed to respond to first or second-line chemotherapy. The inclusion criteria were: (i) histological confirmation of advanced, unresectable NSCLC (stage IIB/IV), which was recurrent or had metastasized; (ii) presence of at least 1 evaluable lesion; (iii) age >18 years; (iv) Eastern Cooperative Oncology Group (ECOG) Performance Score of 0-2; (v) predicted survival time of at least 12 weeks; (vi) patients had received at least 1 course of systemic chemotherapy or radiotherapy, but were not recruited into clinical trials of EGFR inhibitors; (vii) no more than 2 chemotherapeutic regimens, and patients had recovered from the toxicity of previous therapies (Common toxicity Criteria [CTC] ≤1). The time from last therapy to this recruitment was at least 3-4 weeks (14 days for Vinorelbine and other vinca alkaloids or gemcitabine); (viii) absence of history of collagen diseases or xerophthalmia; (ix) granulocyte count ≥1.5×109/L and platelets count >100×109/L; (x) serum bilirubin ≤1.5 times of upper normal limit (ULN); (xi) AST and ALT ≤2 times of ULN (≤5 times of ULN for patients with liver metastasis); (xii) serum creatinine ≤1.5 times of ULN or creatinine clearance rate ≥60 mL/min; (xiii) patients compliable to examination and follow-up; (xiv) negative pregnancy test at 72 hours before study; (xv) patients of child-bearing potential willing to be on contraceptives. Patients with a history of central nervous metastasis or spinal cord compression due to malignancies, who had received and completed their therapy, and who had evidence of stable disease for at least 2 months were also included in the trial.
Exclusion criteria were: (i) presence of a systemic disease such as an active infection, hypertension of grade 4, unstable angina, congestion heart failure, liver, kidney or metabolic diseases; (ii) patients who had received anti-tumor therapy with HER1/EGFR inhibitors (small molecular or monoclonal antibody) before recruitment; (iii) presence of malignancies within the past 5 years (exclusion of cured cervical carcinoma in situ, basal cell skin cancer, and squamous cell skin cancer); (iv) patients with newly diagnosed brain metastasis or spinal cord compression, and/or patients who had not completed the surgery and/or radiotherapy treatment for brain metastasis or spinal cord compression; (v) patients with eye abnormalities, severe dry eye syndrome, Keratoconjunctivitis sicca, Sjögren syndrome, severe exposure keratitis or any disease with the potential to increase damage to the corneal epithelial cells; (vi) patients for whom oral medication was infeasible, and intravenous high-energy nutrition was required; patients with a history of surgery that could influence gastrointestinal absorption; or patients with active gastrointestinal ulcers; (vii) patients who were breastfeeding. The primary end point of this study was overall survival (OS), while the secondary endpoint was PFS.
The study was approved by the IRB of Tongji Hospital and written informed consent was obtained from patients before the study.
Study design
Patients were treated with erlotinib at 150 mg once daily at pre-designed time points each day. The dose was adjusted to 100 mg/d or 50 mg/d in patients with toxic reactions of III or IV degrees. Treatment was discontinued when: (i) disease progression was observed during the treatment period; (ii) patients developed toxic side effects that did not resolve at 2 weeks after treatment discontinuation; (iii) patients developed complications or adverse events, did not conform to the study regimen, failed to return for follow-up visits or discontinued treatment.
Patients were allowed to withdraw from the study when radiotherapy was required due to disease progression. The following supportive therapies were administered: (i) bis-phosphonates to control the pain; (ii) eye lubricant when patients had dry eye symptoms (when the Schirmer's test was abnormal before the study); (iii) the physicians were cautious when concomitant warfarin or coumarin drugs were administered, and the international normalized ratio was monitored closely; (iv) other antitumor drugs (biological/immunotherapy with cytotoxic drugs or radiotherapy at complete dose) were administered only when disease progression was confirmed. After complete discontinuation of erlotinib therapy, patients could receive other therapy according to the judgment of our physicians; (v) patients in the present study could not be treated with any non-antitumor drugs until the therapeutic efficacy had been evaluated (at least 30 days after the last treatment in the present study).
Since patients with contact lenses had an increased risk for adverse effects to the eyes, they were advised to consult the oncologist and ophthalmologist to decide on whether to continue using contact lenses.
Immunohistochemistry for EGFR expression
Tissues were embedded in paraffin and 5-μm sections were prepared and deparaffinized in xylene. The sections were washed in PBS and treated with 3% hydrogen peroxide for 10 minutes to inactivate the endogenous peroxidases. Antigen retrieval was performed in a microwave and the sections were blocked in 10% normal goat serum for 10 minutes before incubating with a 1:50 dilution of mouse anti-human EGFR (Santa Cruz, CA, USA) at 4°C overnight. The sections were washed and incubated with a 1:200 dilution of rabbit anti-mouse IgG at 37°C.
The sections were washed, treated with horseradish peroxidase at 37°C for 30 minutes and visualized with DAB under a light microscope. Sections were washed with double distilled water to stop reaction, dehydrated with ethanol, transparentized in xylene and mounted with neutral gum. EGFR-positive cells were counted under a microscope in 5 randomly selected fields (with a ×100 magnification). EGFR expression was classified based on the proportion of positive cells (low for 5%-25% of positive cells; intermediate for 26%-50%; high for >50% of positive cells).
Spiral CT scanning was performed 7 days before start of the study, at 4 weeks after the first medication, and then once every 4-8 weeks. The therapeutic efficacy was evaluated according to the RECIST criteria and classified as complete remission (CR), partial remission (PR), stable disease (SD), and progressive disease (PD). The total remission rate was calculated as the sum of CR and PR, and disease control rate as the sum of CR, PR, and SD.
Genomic DNA was extracted from embedded tissues to detect EGFR mutations. Briefly, the tissues were deparaffinized with xylene, treated with ethanol to remove xylene and then incubated with OB protease (Mycomebio, Biomedcial Science & Technology Center, P.R. China) at 55°C with continuous shaking for lysis. The samples were incubated at 70°C for 10 minutes in ZZ-II buffer and genomic DNA was purified on Mu-Pu genomic DNA separation columns purchased from Guie Biotech (Guangzou, China). Genomic DNA was subjected to PCR amplification according to the manufacturer's instructions. The primer sequences used for PCR to detect EGFR mutations in exons 19 and 21 are described in the Supplementary Table I. The amplification reaction was performed by subjecting the reaction mix to 35 cycles at 94°C for 1 minute, 45°C for 1 minute, and 72°C for 2 minutes followed by a final incubation at 72°C for 10 minutes. PCR products were sequenced by the Beijing Dingguo Biotech Co., Ltd, and the obtained sequences were searched against Genbank using BLAST.
Demographic Data of the Study Population

Statistical analyses
Data were presented as frequencies. PFS and OS were determined using the Cox regression model and were presented as Kaplan-Meier survival curves. All statistical analyses were two-sided and a p value of <0.05 was considered statistically significant. All analyses were performed using SPSS version 16.0 software (SPSS Inc., Chicago, IL, USA).
Results
A total of 36 patients with stage IIIB/IV NSCLC were enrolled in this study. The study population comprised 26 males and 10 females. Eleven patients were >65 years old and 21 patients had a history of smoking. There were 23 adenomas, 9 squamous cell carcinomas, and 4 bronchioalveolar carcinomas. Thirty of the patients had a stage IV NSCLC. Our immunohistochemistry results showed high EGFR expression in 23 of the patients and low EGFR expression in 13 patients. Mutation analysis showed that 25 patients had no EGFR mutations, while 8 patients had a deletion mutation in exon 19 (del E746-A750) and 3 patients had a mutation in exon 21 (missense mutation L858R). The pathology results, ECOG grades, disease stages, and EGFR expression levels are summarized in Table I.
By univariate analysis, we showed that ECOG grade, stage of disease, presence of EGFR mutations, and EGFR expression levels were all associated with OS in erlotinib-treated NSCLC patients (Tab. II). We included ECOG grade and presence of EGFR mutations in our multivariate Cox regression analysis. Our results showed that an ECOG grade of 2 was a significant negative predictor of OS (95% CI: 1.67-12.47, p=0.0031; Tab. II). Patients with an EGFR mutation also showed a significantly better OS (95% CI: 0.14-0.75, p=0.0083) than those without EGFR mutations (Fig. 1).
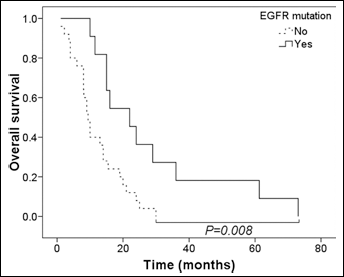
Kaplan-Meier analysis of overall survival in association with EGFR mutations. The overall survival curves of patients with (solid line) or without (dashed line) EGFR mutations were compared.
Cox Regression Analysis of Factors Associated with the Overall Survival of Patients Treated with Erlotinib
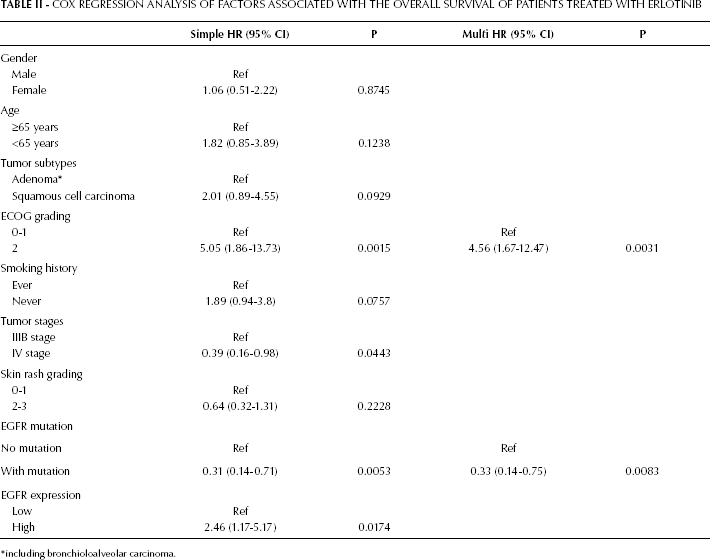
including bronchioloalveolar carcinoma.
We used multivariate analysis to evaluate the association between PFS and the presence of EGFR mutations. We showed that patients with EGFR mutations had a significantly better PFS compared to those without EGFR mutations (95% CI: 0.18-0.82, p=0.0137; Tab. III; Fig. 2).
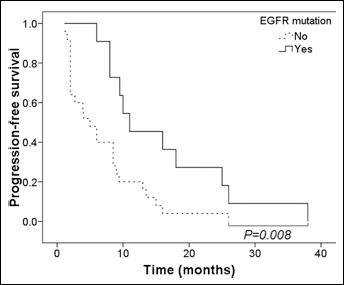
Analysis of progression-free survival in association with EGFR mutations. The progression-free survival curves of patients with (solid line) or without (dashed line) EGFR mutations were compared.
Cox Regression Analysis of Factors Associated with Progression-Free Survival of Patients Treated with Erlotinib
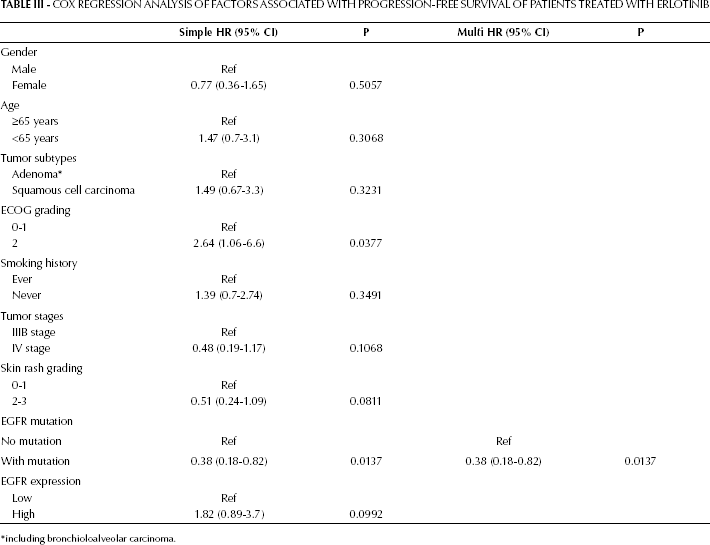
including bronchioloalveolar carcinoma.
Discussion
In this study, we recruited Chinese patients with stage IIIB/IV NSCLC, who had failed to respond to first or second-line chemotherapy and treated them with erlotinib. Our results showed an overall remission rate of 22.2% and a disease control rate of 75%, which were significantly higher than the rates after second-line chemotherapy (effective rate of <10%). We also showed that patients with EGFR mutations had a significantly better PFS compared to those without EGFR mutations. Our findings suggest the possibility of using erlotinib as second or third-line therapy to treat NSCLC stage IIIB/IV patients.
We showed the lack of correlations between remission rate and age, gender, ECOG score, cancer stage, second/third-line therapy, pathological characteristics, history of smoking, and presence of skin rash. Additionally, there was no correlation between disease control rate and age, gender, cancer stage, pathological characteristics, history of smoking or second/third-line therapy. However, the disease control rate in patients with an ECOG score of 0-1 was significantly higher compared to patients with an ECOG score of 2 (82.8% vs. 42.9%, p=0.029). Patients with ECOG scores of 0-1 also had a significantly longer median PFS and OS compared to patients with ECOG scores of 2 (8.5 months vs. 6.0 months; and 15 months vs. 8 months, respectively, both p<0.05). However, there was no correlation between the median PFS and age, gender, pathological characteristics, history of smoking, second/third-line therapy, and skin rash. Our data suggested that patients with a favorable general condition may have a longer period of disease control. In the present study, the median OS time was 13.0 months (95% CI, 7.1-18.9 months), and the median PFS was 8.0 months (95% CI, 5.0-10.9 months).
Our data are consistent with those of the TORCH study (22), which showed that the therapeutic efficacy of erlotinib as second-line therapy was superior to using it as first-line therapy. Asian ethnicity, adenocarcinoma, and history of not smoking were previously shown to be independent predictors of survival in NSCLC patients treated with erlotinib (7). Our study population comprised only Chinese patients, but we found no correlation between smoking status, adenocarcinoma, and survival. This difference could be due to the small sample size of our study. We aim to validate our findings using a larger sample size.
The incidence of EGFR mutations has been shown to be population-dependent, with a significantly higher rate among Japanese and Chinese patients with lung adenocarcinomas compared to Caucasian patients with the same condition (13, 23). Other factors such as female gender and no smoking history also correlated with a higher rate of EGFR mutations (23, 24). In this study, of the 11 patients with EGFR mutations, 72.7% had mutations in exon 19 and 27.3% had mutations in exon 21; these figures agree with previous studies showing that the most common EGFR activating mutations are base-pair deletions in exon 19 and a point mutation (L858R) in exon 21 (25, 26).
EGFR mutations were previously shown to play a role in the therapeutic efficacy of tyrosine kinase inhibitors in NSCLC patients (17, 27–31), suggesting that they can be used as an indicator to predict the therapeutic efficacy of gefitinib and erlotinib (13, 14, 32). NSCLC patients selected on the basis of their EGFR mutations showed significantly better PFS on gefinitib compared to standard chemotherapy (19). In this study, erlotinib-treated patients with EGFR mutations had an effective remission rate of 45.5% and the disease control rate was as high as 100%. Since the effective rate of second-line chemotherapy is about 10%, our data strongly suggest that NSCLC patients with EGFR mutations would benefit from receiving erlotinib rather than standard chemotherapy as second or third-line therapy. Although the mechanism of action remains unclear, patients with exon 19 mutations were reported to show better response rates and longer survival after tyrosine kinase inhibitor therapy, compared to patients with exon 21 mutations (33). It will be interesting to further stratify our data to compare specific mutations with response rates.
Based on our data, we suggest that screening for EGFR mutations in patients who fail to respond to chemotherapy can help determine the feasibility of using a tyrosine kinase inhibitor as second-line therapy. Although erlotinib is currently widely used as second or third-line therapy after chemotherapy, independently of the mutational status, our data suggest important cost and efficacy benefits in testing for EGFR mutations, which would result in sparing some patients from being subjected to a difficult treatment regimen to which they may not respond. EGFR mutations were previously shown to play a role in the therapeutic efficacy of tyrosine kinase inhibitors in NSCLC patients. However, to the best of our knowledge, we are the first to evaluate long-term survival in erlotinib-treated patients carrying EGFR mutations.
It is interesting to note that not all NSCLC patients with EGFR overexpression are responsive to erlotinib therapy, making it important to further investigate the mechanisms underlying the role of EGFR mutations in therapeutic efficacy of tyrosine kinase inhibitors. One important limitation of our study is that it is a single center study with a small sample size. We would like to validate our findings in a larger study population.
Conclusion
In summary, we showed the presence of EGFR mutations in about one third of our Chinese NSCLC patients. Patients with EGFR mutations who were treated with erlotinib had a higher disease control rate, longer duration of PFS and longer OS compared to patients without EGFR mutations. The presence of EGFR mutations may therefore serve as a marker to predict the therapeutic efficacy of erlotinib and the prognosis of NSCLC patients.
