Abstract
Background
Bioactive glass has attracted substantial interest in orthopedics, but it has been less explored as a drug carrier. This study investigated the bovine serum albumin (BSA) release from bioactive 13–93B0 and 13–93B3 glasses.
Methods
Glass disks (13–93B0 and 13–93B3; n = 5) were loaded with 4 mg of BSA and coated under different chitosan-coating conditions. The amount of BSA released in phosphate-buffered saline (PBS) was evaluated, and a degradation study was performed to find out the weight loss and pH of PBS. Secondary structures of BSA on 13–93B0 were characterized by Fourier transform infrared (FTIR) spectroscopy.
Results
One hundred percent protein release occurred by 24 hours for all 13–93B3 groups. However, chitosan coating delayed 100% release up to 72 hours in 13–93B0 groups. The 13–93B3 glass showed higher degradation rates than 13–93B0 regardless of chitosan-coating status. Multilayer and sandwich chitosan coatings further delayed BSA release from 13–93B0. FTIR analysis revealed that α-helical structure was the highest among all groups and significantly higher in the 2% sandwich chitosan coating group (32.0% ± 2.1%), compared with uncoated and 4% chitosan groups.
Conclusions
Chitosan coating can delay the burst release of BSA from 13–93B0 glass and be a potential coating on bioactive glass for drug delivery purposes.
Introduction
Bioactive glass has attracted substantial interest in orthopedics because of its biocompatibility, degradability and ability to bond with host tissues (1, 2). Bioglass® 45S5 is still best known for its excellent bone-bonding ability, but the difficulty of processing 45S5 into a 3D porous scaffold makes bioactive glass 13–93B0, which is also silicate based, more favorable in viscous flow sintering (3). A borate glass 13–93B3, obtained by substituting all of the SiO2 in 13–93B0 glass with B2O3, has been demonstrated to convert faster to hydroxyapatite than 13–93B0 (4). Both 13–93B0 and 13–93B3 have been studied as scaffold materials or injectable bone cements for bone regeneration for non-load-bearing areas (5–7).
Unlike other bone substitute or filler materials, such as polymers and calcium phosphates, bioactive glass has so far received less attention for drug release purposes. Different antibiotics or other pharmaceutical molecules can be directly added into the solution of the sol-gel glass or loaded on glass powders or solid glass surface (8, 9). Recently, bioactive glass has been further explored to deliver growth factors such as bone morphogenetic protein 2 (BMP-2) and vascular endothelial growth factor (VEGF) for promoting bone regeneration (10–12); however, the changes in secondary structures of these growth factors interacting with bioactive glass are rarely investigated.
Among various non-covalent binding methods, passive adsorption of proteins on the surface is the most commonly used approach in the drug delivery of bioactive glass. Protein interactions with materials can be influenced by the surface charge of the materials and isoelectric points of proteins (13, 14). In addition, the electrostatic interactions of the proteins and materials can destabilize the natural conformation of these proteins (15, 16). The surface of bioactive glass in hydrated state is known to form a high density of negatively charged ions such as silanols (Si-O–). However, the conversion of bioactive glass surface into apatite in vitro can affect the adsorption and release, due to an increase of surface area and the less negatively charged apatite layer (11, 15, 17, 18). Furthermore, conformational changes of the protein structures leading to the loss of bioactivity occur with adsorption onto the material surfaces (19). Thus, it is of scientific interest to investigate the release and changes of the protein structures on bioactive glass.
Surface coating is an easy and dynamic approach to provide an immediate protection and modification of the surface properties of a material. Although various polymers have been studied as coating materials to prolong drug release, chitosan is of interest due to its biodegradability, biocompatibility and solubility in both water with inorganic or organic acids. For orthopedic applications in particular, chitosan is demonstrated to facilitate the attachment and proliferation of preosteoblasts and stimulate osteogenic differentiation of mesenchymal stem cells (20,21). Chitosan has been shown to have the potential as a coating material in orthopedics due to its own antimicrobial property or as a carrier of antibiotics on titanium or stainless steel implants (22, 23).
In the present study, chitosan coating was examined to explore its effects on the release and secondary structures of bovine serum albumin (BSA) loaded into bioactive glasses. BSA was chosen as it is a model protein which has been widely investigated for release and secondary structures. Silicate-based glass 13–93B0 and borate-based glass 13–93B3 were studied with chitosan of 2 degrees of deacetylation. Degradation study was also performed on 13–93B0 and 13–93B3. Secondary structures of the BSA on the bioactive glass 13–93B0 were characterized by Fourier transform infrared (FTIR) spectroscopy and further analyzed using Gaussian peak analysis.
Materials and Methods
Disk fabrication
Glass disks were fabricated from bioactive glass 13–93B0 (BO) and 13–93B3 (B3) by pellet pressing and sintering. The chemical compositions of these 2 types of glasses share in common are 4% P2O5, 20% CaO, 5% MgO, 6% Na2O and 12% K2O (in weight %), while BO is a silicate (53% SiO2)-based and B3 is a borate (53% B2O3)-based glass; the glass preparation is reported in detail in the literature (24).
BO and B3 glass particles of size 75–150 μm were used for this study. Glass disks were made by putting 450 mg glass powder into a 13 mm evacuable pellet die and pressing with 2 tf. The resulting disks were sintered using a tube furnace (Lindberg/Blue M, Waltham, MA, USA) at 650°C (BO) or 540°C (B3) at a heating rate of 5°C/min with no holding time. BO and B3 disks were sintered at a lower temperature compared with other studies, to avoid complete fusion of the glass particles allowing liquid absorption. The sintered disks had a diameter of 12.78 mm and thickness of 1.95 mm for the release and degradation studies; pellets of the same diameter but 1 mm thickness were prepared for BSA loading for FTIR analysis. BO and B3 glass powders were provided by Mo-Sci Corporation (Rolla, MO, USA) and other chemicals were purchased from Sigma-Aldrich (St. Louis, MO, USA) unless otherwise specified.
Porosity measurement
The porosity of the BO and B3 glass disks used for the release and degradation studies was estimated by the density of the corresponding glass. Five disks of each type of glass were fabricated as described in the previous section by pellet pressing and sintering. Dimension and weight of each disk was measured by a digital caliper and a digital scale. The mass to volume ratio of the disk was then estimated by the mass of the disk divided by the volume. Porosity of each type of glass was then calculated by dividing the mass to volume ratio over the corresponding density of the glass provided by Mo-Sci Corporation. The densities of BO and B3 are 2.66 kg/m3 and 2.30 kg/m3, respectively.
BSA loading
BSA solution at a concentration of 40 mg/mL was prepared by dissolving BSA with distilled water and sterile filtered the resulting solution with a 0.2 μm syringe filter. Inside a biological safety cabinet, 50 μL of BSA solution was pipetted to the top surface of each disk laid on a Petri dish. The disks were allowed to dry inside the cabinet for 3 hours; the disks were then flipped over, and another 50 μL of BSA solution was added to the bottom surface of the disks. The disks, with a total BSA loading of 4 mg, were allowed to dry inside the cabinet overnight prior to the next process. Plain glass disks without BSA loading were used as the control.
Chitosan coating
Chitosan coating of the glass disks was performed with different concentrations and 2 degrees of deacetylation of chitosan by different coating approaches. The 2 degrees of deacetylated (DEA) chitosan investigated were 80% and 94%. Chitosan powder was dissolved in 1% (w/w) acetic acid solution to prepare 2% (w/w) of 94% DEA chitosan solution and 0.5%, 2% and 4% (w/w) of 80% DEA chitosan solutions. After overnight stirring, a homogenous mixture was achieved. Coating was performed by adding just enough chitosan solution to cover each glass disk and then spreading the solution with a metal spatula on the top surface of the disk. Disks were dried inside a chemical hood for 1 hour and then the same coating procedure was repeated for the bottom surface. Release of BSA from B0 and B3 was studied with coating the disks once with 2% (w/w) of 94% DEA, 0.5%, 2% or 4% (w/w) of 80% DEA chitosan solutions. Release of B0 was further studied with 80% DEA chitosan using different coating approaches. Sandwich coating was done by first coating the disks with a layer of 2% of 80% DEA chitosan solution before BSA loading. BSA solution was loaded on the chitosan coating as previously described, and the disks were then coated with another layer of 2% of 80% DEA chitosan solution. The 3-time coating group was prepared by coating the BSA-loaded glass disks with 2% of 80% DEA chitosan solution 3 times.
In vitro release of BSA
To determine the release kinetics of BSA from the glass disks, the specimens were placed in 50 mL centrifuge tubes with 2 mL sterile phosphate-buffered saline (PBS) and incubated at 37°C. The elution of BSA was examined at 1, 2, 4, 8, 24, 72 and 168 hours by collecting old PBS and replacing with fresh PBS at the corresponding time point. Collected PBS samples were kept at 4°C and analyzed at once. The amount of BSA in the PBS samples was measured by bicinchoninic acid (BCA) protein assay (Pierce, Waltham, MA, USA). BSA solution used for BSA loading was diluted accordingly and used for the BCA protein assay standard curve.
Diffusion Coefficient
To understand the mechanism of BSA release kinetics, Fick's second law is an established model used to estimate the early-stage diffusion (Mt/M∞ ≤ 0.6) of drug release from materials (25, 26). The following equation (Eq. [1]) was used to model the BSA release at the initial stages:
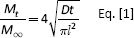
which
Degradation Study
The degradation of the B0 and B3 glass disks with chitosan coating but no BSA loading was evaluated as a function of immersion time of the disks in PBS. Chitosan coatings with 1 time and 3 times 2% chitosan solution of 80% DEA were examined. Before soaking the disks in PBS, the weight of each disk was measured as W0. In vitro degradation was performed with the disks placed in 12-well plates and immersed in the same amount of PBS (2 mL) as in the release study. The plate was incubated at 37°C until the time point (1, 2, 4, 8, 24, 72 and 168 hours) was reached. At the time point, the disks were removed from PBS, dried at 90°C overnight and weighed the following day as WT. Weight loss of the disk was calculated from WT – W0. The collected PBS was cooled to room temperature, and its pH was measured with a pH meter. PBS of the remaining plates for other time points was replaced with fresh PBS at each time point.
Secondary structure characterization
FTIR measurements were conducted with only B0 glass slices with different chitosan coatings using Nicolet 380 FTIR (Thermo Fisher, Waltham, MA, USA) to study the change of BSA secondary structure on glass surface. BSA solution at 250 mg/mL was prepared to load 10 mg of BSA onto each B0 slice. Glass slices were coated with 2%, 4%, 2% sandwich and 3 times 2% chitosan solution with 80% DEA. To prepare background spectra for the FTIR measurements, glass slices without BSA were prepared with the same coating approaches as above. The potassium bromide (KBr) pellet approach was used to prepare the pellet for FTIR measurement; the spectra of BSA loaded pellets were subtracted from the spectra of the corresponding glass slices without BSA. FTIR spectra were smoothed with a 13-point Savitsky-Golay smoothing function. Secondary structures of amide I (1,600–1,700 cm1) were resolved using Gaussian peak analysis using OriginPro (version 8.1; Northampton, MA, USA) software with a linear baseline fitted. In the amide I region, the Gaussian peaks were assigned to distinct secondary structures: 1,600–1,620 cm1- side chain; 1,620–1,640 cm1 and 1,686–1,700 cm-1 - β-sheet; 1,640–1,650 cm-1 - random structures; 1,650–1,660 cm-1 - α-helix; 1,660–1,686 cm-1 - β-turn (27–30). Percentages of area of these peaks were analyzed to find the quantitative contribution of each structure.
Statistical analysis
The results presented in this study are expressed as means ± standard deviation. Statistical analysis of the α-helical content was performed with the Statistical Package for Social Sciences (SPSS 17.0.1; Norusis/SPSS Inc., Chicago, IL, USA) software. Analysis of variance (ANOVA) was applied to calculate the significance level of all data. Statistically significant differences were considered as p≤0.05, and the Bonferroni correction was used if statistical significance was obtained.
Results
Porosity of glass disks
The estimated porosity and other physical parameters of the B0 and B3 glass disks can be seen in Table I. Statistical differences were found in the radius and mass between these 2 groups of glasses, but no statistical difference was found in the mass to volume ratio, which was later used for the porosity calculation. Based on the mass to volume ratio measured from the disks, the porosity of B0 and B3 was found to be 66.77% ± 1.32% and 77.38% ± 0.97%, respectively.
Physical characteristics of the BO and B3 glass disks

No statistical analysis was performed on the estimated porosity.
p<0.01.
Release from B0 and B3 glasses
Release of BSA from B0 and B3 disks was performed with 80% and 94% DEA chitosan to study the effects of chitosan coating on these 2 types of glasses. Cumulative BSA release from BO and B3 disks at all time points is shown in Figures 1 and 2, respectively. For all B3 groups, 100% BSA release occurred by 24 hours, with no obvious difference found between the groups with and without chitosan coatings (as seen in Fig. 2B). On the other hand, 2% chitosan, both 80% and 94% DEA, coatings delayed the total release of BSA from B0 disks to 72 hours. The effect of different degrees of chitosan deacetylation on BSA release was not observed with 2% chitosan coating on B0 glass.
Cumulative release profiles of bovine serum albumin (BSA) from BO disks coated with 2% (w/w) of 94% deacetylated (DEA) chitosan solution and 0.5% 2% and 4% (w/w) of 80% DEA chitosan solutions for 7 days (A) and initial 24 hours (B).
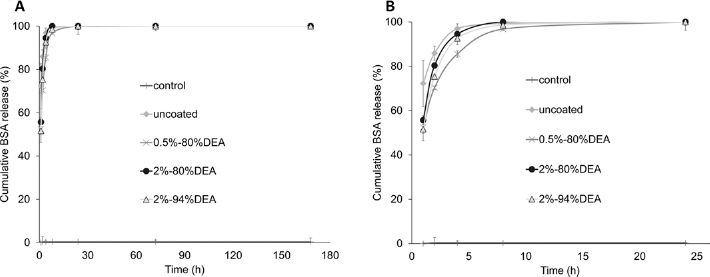
Cumulative release profiles of bovine serum albumin (BSA) from B3 disks coated with 2% (w/w) of 94% deacetylated (DEA) chitosan solution and 0.5%, 2% and 4% (w/w) of 80% DEA chitosan solutions for 7 days (A) and initial 24 hours (B).
Degradation of the disks
The weight loss of B0 and B3 disks and the change in pH of PBS, as presented in Figure 3A and B, were used to determine the degradation of the glasses with or without chitosan coating. The weight loss of most groups followed a similar trend of increasing weight loss over time in the first 24 hours. By comparing the different B0 and B3 groups, it could be seen that the weight loss of B3 groups was higher than that of the corresponding B0 groups with or without chitosan coatings at most time points. Chitosan coating reduced the weight loss of 1 time and 3 times 2% chitosan-coated groups in B0 glass. In contrast, both chitosan-coated groups for B3 glass had greater weight loss than the uncoated group. The pH of the uncoated B3 group was only higher than that of the uncoated B0 group initially, with a minimal difference until 72 hours, but then the pH of B0 was higher than B3 by 0.93 at 168 hours. Chitosan coating could effectively lower the pH of PBS of B0 glass in the first 24 hours; however, there was no clear trend for the use of chitosan coating in the pH of PBS of B3 glass.
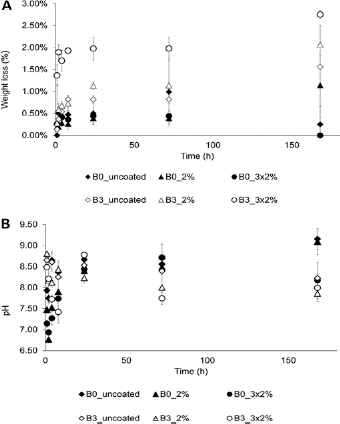
(A) Weight loss of BO and B3 disks, as a function of immersion time in phosphate-buffered saline (PBS), for BO and B3 disks with different chitosan coatings. (B) pH of BO and B3 disks, as a function of immersion time in PBS, for BO and B3 disks with different chitosan coatings.
BSA Release from BO Glass
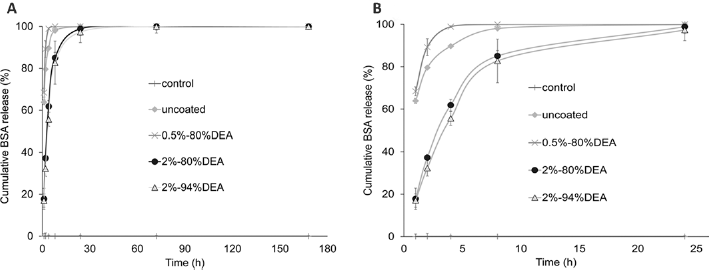
Due to the limited effect of chitosan coating on B3 glass, BSA release was further studied only on BO glass with different coating approaches. The cumulative BSA release shown in Figure 4 clearly demonstrates that different chitosan-coating approaches delayed the release of BSA in various degrees. Release of BSA from the uncoated BO disks was a burst release, with almost total release (98.9%) within the first 8 hours. One layer of chitosan coating could slightly delay the initial burst release of BSA, with slower release achieved by the 4% chitosan coating as compared with the 2% chitosan coating. Sandwich and 3 times 2% chitosan coatings greatly reduced the BSA release in the first hour to about one third that of the uncoated disks. Three times 2% chitosan-coated BO disks had the slowest BSA release rate among all groups.
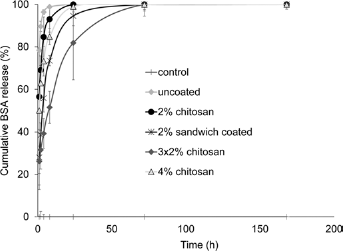
Cumulative release profiles of bovine serum albumin (BSA) from BO disks coated with 80% deacetylated (DEA) chitosan using different coating approaches, for 7 days.
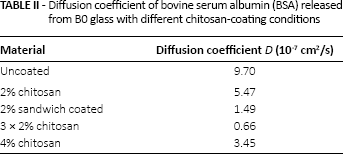
Diffusion coefficients of BO glass
The plots of Mt/M∞ against t1/2 of BSA released of BO and B3 glasses with different chitosan coatings and BO glass with different chitosan coating conditions are shown in Figures 5 and 6, respectively. The curves of all groups of both BSA studies were considerably linear at the initial stage, but diffusion coefficients (as seen in Tab. II) were only calculated with the latter BO study for an in-depth analysis of the BSA release. The diffusion coefficient of BO control (no BSA) was not calculated due to the fact that no BSA was released from this group. The values of the diffusion coefficients of other groups followed the trends of the BSA release with the highest at 9.70 x 10-7 cm2/s and the lowest at 0.66 x 10-7 cm2/s obtained from uncoated and 3 x 2% chitosan groups, respectively.
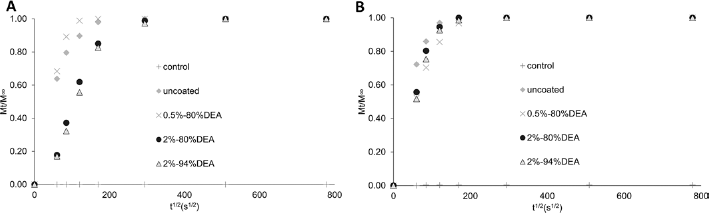
Plot of Mt/M∞ against t1/2 of bovine serum albumin (BSA) released from BO (A) and B3 (B) glasses with different chitosan coatings, over a period of 7 days.
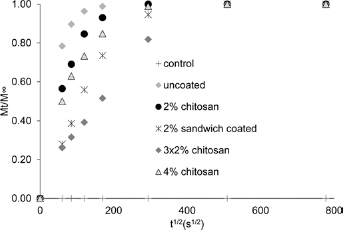
Plot of Mt/M∞ against t1/2 of bovine serum albumin (BSA) released from BO glass coated with 80% deacetylated (DEA) chitosan using different coating approaches, over a period of 7 days.
Diffusion coefficient of bovine serum albumin (BSA) released from BO glass with different chitosan-coating conditions
Secondary Structure Analysis
Five samples from each group were taken for FTIR measurement to determine the structure of BSA on BO disks with or without chitosan coating. In general, 6–12 peaks were resolved in the amide I region by Gaussian peak analysis, with the representative spectra shown in Figure 7. The resolved bands were then classified into corresponding secondary structures with the percentages of areas presented in Figure 8. Among these secondary structures, β-turn, random coils and side-chain are regarded as unordered structures while α-helix and β-sheet are ordered structures; the main indicator of change of protein conformation of BSA is widely suggested to be the α-helical content (27, 31). Considering the use of lyophilized BSA powder as a control, no statistical difference was found between all of these experimental groups and the lyophilized powder. The highest α-helical content was found in the 2% sandwich chitosan-coating group with a value of 32.0% ± 2.1%, which was significantly different from that of the uncoated and 4% chitosan coating groups but not different from lyophilized BSA powder (24%).
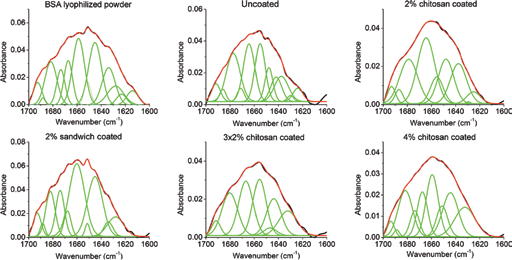
Representative results of the amide I curve fitting of bovine serum albumin (BSA) on BO glass coated with 80% deacetylated (DEA) chitosan using different coating approaches.
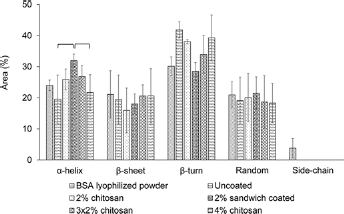
Distribution of secondary structure elements analyzed by Fourier transform infrared (FTIR) spectroscopy. Brackets indicate statistical differences for p<0.05 in the analysis of α-helix only. BSA = bovine serum albumin.
Discussion
There is a growing interest in the use of bioactive glass for repairing non-load-bearing bone defects. Aside from the promising characteristics of 13–93B0 and 13–93B3 glasses for bone regeneration, loading the materials with growth factors such as the osteoinductive BMP-2 has been proven to notably enhance the capacity of the materials to form new bone (17,32). While the use of bioactive glasses as delivery devices for proteins or growth factors has gained less attention, the interaction between these glasses and proteins needs to be explored in more detail.
The mechanism of drug release in a degradable material is controlled by a combination of processes including surface elution, diffusion and degradation of the material. Surface coating with chitosan was applied in this study to delay the release of BSA from the surface as well as diffusion from the core. In the first BSA release study on BO and B3 glasses, a slightly faster BSA release rate was observed in the B3 uncoated group, and the chitosan coating did not show any effect on B3 glass. Fu et al have reported a weight loss of 8% for BO and 67% for B3 at around 200 hours (4); however, the glasses were soaked in simulated body fluid with about 25 times more liquid than this current study. Regardless of the difference in experimental conditions, the weight loss of B3 was still demonstrated to be larger than that of B0 in the corresponding groups, as previously reported. The pH of PBS is assumed to be affected not only by the degradation products of B0 or B3 glass but also by the residual acetic acid in the chitosan coating. The pH fluctuation and larger weight loss of chitosan-coated B3 groups confirmed the faster degradation of the disks and coating, which led to the lack of effect of chitosan coating on BSA release from B3 glass.
Chitosan is an attractive coating material to modify the surface properties and delay drug elution of various materials for different applications with the flexibility of modifiable properties. Degree of deacetylation of chitosan is inversely proportional to its degradation (33, 34). Because they are applied as a thin layer of coating, the difference between 80% and 94% DEA chitosan coatings on BSA release from B0 glass was not influential. High chitosan concentration and multilayer coating techniques are demonstrated to possibly further delay BSA release from B0 glass, but there are also some limitations to these 2 approaches. Solubility of chitosan is affected by the degree of deacetylation, and it is only soluble in acidic conditions, with the exception of 50% DEA (35). The 4% chitosan solution used, which had a workable gel-like consistency, was the highest concentration achieved in this study. While multilayer coating can be easily performed, the added weight and thickness of multilayers may alter the overall geometry and adherence of the coating on the bulk material.
The use of Fick's law in modeling the diffusion behavior of filler or drug release has been applied in dental and orthopedic materials (25, 36). Linearity of the Mt/M∞ (≤0.6) against t1/2 in the initial stage indicates that the release of BSA from all groups of B0 and B3 glasses followed Fickian diffusion. The diffusion coefficients of B0 groups with different chitosancoating conditions are in the range of 0.66 x 10-7-9.70 x 10-7 cm2/s; these values, except those for the 3 x 2% chitosan group, are 1 to 5 orders of magnitude higher than the diffusion coefficients found in dimethacrylate resin–based dental materials (25, 26, 37).
The diffusion coefficients of BO glass investigated in this study are not intended to lead to a direct comparison with polymeric-based materials; however, the degree of difference can demonstrate a much faster release of BSA from BO glass, making it more suitable for delivering drugs, with the concerns regarding the loss of bioactivity of proteins or the development of drug resistance – e.g., in antibiotics.
Understanding the interaction of proteins with material surfaces is of primary importance to consider applying materials as a carrier without sacrificing the bioactivity of proteins. Electrostatic charges of proteins and material surfaces can have effects on the release rate and conformation of the released proteins (14, 16). Proteins adsorbed on the surfaces having the same charge normally have less distortion of protein structures but faster release rates. Thus, sandwich chitosan coating was studied because glass and BSA are negatively charged while chitosan is positively charged (13, 17, 38). The highest α-helical content of 32.0% was found in the sandwich coating group, which fell in the range of an α-helical content of 31%–32.6% as found in lyophilized BSA powder, as reported in the literature (27, 39). In a previous study of electrically induced neutral, positively and negatively charged biphasic calcium phosphate surfaces, the highest α-helical content of adsorbed BSA was found on the unpoled surface, while positively charged surfaces had the lowest α-helical content (13). The lowest α-helical content was found in the uncoated group showing that chitosan, even if it had an opposite charge to the glass surface and BSA, as a coating material provided a certain level of BSA structure retention. Considering that the sandwich coating was done with only a thin anionic chitosan coating on top of the highly cationic glass, perhaps the surface could be detected by BSA as a neutral or less cationic surface with minimal conformational change of the secondary structures. On the other hand, while opposite-charged surfaces may not be favorable for the structures of the adsorbed proteins, the electrostatic attraction is actually beneficial for slower release rates. The release of anionic histone has been shown to be faster from chitosan due to electrostatic repulsion than from opposite-charged cationic alginate or chondroitin-4-sulfate added to chitosan (40). Comparing the 2% chitosan group to the 2% sandwich group, with only 1 layer of chitosan coating applied to cover the BSA loaded in both groups, the slower release in the 2% sandwich group could be taken as an effect of the loading of negatively charged BSA on the positively charged chitosan on the bottom layer. As a result, the favorable results for BSA release and secondary structure analysis of sandwich coating could be explained by the electrostatic interactions of BSA with chitosan and BO glass.
Conclusions
This study investigated the BSA release and secondary structure, as well as the degradation, of chitosan-coated 13–93B0 and 13–93B3 glasses. Parameters such as surface charge, material properties and composition are all critical in the release and conformation of proteins on material surfaces and have to be taken into consideration. Due to the fast degradation of borate-based 13–93B3 glass, chitosan coating did not show any effect on BSA release. However, chitosan coatings –multilayer and sandwich, in particular – reduced the burst release of BSA from silicate-based glass 13–93B0. Moreover, the α-helical content suggests better prevention of BSA unfolding of the sandwich coating on BO glass surface. Based on the observations from this study, we conclude that chitosan coatings can be considered as a potential approach for modifying bioactive glass for drug delivery purposes.
Footnotes
Acknowledgements
The authors would like to thank Mr. Cary Pritchard of the Department of Chemistry and Chemical Biology of Indiana University–Purdue University Indianapolis, for his assistance in FTIR measurement.
Financial support: This project was supported by the Office of Naval Research (contract number N000014-11-M-0113-1).
Conflict of interest: The authors declare that they have no conflict of interest.
