Abstract
Background
Titanium has outstanding corrosion resistance due to the thin protective oxide layer that is formed on its surface. Nevertheless, in harsh and severe environments, pure titanium may suffer localized corrosion. In those conditions, costly titanium alloys containing palladium, nickel and molybdenum are used. This purpose investigated how it is possible to control corrosion, at lower cost, by electrochemical surface treatment on pure titanium, increasing the thickness of the natural oxide layer.
Methods
Anodic oxidation was performed on titanium by immersion in H2SO4 solution and applying voltages ranging from 10 to 80 V. Different anodic current densities were considered. Potentiodynamic tests in chloride- and fluoride-containing solutions were carried out on anodized titanium to determine the pitting potential.
Results
All tested anodizing treatments increased corrosion resistance of pure titanium, but never reached the performance of titanium alloys. The best corrosion behavior was obtained on titanium anodized at voltages lower than 40 V at 20 mA/cm2.
Conclusions
Titanium samples anodized at low cell voltage were seen to give high corrosion resistance in chloride- and fluoride-containing solutions. Electrolyte bath and anodic current density have little effect on the corrosion behavior.
Introduction
Titanium has outstanding corrosion resistance due to the thin amorphous, nonstoichiometric titanium dioxide protective layer (maximum thickness 10 nm (1)). The film is spontaneously formed on the titanium surface when exposed to an aerated environment. This protective layer is very stable and allows titanium to be used in very severe working conditions, such as offshore drilling at up to 260°C, acid environments, in the aerospace industry (2, 3), in high-temperature environments and in the automotive, chemical and food industries (4–5–6).
Nevertheless, commercially pure titanium may suffer different forms of localized corrosion in very harsh environments. Titanium may undergo pitting corrosion in hot, salty water above 200°C as well as in hot hydrogen halides, due to the combined effect of the acidic condition, high temperature and presence of halides. Small amounts of fluorides may dissolve the passive film, due to formation of a titanium soluble salt, TiF4 (7, 8). Titanium may suffer hydrogen embrittlement, occurring on α and α-β titanium alloys due to low hydrogen solubility in α-Ti, and stress corrosion cracking in specific environments, such as anhydrous methanol, nitrogen tetroxide, red-fuming nitric acid or solid cadmium (9).
To face these potential issues, specific alloys containing palladium, nickel and molybdenum were introduced (9, 10). These elements promote cathodic reaction, reducing overvoltages, allowing a polarization of the cathodic curve to more noble potentials, forcing the metal to work in the passive region; consequently, the corrosion rate is greatly reduced (11). However, titanium alloys based on addition of Pd, Mo and Ni are very expensive.
To overcome this problem, the corrosion resistance of pure titanium may be increased by a variety of surface treatments. Nitration, consisting of the introduction of nitrogen into the first micrometers of titanium, promotes the formation of TiN and increases corrosion resistance (12, 13). The thickness of the natural titanium oxide layer, and consequent resistance to corrosion, may be increased by using an electrochemical anodizing treatment. An anodic polarization of several volts, up to 100 V, is applied on the titanium surface in a proper electrochemical bath, promoting the increase of the oxide layer from about 10-20 nm, up to 250-300 nm (14–15–16–17). At voltages higher than 100 V, the anodizing treatment causes the instauration of microarcs in the insulating oxide layer; this regime is called anodic spark deposition (ASD) or plasma electrolytic oxidation (PEO). The titanium oxide is several micrometers thick, but with a porous structure. This growth mechanism happens on different light metals (18) and is reported to increase titanium corrosion resistance (19); nevertheless, due to the high energy consumption, it is rarely used, mainly to dope the oxide film with a very short treatment (20).
Thermal oxidation, typically carried out at temperatures higher than 400°C, accelerates the spontaneous formation of titanium film. The oxide obtained is more crystalline, with the formation of an anatase phase at low temperature and a rutile phase above 600°C (21, 22).
Sol-gel treatment of pure titanium showed a moderate increase of corrosion resistance: due to its low stability and reproducibility, this treatment is not generally considered practical (23). The same considerations are applicable to other coating techniques, such as vacuum plasma spray coating, plasma spraying and chemical vapor deposition. Among all of the available treatments, electrochemical anodizing has been preferred, because of its low cost and the feasibility of the treatment from an industrial point of view.
In this present study, anodic oxidation was performed to tune the TiO2 layer to increase its thickness and to obtain a mostly amorphous phase, with the final aim to increase pure titanium corrosion resistance up to the level of more expensive alloys. The anodic oxide film formed on the titanium strongly depends on the electrolyte composition, the applied voltage, the current density, the temperature and the surface preparation (24). In this work, the effects of the first 3 parameters were investigated, keeping constant temperature and surface preparation. Due to promising preliminary results (25) in increasing corrosion resistance of the anodizing treatment at low voltage, below 100 V, a deeper investigating was performed and is reported here. Furthermore, it is possible to combine the increased corrosion resistance with the possibility to make use of other well-known properties of low-voltage films, such as interference coloration, super hydrophilicity and photocatalyzation.
Materials and Methods
Four titanium alloys were tested: 2 commercially pure titanium samples with different amounts of impurities and 2 alloys containing palladium, nickel and molybdenum. Table I reports the composition of each metal.
Composition of metals used
Samples were tubular in shape. To test external surfaces, tubes were cut longitudinally. Specimens were then covered with an epoxy-based electrical insulator resin, one part with a 2-cm2 area used for testing and a smaller connection region to ensure electrical contact with testing instruments, as visible in Figure 1.

Specimen covered with a green epoxy-based resin: on the left, the 2-cm2 area used for testing; on the right, the bare surface used for electrical connection.
All samples were polished with 600-grit abrasive to ensure the same surface finish. To analyze surface finish effect, the same samples were polished with 2400/4000 grit, and then with 6 µm alumina particles until mirror finish.
Titanium samples were anodized through an electrochemical anodization treatment. Samples were connected to the positive pole of a DC feeder (AimTTi PLH120 DC power supply, able to operate up to 120 V and 0.75 A). The counter-electrode, an activated titanium net, was connected to the negative pole. Anodizing started just after sample immersion. The anodizing bath was a 0.5M sulfuric acid solution. Treatments were carried out at 20 mA/cm2 constant current density varying the cell voltage in the range 10 V to 80 V. At 20 V feeding voltage, tests were performed also at 5 mA/cm2and 50 mA/cm2, to study the effect of the current density. At the same voltage, anodizing treatment was performed in 0.5M Na2SO4, 0.5M (NH4)2SO4 and in 0.5M NH4BF4 to analyze the effect of the bath composition on the quality of the titanium oxide. Anodization parameters were chosen based on previous knowledge about the formed oxide, balancing the need to obtain a thick and protective oxide layer and the necessity to not reach potentials that may lead to oxide crystallization (24, 26–27–28).
Thermal treatment was also considered to oxidize the titanium, which is a well-known process to produce a thick and protective oxide layer (21, 22). Calcinations were conducted at 550°C for 1 hour; this temperature was selected to obtain oxide thickness similar to the one obtained with anodization at 20 V, according to the interference color acquired by the sample after treatment (26).
The corrosion resistance of the anodized titanium samples was determined by electrochemical potentiodynamic tests performed with an EG&G Princeton Applied Research potentiostat/galvanostat. Samples were immersed in a 1 L cell, in 2 electrolytes with increasing halide concentrations: 35-g/L and 100-g/L NaCl, and 1-g/L, 3.5-g/L, 10-g/L, 20-g/L and 35-g/L NaF. All tests were performed at room temperature. Only the 100-g/L NaCl solution tests were also performed at 95°C.
Potentiodynamic tests were conducted starting -0.1 V as the free corrosion potential, measured 30 minutes after sample immersion in the corrosive solution, up to potentials between 1.5 V and 7 V, depending on the corrosive electrolyte. The potential scan was inverted when a 10 A/m2 anodic current density was reached. The potential scan rate was 20 mV/min. A silver/silver chloride in a KCl saturated solution was used as reference electrode. Since the duration of a test did not exceed 6 hours, no sealing was necessary, and neither chemical (ΔpH ≈ 0.05) nor physical (ΔT ≈ 2°C) deviations were detected.
To study the effect of the electrochemical anodizing treatment on the cathodic overvoltages, cathodic potentiodynamic tests were conducted in 10% HCl solution, starting at free corrosion potential, decreasing the potential to -1 V Ag/AgCl, with a potential scan rate of 20 mV/min.
All tests were performed in triplicate, with high reproducibility.
Results and discussion
Corrosion resistance in chloride-containing solution
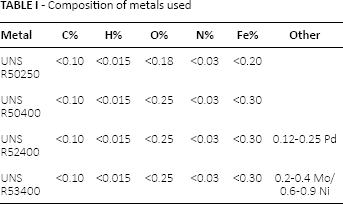
Figure 2 compares the corrosion behavior of the 4 tested titanium alloys in 35-g/L NaCl at 25°C. In each graph, for each alloy, the untreated sample, the electrochemical anodized samples at 20 V and 30 V in sulfuric acid and the thermal anodized sample at 550°C for 1 hour are shown.
Potentiodynamic tests in 35-g/L NaCl at 25°C on untreated titanium, on titanium anodized at 20 V and 30 V, and on titanium thermally treated (TT). (
For both of the 2 commercially pure titanium samples – UNS R50250 (Fig. 2A) and UNS R50400 (Fig. 2B) – those anodized at 20 V showed the lowest anodic current density, and consequently the best corrosion resistance, for almost all of the potential range. In fact, the corrosion current density was 1 order of magnitude lower than that obtained on the untreated commercially pure titanium. Thermal oxidation did not promote any corrosion improvement.
The 2 titanium alloys (Fig. 2C, D) showed a similar trend. The worst corrosion resistance was shown by the untreated samples. In the case of the thermal oxidized sample, an inversion in the corrosion behavior with respect to the untreated one was observed: calcination gave a worsening of corrosion resistance at potentials below 0.7 V.
This is probably due to the less homogeneity of the titanium oxide.
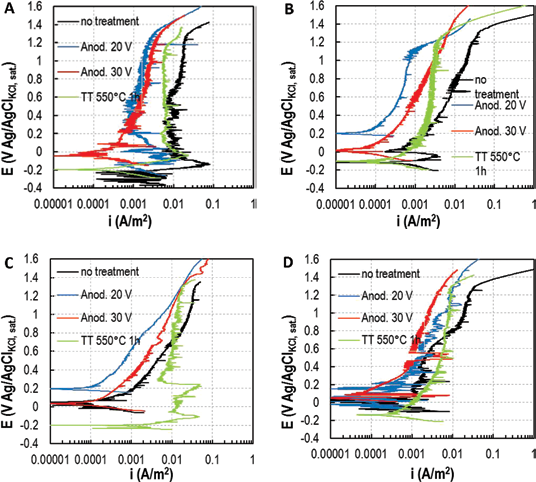
Test were then performed at high temperature, 80°C and 95°C, to favor localized corrosion. At this stage, the roughness effect was also studied by polishing samples until they had a mirror-like finish. Results of commercially pure titanium UNS R50400 tested at 80°C and 95°C with anodizing or polishing treatments are shown in Figure 3. At 80°C, the untreated titanium sample showed a pit initiation at 6.5 V Ag/AgCl, confirmed by the increase of the anodic current density at values higher than 10 A/m2. At 95°C, the pitting potential of the commercially pure titanium was reduced to 5.5 V Ag/AgCl. A corrosion resistance enhancement could be seen both with anodic oxidation treatment at 20 V and with mirror polishing treatment, especially at high potential. Anodization showed better behavior at low potential, which is more interesting for practical applications, while polishing increases the potential at which an anodic current density of 10 A/m2 is reached – that is, pitting corrosion resistance. It is worth noting that the anodic curves became very disturbed at potentials close to the pitting value, due to the formation of meta-pits, in which the material is subject to local failure and successive repassivation.
Potentiodynamic tests on UNS R50400 at high temperatures, with anodization and polishing treatments.
NaCl concentration was raised to 100 g/L, to promote localized corrosion. Nevertheless, no significant variation in the pitting potential was observed: the potential at which the current suddenly increased was between 5.5 V and 6.0 V Ag/AgCl.
Corrosion resistance in fluoride-containing solution
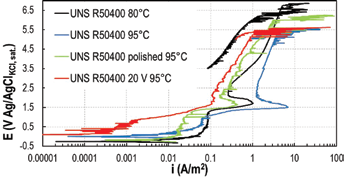
Tests were first performed on untreated commercially pure titanium UNS R50400 with increasing NaF concentrations, to define the proper fluoride content and avoid a very severe solution. NaF solutions from 1 g/L to 35 g/L were tested. Comparisons between untreated titanium and anodized titanium were conducted at low NaF concentrations.
The results are reported in Figure 4. At NaF content higher than 3.5 g/L, passivity of untreated titanium was completely destroyed: anodic curves showed the typical behavior of an active metal with an increasing current density at increasing potential. At NaF concentrations lower than 3.5 g/L, untreated titanium showed a passive behavior, with a lower passive current density at lower NaF concentration.
Potentiodynamic tests of UNS R50400 in different concentrations of NaF with and without 20 V anodization treatment at 25°C.
Potentiodynamic tests on commercially pure titanium electrochemically anodized at 20 V were performed in solution with a NaF content of 1 g/L, 3.5 g/L and 10 g/L. It was clearly visible (Fig. 4) that the anodizing treatment improved the corrosion resistance of the commercially pure titanium. The reason for this was twofold:
At the same fluoride content, the passive current density of anodized titanium is lower than passive current density of untreated titanium. Figure 5 shows the anodic current density measured at 1.5 V Ag/AgCl: the current density of the anodized titanium was more than 1 order of magnitude lower than the value of the untreated titanium.
The critical NaF content to activate passive titanium increased from 3.5 g/L in the case of untreated titanium, to 10 g/L in the case of anodized titanium.
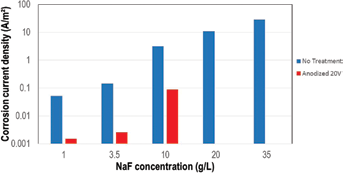
Anodic current density of untreated titanium and titanium anodized at 20 V in different concentrations of NaF at anodic potential 1.5 V vs. Ag/AgCl.
Tests in 3.5-g/L NaF solution were performed to evaluate the effect of the anodizing voltage (voltage range 10-80 V). Each test was repeated 3 times to guarantee reproducibility. Potentiodynamic curves are shown in Figure 6. A slight correlation between electrochemical behavior and anodizing voltage was visible: titanium samples anodized at voltage lower than 40 V appeared more corrosion resistant than samples anodized at higher voltages. However, the behavior of all of the oxidized titanium samples was similar, and corrosion current density was 1 order of magnitude lower with respect to the value measured on the untreated titanium.
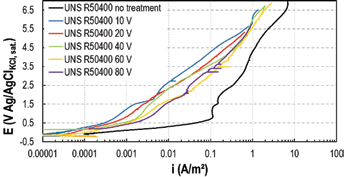
Potentiodynamic tests in 3.5-g/L NaF on UNS R50400 anodized at different voltages at 25°C.
From these results, it appears that titanium oxide layer thickness, which increases proportionally to the increase of the anodizing voltage (for the treatments carried out between 30 nm and 150 nm (24)), is not directly correlated to the corrosion resistance. Conversely, the better corrosion behavior obtained at lower anodizing voltages suggests a possible effect of oxide crystallinity.
Effect of the anodizing current density
Electrochemical anodizing treatments were performed at constant current density, increasing the cell voltage to the desired final value. The parameter
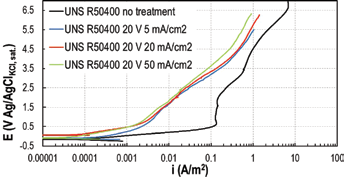
Potentiodynamic tests in 3.5-g/L NaF on UNS R50400 anodized at different current density at 25°C.
Effect of anodizing electrolyte
To investigate the effect of anodization bath, 3 solutions were tested apart from sulfuric acid: 0.5M (NH4)2SO4, 0.5M Na2SO4 and 0.5M NH4BF4. These solutions were chosen to find a treatment suitable for industrial applications; in fact, the use of sulfuric acid solution would have some drawbacks in handling the cells and in the disposal of the acid used. Commercially pure titanium was anodized at 20 mA/cm2 up to 20 V in every considered bath and then tested in 3.5-g/L NaF solution. Results are reported in Figure 8: the electrolytes did not affect the corrosion behavior.
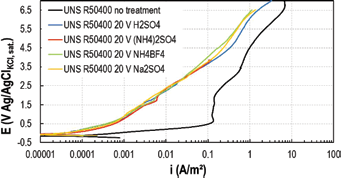
Potentiodynamic tests in 3.5-g/L NaF on UNS R50400 anodized in different electrolytes at 25°C.
Effect of electrochemical anodization on cathodic behavior
Cathodic potentiodynamic curves were graphed to investigate the effect of the electrochemical anodized titanium on the slope of the hydrogen evolution reaction. It is well-known that the enhanced corrosion resistance of the Ti-Pd alloy UNS R52400 is related to the decrease of hydrogen overvoltage given by the addition of palladium to the alloy (29). Tests were performed in 10% HCl. The results are shown in Figure 9. Cathodic Tafel slopes in the order of 130 mV/decade, 150 mV/decade and 170 mV/decade were measured on UNS R52400, UNS R50400 anodized and UNS R50400, respectively. Anodized UNS R50400 has an effect on the cathodic behavior that is between that of the nonanodized titanium and the Ti-Pd alloy; however, the difference in the Tafel slopes was too small to draw any reasonable conclusions.
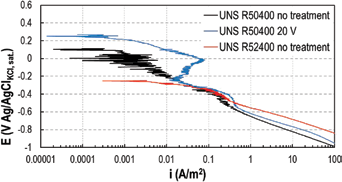
Cathodic curves in 10% HCl at 25°C.
Conclusions
Electrochemical anodization treatment can be used to improve the corrosion behavior of commercially pure titanium. Treated titanium has a higher corrosion resistance in both chloride- and fluoride-containing solutions. The best improvement was achieved at anodizing voltages lower than 40 V, due to the formation of an almost amorphous compact oxide layer. This enhanced corrosion resistance was independent from the anodizing current density and the anodizing bath. Treatments at low current density are therefore preferable because of the lower required energy and cost. However, while a lower current requires less power, a higher current accelerates the process, reducing the time required. Since the anodizing bath has a minimum influence on the quality of the film formed, it is suggested that baths be used at neutral pH – e.g., based on sodium sulfate.
Footnotes
Acknowledgement
Titanium was supplied by A.D. Tubi inossidabili SpA.
Financial support: No grants or funding have been received for this study.
Conflict of interest: None of the authors has any financial interest related to this study to disclose.
