Abstract
Background
Silver ions (Ag+) have strong antibacterial effects, and silver-coated materials are in widespread clinical use. However, the application of silver-coated medical devices is not without concerns: its use with direct bone contact is not established, and systemic toxic side effects of released Ag+ have been described. Therefore, alternative bactericidal coatings with a more localized way of acting – e.g., calcium dihydroxide, Ca(OH)2 (CH) – would be advantageous.
Methods
A new rat model of the animal's tibial metaphysis was developed. In the left proximal tibiae of 36 male Wistar rats, titanium screws were implanted. The screws were coated with hydroxyapatite (HA; 12 animals: group I), low-dosed HA silver (HA-Ag; 12 animals: group II) and CH (12 animals: group III). After 6 weeks, all rats were sacrificed. The implants were evaluated for morphological changes on their surfaces, by light microscopy, scanning electron microscopy and energy-dispersive X-ray spectroscopy; for osteointegration, by measurement of resistance to removal; and for bacterial colonization, by quantitative culture analysis. Additionally, the tibial bone was investigated histologically for signs of osteomyelitis and sonicated to detect bacterial loads.
Results
(i) No microbiological or histological signs of infection could be determined on any of the screws or the surrounding bone. (ii) The bone-implant interface analysis revealed extensive bone formation and direct bone-implant contact on all HA, HA-Ag and HA-CH coated screws. (iii) HA and HA-Ag were partially, and CH was fully, degraded on the screw coating, allowing host bone to osteointegrate.
Introduction
Peri-implant infections are severe complications in musculoskeletal surgery, representing a major health care and economic burden (1). Management of these infections often requires several surgical interventions and the use of long-term antibiotics (2, 3). An unsolved problem in the treatment of peri-implant infections is the formation of biofilm on implanted medical devices (4). Once biofilm has formed on the implant, eradication of infection is only possible if the foreign material is removed (3).
To decrease infection rates, several strategies have been employed, but increasing incidence rates of antibiotic resistance among bacteria make antibiotic-based methods progressively more useless (5–6–7–8–9). Therefore alternative strategies are of more and more interest. Auspicious could be the use of “active” antiinfective coatings able to release bactericidal agents – e.g., silver ions (Ag+) (9–10–11) or calcium dihydroxide (CH) (12–13–14). In comparison with materials used in conventional antibiotic strategies, these substances act more widely against several entities of bacteria. Ag+ acts by interacting with bacterial components such as enzymes, membranes and nucleic acids (10). This bactericidal effect has been confirmed experimentally and also clinically with a relevant decrease of infection rates in revision total joint arthroplasty (15). However, toxic side effects on eukaryotic cells from silver applications have been mentioned and limit its widespread use in total joint arthroplasty (11).
In this context, CH could be a promising alternative. CH has been extensively studied in odontology for years, where it is a widely used substance for canal disinfection, with favorable antiinfective potency (14). CH creates a bactericidal, antiinfective alkaline environment at the level of the wound site by releasing hydroxides and calcium ions. This acting mechanism is localized, and therefore systemic toxic side effects do not appear. Another favorable feature of CH is that its bactericidal potency does not seem to interfere with bone regeneration. In this context, studies in animals and also in humans have shown that the use of CH led to accelerated bone regeneration around the natural tooth after treatment with an oily CH suspension (13). Nevertheless, its use on medical devices at the level of extremity bones has so far not been attempted.
Hence, the aim of the current study was to develop an animal model with a customized screw implant to carry out biocompatibility testing of different coatings with direct bone contact (hydroxyapatite [HA], HA containing silver [HA-Ag] and calcium dihydroxide [CH]).
MATERIALS and methods
Implants
The implant device in the present study was a machined titanium (Ti6Al4V) screw (Fig. 1). The screw was further coated with HA (group I: control group), HA-Ag (group II) and CH (group III) (Atesos Medical AG/Medicoat AG, Bern, Switzerland) according to a manufacturing process previously described (Fig. 1) (16). Briefly summarized, a modified technique of vacuum plasma spray (VPS) coating was used so that a thickness of ∼100 µm was achieved for the HA- and HA-Ag coatings. The HA-Ag coating contained low amounts of Ag (45 parts per billion [ppb]). The CH deposition took place in a solution containing calcium ions (among other ion species). The electrochemical deposition of CH onto the surface of the implants was performed to achieve a final layer thickness of about 20 µm.

(
The morphology of the surface was analyzed by scanning electron microscope (SEM; Hitachi TM-3030 Plus; Hitachi, Berlin, Germany) using a secondary electron detector. The elemental composition of the HA-, HA-Ag- and CH-coated implants was determined by energy-dispersive X-ray spectroscopy (EDX; Quantax 70; Bruker, Billerica, MA, USA). The surface roughness was determined on the apical portion of the implant by a confocal laser scanning microscope (Lext OLS 3100; Olympus, Tokyo, Japan) equipped with a ×50 objective. The arithmetic average roughness (Ra) and Roughness depth (Rz) 1D values were determined by averaging 10 profiles of length 260 µm, using a cutoff wavelength of 26 µm. The SRz 2D values were defined by 5 areas of 295 × 20 µm, also applying a cutoff wavelength of 26 µm.
Animals and operative procedure
The study was approved by the Animal Experimentation Ethics Committee of Bavaria (Reg. No. 146-14). In accordance with the committee's recommendation to minimize the number of experimental animals, the sample size was 12 rats per group. To avoid interactions between the different coatings within the same animal, only the left tibiae were investigated. Hence, 36 male, 5-month-old Wistar rats (Charles River Laboratories, Sulzfeld, Germany) with a mean body weight (BW) of 388.4 g (range 343-411 g) were used.
Surgery was performed under general anesthesia (medetomidine 150 µg/kg BW; midazolam 200 µg/kg BW and fentanyl 5 µg/kg BW).
Animals were prepared for surgery as follows (Fig. 2A–B–C–D–E): The left hind leg was shaved, antisepticized with povidone-iodine, and dried. Bodies were covered with sterile sheets except for the leg. A skin incision (length, 1-2 cm) was made over the proximal lateral tibial metaphysis. A unicortical hole with a depth of ∼8 mm was drilled using a 1.8-mm diameter drill bit. The drill hole was then tapped with a custom-made stainless steel tap and dried with gauze. After removal of the gauze, the implant was immediately inserted into the cavity using a dedicated instrument. Soft tissue was irrigated with saline solution, the fascia was closed using absorbable suture material (Vicryl Rapid, size 6-0; Ethicon Inc., Cincinnati, OH, USA), the skin was closed by continuous intracutaneous (4-0 Monocryl; Fa. Ethicon, Norderstedt, Germany) and interrupted sutures (4-0 Prolene; Fa. Ethicon, Norderstedt, Germany). After the operation, anesthesia was antagonized by a subcutaneous injection of atipamezole 750 μg/kg BW (Alzane; Pfizer, Berlin, Germany), flumazenil 200 μg/kg BW (Flumazenil-Ratiopharm; Ratiopharm, Ulm, Germany) and naloxone 120 μg/kg BW (Naloxon-Ratiopharm; Ratiopharm, Ulm, Germany).
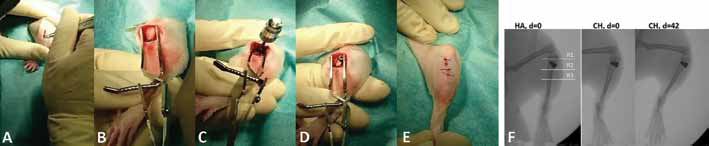
Operative procedure (left tibia) and X-rays: (
The animals were sacrificed after 42 days. Under sterile conditions, tibiae of the left legs were dissected. Swabs were obtained from the subcutaneous tissue and the surface of the implant before removal. All muscle tissue was then removed, and the implant was unscrewed. After removal, the proximal tibia was separated from the distal half and used for further microbiological and histological investigations.
Radiographic evaluation
Radiographs were taken in 2 planes immediately after implantation and on the day of sacrifice. For X-rays, digital films and a Mobilett Plus X-ray unit (Siemens AG, Erlangen, Germany) were used. Three regions of interest (ROIs) were determined and investigated separately (Fig. 2F): R1 (epiphysis), R2 (metaphysis with implant) and R3 (proximal diaphysis). The radiographic scans were evaluated according to an established method, with a minimum score of 0 (well-osteointegrated implant without signs of infection) and a maximum score of 47 (loose implant with severe osteomyelitis) (17).
Evaluation of osteointegration
The implant was removed manually using a sterile custom-made screwdriver (resistance to removal [RTR]) and investigated macroscopically for attached bone remnants (ABRs). A scoring system ranging from 1 to 5 points for either item (RTR and ABRs) was applied (Tab. I; Fig. 3).
Scoring system for resistance to removal (RTR) and attached bone remnants (ABRs)
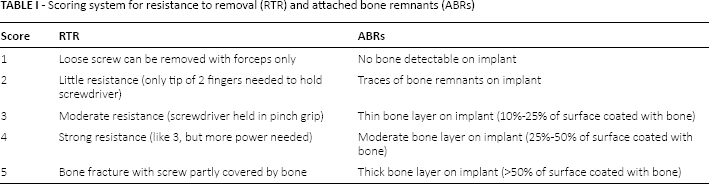

Scoring system to classify bone adherence (red marks) on the retrieved screw implants; 1 = no bone detectable; 2 = spot-like; 3 = thin bone layer (10%-25% of surface coated with bone); 4 = moderate bone layer (25%-50% of surface coated with bone); 5 = thick bone layer (>50% of surface coated with bone).
Histological evaluation
Two randomly chosen tibiae from each group (I, II and III; in total n = 6) were fixed for 2 days in 5% formaldehyde and decalcified in 5% nitric acid solution. The whole proximal half of the tibia was embedded in paraffin and cut into 4-mm-thick sections. Slices were stained with toluidine blue. Three ROIs were determined:
epiphysis,
metaphysis (implant site),
proximal diaphysis.
Qualitative histopathological analysis of the stained slides was performed on all samples using a light microscope and evaluated on the following parameters of infection, according to a modified version of the evaluation approach used by Petty et al and Lucke et al (18, 19):
infiltration of granulocytes;
sequestrum formation;
infiltration of mononuclear cells (lymphocytes, plasma cells, macrophages) and bone marrow fibrosis;
enlargement of cortical bone;
erosion and/or destruction of cortical bone;
general impression.
Parameters 1 to 5 were scored with 0 (absent) or 1 (present). Parameter 6 was scored from 0 (absent), 1 (mild) to 2 (severe). The maximum score was 21 (3 × ROIs for parameters 1-5 × max. 1 point) + (3 × ROIs for parameter 6 × max. 2 points).
Microbiological evaluation
To rule out bacteriological contamination, which would interfere significantly with osteointegration, meticulous microbiological investigations of the implant and the surrounding bone with adjacent soft tissues were carried out.
Examination of bacteriology swabs
Bacteriology swabs from the subcutaneous tissue and the implant site were obtained bilaterally from all animals. Swabs were moistened with 1 drop (20 µL) of sterile phosphate-buffered saline (PBS), then evenly streaked onto a plate each of Columbia Agar and Schaedler Agar, and immersed in thioglycollate broth. The solid and liquid media were incubated for 10 days under the conditions listed below to detect even slowly growing bacteria (e.g., Propionibacterium):
Columbia Blood Agar: 37°C;
Schaedler Agar: 37°C under anaerobic conditions;
thioglycollate broth (prepared in-house): 37°C.
After 10 days, the solid and liquid media were analyzed by conventional bacteriological techniques. The identity of potential isolates was determined by catalase and coagulase testing or by matrix-assisted laser desorption/ionization time of flight mass spectrometry (MALDI-TOF-MS; Bruker Corp., Billerica, MA, USA) if the catalase/coagulase testing was unclear.
Examination of the implant
The implant was immersed in 1 mL of PBS for low-frequency ultrasound treatment using glass beads (5 minutes at 80% intensity; Sonorex Digital 10P; Bandelin, Berlin, Germany). After the treatment, tubes were centrifuged at 4,000 rpm for 10 minutes at 4°C (Heraeus Varifuge 3.OR; Kendro Laboratory Products, Osterode, Germany). The supernatant was resuspended in 300 μL PBS. A serial tenfold dilution of this irrigated PBS was incubated on agar plates at 37°C for 48 hours, and the number of possibly inoculated viable bacteria was then calculated.
Examination of peri-implant bone
The remaining tibia was separated from all soft tissues and ultrasonically irrigated in 10 mL of PBS for 5 minutes. A serial tenfold dilution of this irrigated PBS was incubated on agar plates at 37°C for 48 hours, and the number of inoculated viable bacteria was then calculated.
Statistical Analysis
All results are given as means ± standard deviation (SD), and tests were performed with use of SPSS (version 20.0; SPSS Inc., Chicago, IL, USA). Statistical significance was computed using the following testing procedures: Kruskal-Wallis test, Mann-Whitney U-test and Wilcoxon test. Differences were considered significant for p values <0.05.
Results
Clinical evaluation of animals
The animals recovered quickly after surgery and showed no signs of discomfort or local infection (mean weight gain: 32 ± 11 g for group I, 29 ± 17 g for group II, 37 ± 15 for group III).
X-ray examinations at day of sacrifice
X-rays showed no signs of osteolysis, periosteal elevation or implant loosening at day of sacrifice (Tab. II). The implant was radiographically well integrated and regularly positioned in the tibial metaphysis throughout the study period (Fig. 2F).
Surface roughness and outcomes of radiographic, histological and osteointegration assessments, as well as elemental analysis of chemical surface composition of implants and explants
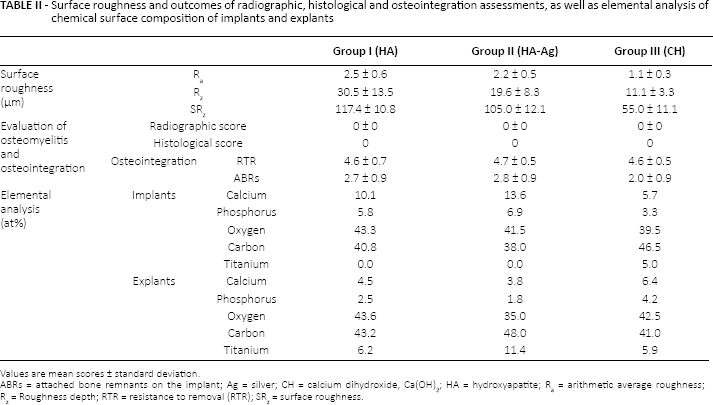
Values are mean scores ± standard deviation.
ABRs = attached bone remnants on the implant; Ag = silver; CH = calcium dihydroxide, Ca(OH)2; HA = hydroxyapatite; Ra = arithmetic average roughness; Rz = Roughness depth; RTR = resistance to removal (RTR); SRz = surface roughness.
Implants
Due to the titanium plasma-spray (TPS) coating of the machined titanium screw, the surface of all 3 implant types showed rough surface morphologies (Fig. 1). The additional HA- and HA-Ag coating led to a rough surface in the samples of group I (HA) and group II (HA-Ag) (Tab. II). The supplementary CH coating (group III) with a thickness of about 5 µm, led to a smooth flower-like decoration (Fig. 4A), with an arithmetic average roughness Ra of 1.1 ± 0.3 µm. Its roughness was less compared with that of groups I and II because no VPS coating of either HA or HA-Ag was present. Similarly, the 2D surface roughness parameter SRz (maximum height) listed in Table II was also lowest in group III. All 3 implant types showed a closed and homogeneous coating (Fig. 4A).
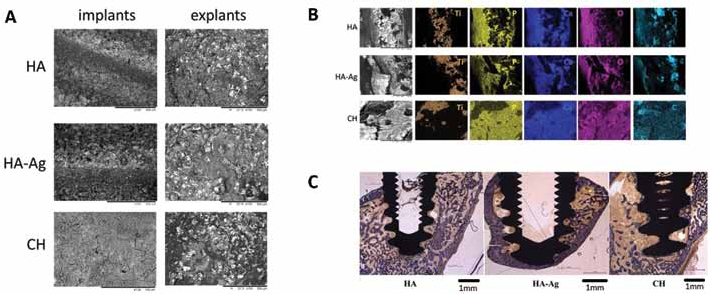
(
The elemental composition of the implants is given in Table II. The thickness and closeness of the surface layers of groups I and II prevented any titanium signal from the adjacent TPS layer and titanium substrate. No silver was found in the HA-Ag coating because the concentration was below the detection limit. Analogous to the HA and HA-Ag surfaces, the CH surface also exhibited significant carbon signals due to atmospheric adsorbates (Tab. II). As the CH coating was built up of Ca(OH)2, mainly calcium and oxygen were found. Traces of titanium (5.0 at%), related to tiny uncoated spots, were detected.
Explants
Compared with the preoperative situation, the surface morphologies of all implants had significantly changed: The surface layers could not be observed anymore on the explants. The coatings had completely disappeared and disclosed the titanium from under the subjacent TPS coating. These TPS coatings are shown as bright zones in the backscattered SEM images of the explants (Fig. 4A). The EDX maps of retrieved explants from all 3 groups clearly identified these bright areas with exposed titanium (shown in orange in Fig. 4B). The elemental composition is given in Figure 4B and Table II. The elemental distribution of calcium, phosphorus and oxygen was at least partially caused by the presence of bone fragments on the explants’ surfaces. Therefore, additional roughness analysis on explants was not performed.
Microbiological evaluation
Retrieved implants and adjacent bone showed no bacterial growth after 10 days of incubation. Additionally, nearly all smears from the entry site were found to be negative. Only 2 smears showed contamination: with Staphylococcus epidermidis (animal no. 3) and Micrococcus luteus (animal no. 16).
Histological evaluation
No specimen showed signs of bone infection in any of the analyzed ROIs (Tab. II). On the other hand, osteoconduction and integration, with no evidence of inflammation or macroscopic particles, were found within the tissue (Fig. 4C). Specimens of all 3 groups had large masses of newly formed bone filling the thread spaces.
Osteointegration
All implants showed good osteointegration, with mean score values ranging from 2 to 5 for RTR and ABRs (Tab. II). Removal of the screws almost always ended up in the destruction of at least small parts of the tibial metaphyses due to strong bone-implant interlinkage.
Discussion
Modern biomaterials intended for use with direct bone contact in the human body have to meet several requirements – e.g., to allow the regeneration of tissue and to lower the risk of implant-related infections. One field with still unresolved problems regarding osteointegration, on the one hand, and antibacterial effects, on the other, is total joint arthroplasty. Until today no antibacterial agents are used routinely with direct bone-implant contact, making a large surface susceptible to bacterial adhesion (9). This study therefore aimed, first, to develop an animal model to investigate osteointegration; second, to compare osteointegration of well-established biocompatible surface coatings (i.e., HA) with modified antiinfective surface coatings intended to also be used with direct bone contact (i.e., HA-Ag and CH); and third, to investigate degradation of surface coatings in vivo throughout the study period.
The present study showed that all tested implants were inserted without bacterial contamination so that results regarding osteointegration can be interpreted as depending only on the interaction between coating and host tissue and not on additional bacterial interactions. Another finding was that surface coatings with potentially local bactericidal activity (HA-Ag and CH) showed the same osteointegration as established osteoconductive HA coatings. Lastly, all 3 coatings were completely degraded over time in vivo, allowing good long-term biocompatibility and decreasing the risk of local toxicity against host cells.
The implant we chose in the present study has some important features (Fig. 1): The shape of the implant allows press-fit insertion, the most commonly used method of fixing cementless orthopedic devices. In addition, the cone-shaped profile provides sealing of the drilled hole. This could be important for future studies with our model if applied to answer questions regarding implant-associated infections. In this context, bacterial solutions could be easily inserted into the predrilled hole without the fear of losing the inoculum into the surrounding soft tissue. In several infection models, wax, fibrin glue or dental gypsum are used to seal the entry site of the application to avoid leakage of bacterial inoculum into the adjacent tissue (20–21–22). These foreign materials can interfere with locally inserted bacteria, making the model error-prone. Finally, the placement of the screw implant in the current study was decided to be in the metaphysis close to the joint. This part of the bone is very interesting for orthopedic questions regarding joint replacement, and has been used in rat models by only a few authors so far (17, 19, 20). Another important feature of the screw is that it was manufactured from titanium alloy, was treated by TPS and then coated with HA (group I), HA-Ag (group II) and CH (group III). The use of titanium alloys in surgical implants has increased in recent years, due to superior mechanical properties compared with pure titanium (23, 24). The addition of low-dose Ag was to allow sufficient osteointegration and, on the other hand, to provide potential bactericidal activity (25). Ag+ concentrations in the powder of the HA-Ag coatings of the present study were 45 ppb and therefore rather low. In this context, a previous study evaluating similar coatings showed high bactericidal activity of HA-Ag samples in vitro for a short time (16). On the other hand, contradictory data still exist regarding the reproducibility of antibacterial effects of Ag in vitro and in vivo showing that the effects in vitro do not necessarily have to be evident in vivo (26). However, such coatings have rarely been applied to the surface of implants intended for direct bone contact, due to concerns about potential toxicity. To solve this dilemma, we used HA as a support material for Ag+, because it offers good osteoconductivity and biocompatibility. CH is another promising additive to provide bactericidal potency for surface coatings. CH acts by creating an alkaline (pH 8-9) environment due to the release of hydroxides and calcium ions (27). The pH plateau created by CH is thought to stimulate local host tissue, and seems to inhibit bacterial activity. The big advantage is its short-acting local effect without systemic toxicity. Other studies have depicted antiinflammatory and analgetic features if used in on exposed bone surfaces (13, 27). As an oil or aqueous solution, CH is a broadly used substance for canal disinfection in dentistry, but so far it has not been applied on coatings used with direct bone contact in the extremities (12, 13).
In the current study, various methods were applied to evaluate the implants’ biocompatibility. We used EDX for chemical analysis of the implant as well as explant surfaces. The HA and HA-Ag coatings were composed of calcium, phosphate and oxygen. The carbon signal was probably derived from a carbon surface layer typically formed from the atmosphere during storage (28). No titanium was detected preoperatively on the implants, because these coatings were thick and closed. The CH coating mainly consisted of calcium and oxygen. On implants of group III, minor traces of titanium were found and assigned to shown as tiny uncoated spots. Similar to all groups, carbon signals were detected arising from superficial atmospheric adsorbates. On the retrieved explants, however, the surface chemistry and morphology significantly differed from the initial implant surface. All 3 coatings were completely degraded and could not be identified anymore. All explants released titanium from the subjacent TPS coating and were decorated with bone fragments.
Other studies have reported the chemical composition of implants studied using different analytical techniques, such as X-ray photoelectron spectroscopy (XPS), which makes comparison with the findings of the present study difficult (29–30–31). EDX and XPS are equally suitable for accurate determination of the mean atomic composition of surface areas. However, XPS analysis is more surface-specific and can determine the chemical state of detected elements – e.g., after chemical modification – and is more commonly used in chemical analysis of implant surfaces (32). Furthermore, the immersion of the explants in ethanol before surface analysis has to be considered. The exact chemical surface composition has to be interpreted with caution.
Biomechanical testing in the present study was carried out via a semiquantitative measuring of RTR. HA was used as the gold standard coating, since it is in widespread use in orthopedic and trauma surgery and has been extensively studied for its excellent biocompatibility. The findings suggest excellent osteointegration of the tested antibacterial coatings of groups II and III in our study (Tab. II). Comparing the macroscopically detectable bone remnants on the screw with the average score values for RTR, similar high values were found. These results suggest that both methods used to evaluate osteointegration confirmed the implant's performance in the same direction. However, several methods of determining osteointegration are described in the literature making comparison among different studies difficult (14, 29, 33, 34). Nevertheless, even if quantitative methods are used, correlations between biomechanical testing and osteointegration are not always verifiable (30, 35). In this context, histological evaluation of the bone provides important insights. Our qualitative histological observations showed evidence of osteoconduction and absence of osteomyelitis in all implant surfaces analyzed (Fig. 4C). The bone inside the threaded area of the implant appeared to be more homogenous and densely mineralized compared with the cancellous bone of the metaphysis. In contrast to other investigators, we did not see large masses of disorganized calcified material or debris between the threads (29). Ivanoff et al reported a higher incidence of inflammatory cells and nonresorbed remnants of old bone entrapped in the bone tissue in contact with machined implant surfaces, than on spark-anodized surfaces with a 10-µm-thick oxide layer (36). The results of qualitative histological observations obtained in the present study suggest that all tested surfaces were highly osteoconductive.
In summary, the model presented is suitable for studies of implant-related effects on bone. The tested materials showed early integration for all surface coatings. In particular, the antibacterial coatings tested showed excellent osteointegration in extremity bones.
Footnotes
Abbreviations
Acknowledgement
We thank Fa. Atesos (Switzerland) for providing the implants and Fa. Medicoat AG (Switzerland) for carrying out coating of implants. We sincerely thank Prof. Dr. F.E. Weber from the University of Zurich for the preparation of the histological sections.
Financial support: This study was partially funded by the Wilhelm-Sander Foundation (Fördernummer: 2009.905.2), a charitable, non-profit foundation whose purpose is to promote cancer reasearch.
Conflict of interest: The authors declare that they have no competing interests.
